BIOLOGICAL COMPOUNDS NUTRIENTS 1 Organic molecules Molecules are

BIOLOGICAL COMPOUNDS & NUTRIENTS 1

Organic molecules • Molecules are called organic compounds if they: • contain carbon-to-carbon bonds AND • are made exclusively by plants and/or animals, • (This naturally excludes compounds like CO 2, which is made by living organisms, but not exclusively— plenty-of other processes produce CO 2). 2

INORGANIC COMPOUNDS 1. Do not contain the 1. Always contain the element CARBON element carbon (except as single carbon (forming chains) atoms - CO 2 ) 2. They are usually raw 2. Made by Living materials from the organisms environment 4. Do not burn 4. Burn (Combustible) 3

ORGANIC COMPOUNDS • Contains CARBON with either a HYDROGEN or another/other CARBON/S attached to it. • CO 2: INORGANIC • CH 4 ORGANIC • CCl 4: ORGANIC • C 6 H 12 O 6: ORGANIC 4
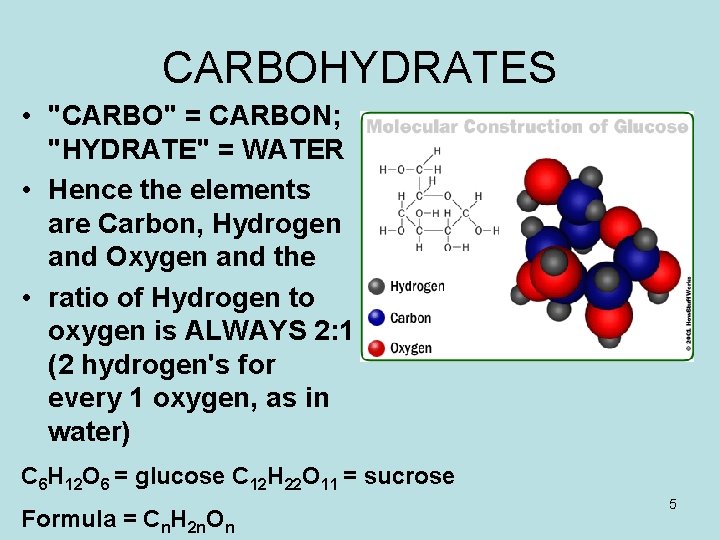
CARBOHYDRATES • "CARBO" = CARBON; "HYDRATE" = WATER • Hence the elements are Carbon, Hydrogen and Oxygen and the • ratio of Hydrogen to oxygen is ALWAYS 2: 1 (2 hydrogen's for every 1 oxygen, as in water) C 6 H 12 O 6 = glucose C 12 H 22 O 11 = sucrose Formula = Cn. H 2 n. On 5
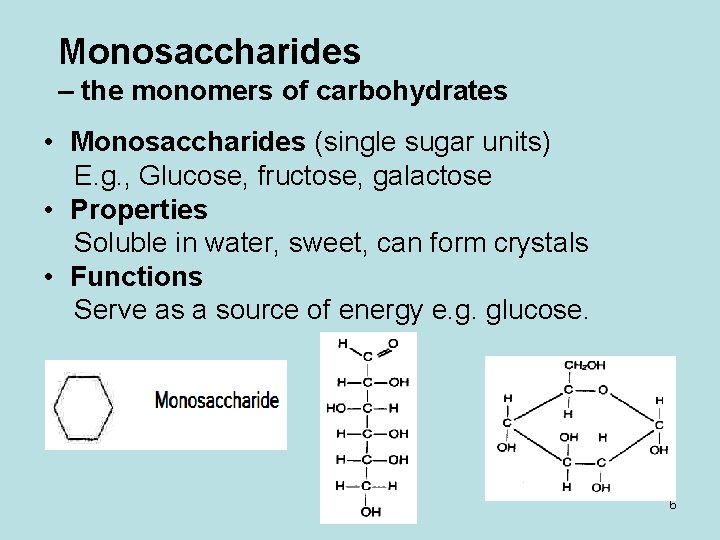
Monosaccharides – the monomers of carbohydrates • Monosaccharides (single sugar units) E. g. , Glucose, fructose, galactose • Properties Soluble in water, sweet, can form crystals • Functions Serve as a source of energy e. g. glucose. 6
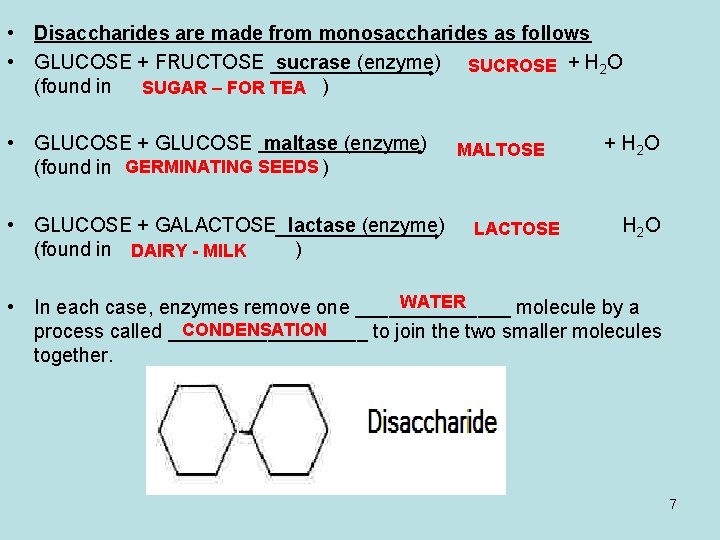
• Disaccharides are made from monosaccharides as follows • GLUCOSE + FRUCTOSE sucrase (enzyme) SUCROSE + H 2 O (found in SUGAR – FOR TEA ) • GLUCOSE + GLUCOSE maltase (enzyme) (found in GERMINATING SEEDS ) • GLUCOSE + GALACTOSE lactase (enzyme) (found in DAIRY - MILK ) MALTOSE LACTOSE + H 2 O H 2 O WATER • In each case, enzymes remove one _______ molecule by a CONDENSATION process called _________ to join the two smaller molecules together. 7
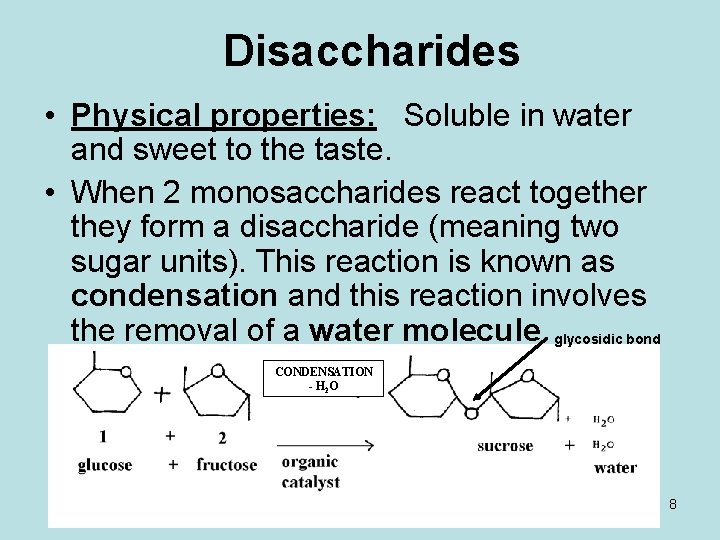
Disaccharides • Physical properties: Soluble in water and sweet to the taste. • When 2 monosaccharides react together they form a disaccharide (meaning two sugar units). This reaction is known as condensation and this reaction involves the removal of a water molecule. glycosidic bond CONDENSATION - H 2 O 8

POLYSACCAHRIDES • A polysaccharide is a POLYMER ("poly" = many), made of many single units or monosaccharides called MONOMERS • Polysaccharides are formed by condensation reactions where many monosaccharides are joined together, in a process generally called POLYMERISATION. Polysaccharides are hydrolysed (by the addition of water) into simple monosaccharides. • They are insoluble in water and are not sweet. 9

Examples of polysaccharides are • • • CELLULOSE - CELL WALLS – PRIMARY CELL WALLS –SECONDARY LIGNIN -(WATERPROOF) GLYCOGEN - IN LIVER AND MUSCLES STARCH –IN LEUCOPLASTS PECTIN - IN MIDDLE LAMELLA 10
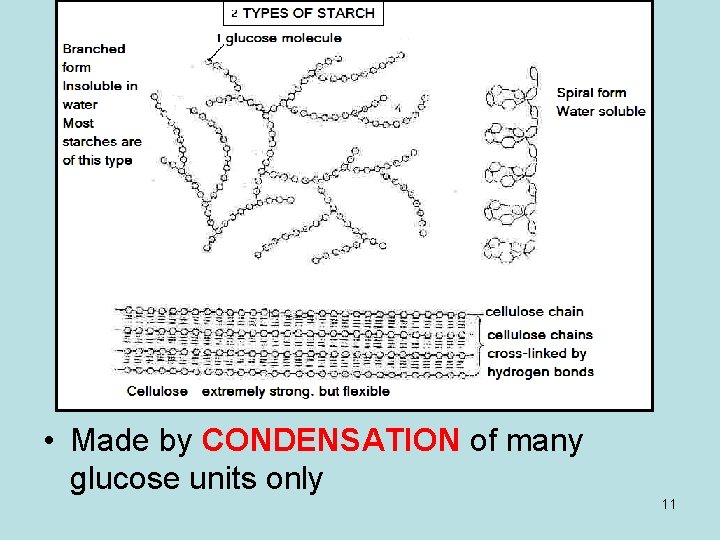
• Made by CONDENSATION of many glucose units only 11

Physical properties of Carbohydrates • Simple carbohydrates are soluble in water • Polysaccharides are generally insoluble in water • Monosaccharides and most disaccharides taste sweet 12

FOR ALL ORGANISMS CHEMICAL POTENTIAL ENERGY 1. They are a good source of ____________________ energy, such as glucose. 2. They form part of nucleic acids DNA and RNA. FOR PLANTS FOR ANIMALS 3. Plants store food as 3. Animals store food as GLYCOGEN STARCH ________ in seeds and ______in muscles and liver. bulbs. 4. Plants transport food as glucose in 4. Animals transport food as glucose BLOOD a tissue called in the ______ PHLOEM STREAM _________. 5. Carbohydrates like CELLULOSE LIGNIN CHITIN _______ & _____________ form part of the form part of cell walls. exoskeleton of insects. PECTIN CELLULOSE 6. _________ in plants 7. _________ forms an keeps cells glued together important source of roughage in our diets. 13
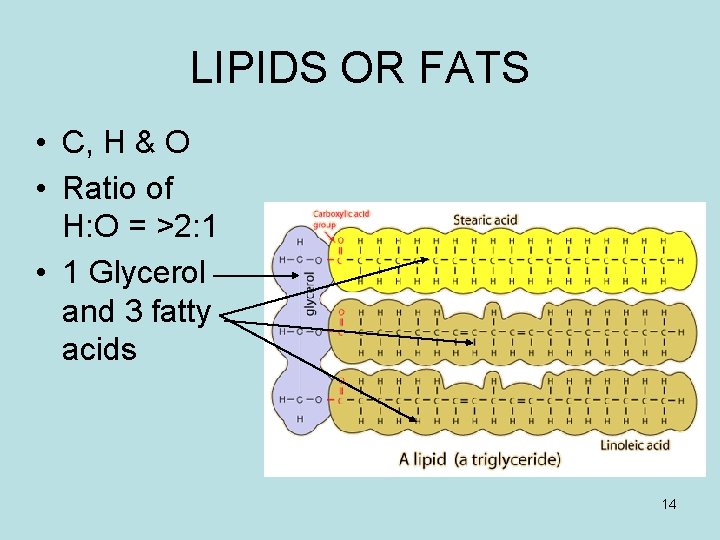
LIPIDS OR FATS • C, H & O • Ratio of H: O = >2: 1 • 1 Glycerol and 3 fatty acids 14
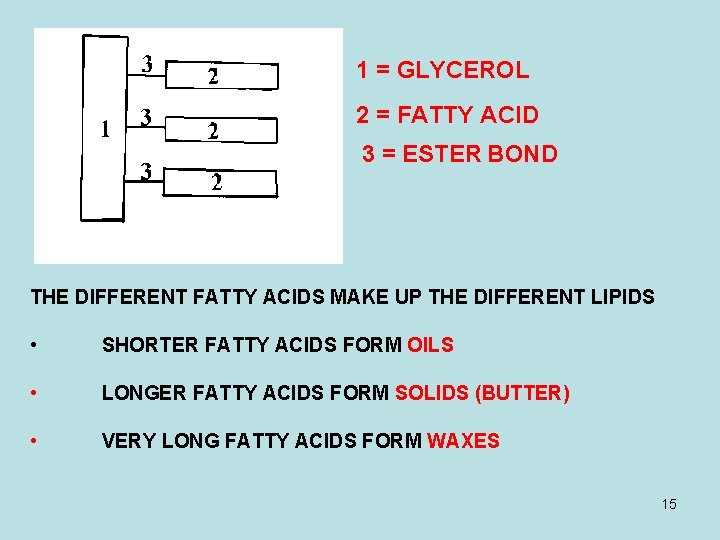
1 = GLYCEROL 2 = FATTY ACID 3 = ESTER BOND THE DIFFERENT FATTY ACIDS MAKE UP THE DIFFERENT LIPIDS • SHORTER FATTY ACIDS FORM OILS • LONGER FATTY ACIDS FORM SOLIDS (BUTTER) • VERY LONG FATTY ACIDS FORM WAXES 15
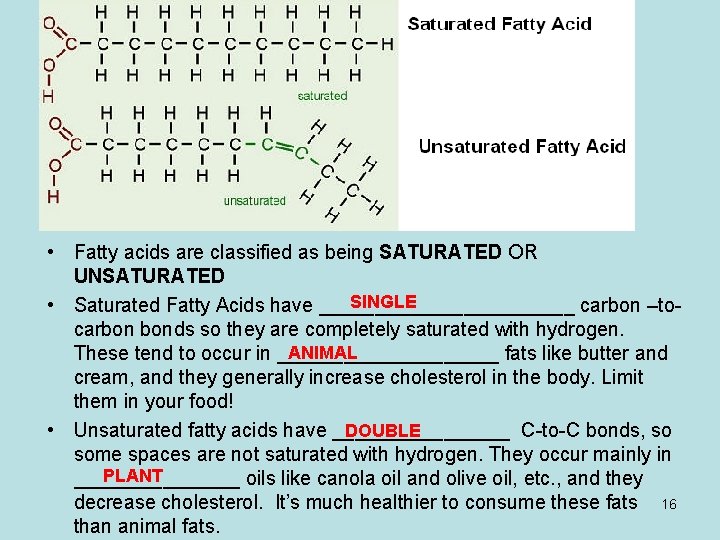
• Fatty acids are classified as being SATURATED OR UNSATURATED SINGLE • Saturated Fatty Acids have ____________ carbon –tocarbon bonds so they are completely saturated with hydrogen. ANIMAL These tend to occur in __________ fats like butter and cream, and they generally increase cholesterol in the body. Limit them in your food! • Unsaturated fatty acids have ________ C-to-C bonds, so DOUBLE some spaces are not saturated with hydrogen. They occur mainly in PLANT ________ oils like canola oil and olive oil, etc. , and they decrease cholesterol. It’s much healthier to consume these fats 16 than animal fats.
Cholesterol and heart disease: • Your body needs cholesterol to make new cells, insulate nerves and produce some LIVER hormones. Normally the _____makes all the cholesterol you need, but people also take in cholesterol with foods like EGGS MILK _____, ________ and RED MEAT ____________. 17
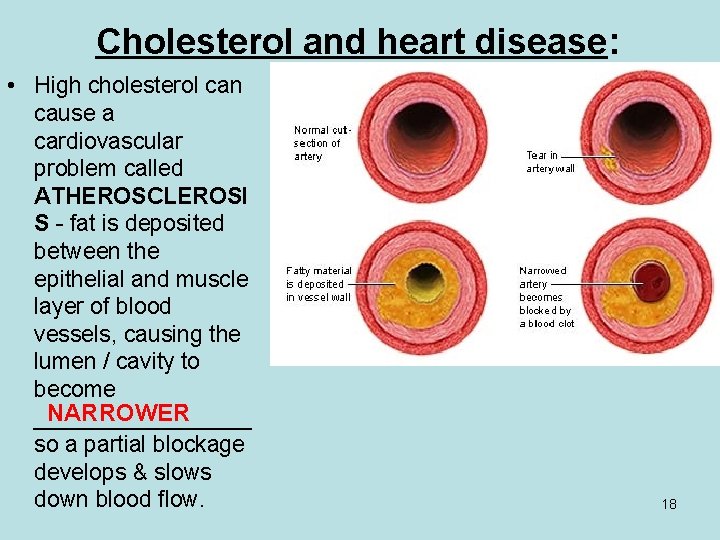
Cholesterol and heart disease: • High cholesterol can cause a cardiovascular problem called ATHEROSCLEROSI S - fat is deposited between the epithelial and muscle layer of blood vessels, causing the lumen / cavity to become NARROWER _________ so a partial blockage develops & slows down blood flow. 18

• In the heart, this causes a reduced supply of oxygen to PAIN the heart muscle, causing _____ called ANGINA HEART ATTACK • ________. A __________ will result if the obstruction causes a complete blockage. • Cholesterol is transported in the blood in 2 forms; • LDL's (Low Density Lipoproteins): also called ________ cholesterol. This contains little protein BAD and much cholesterol and Is the chief agent causing BUTTER arterial blockage (e. g. , ________, CREAM ________) • HDL's (High Density Lipoproteins): Also called ______ cholesterol. This form has lots of proteins GOOD and little cholesterol. If actually works to ______ REDUCE fatty deposits in the blood stream. 19

• REMEMBER: High cholesterol levels in the blood stream cause NO SYMPTOMS at all • Everyone over the age of 20 should have their cholesterol levels checked at least every 5 years, thereby decreasing their chances of heart disease. It's definitely also advisable to ANIMAL decrease ___________ fats and INCREASE PLANT FATS, fruit and vegetables in the diet. 20

• Fats or lipids are formed when I glycerol and 3 fatty acids are joined by enzymes by means of CONDENSATION ________________. • 1 glycerol + 3 fatty acids 1 lipid + 3 H 20 HYDROLYSIS • The process __________can be used to break the lipid down again: • 1 lipid + 3 H 20 molecules 1 glycerol + 3 fatty acids • 2. 4 Physical properties of lipids: WATER • They are insoluble in ____________. • They are soluble in ALCOHOL, ETHER, AMMONIA ___________________ etc. • They are hydrophobic, i. e. , REPEL WATER _____________. 21

Biological importance of Lipids: FOR ALL ORGANISMS CHEMICAL POTENTIAL 1. They are the best form of ________________ energy in the form of stored fats). Gram for gram, they have more than TWICE the energy in carbohydrates). CELL MEMBRANE 2. They form part of all ____________ in plants and animals. 3. Lipids are important for waterproofing leaves and insects by means of a CUTICLE waxy layer called the ________________. FOR ANIMALS PROTECT delicate organs like kidneys and eyes from injury. 1. Lipids _____ INSULATE 2. Lipids under our skin _______________ the body against heat loss in cold weather. 3. Lipids are important for the absorption of fat-soluble ________ VITAMINS like Vit. A and D. 22
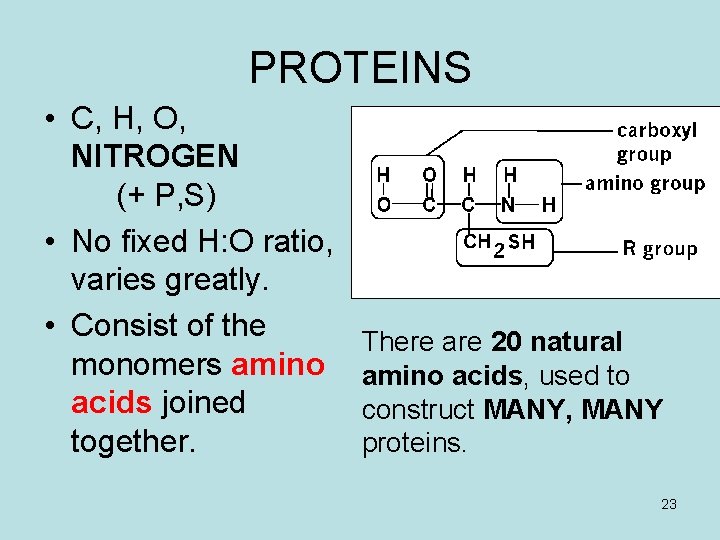
PROTEINS • C, H, O, NITROGEN (+ P, S) • No fixed H: O ratio, varies greatly. • Consist of the monomers amino acids joined together. There are 20 natural amino acids, used to construct MANY, MANY proteins. 23

Monomers of Proteins AMINO ACIDS • The __________ form the basic molecules (monomers) of which proteins are constructed. 24
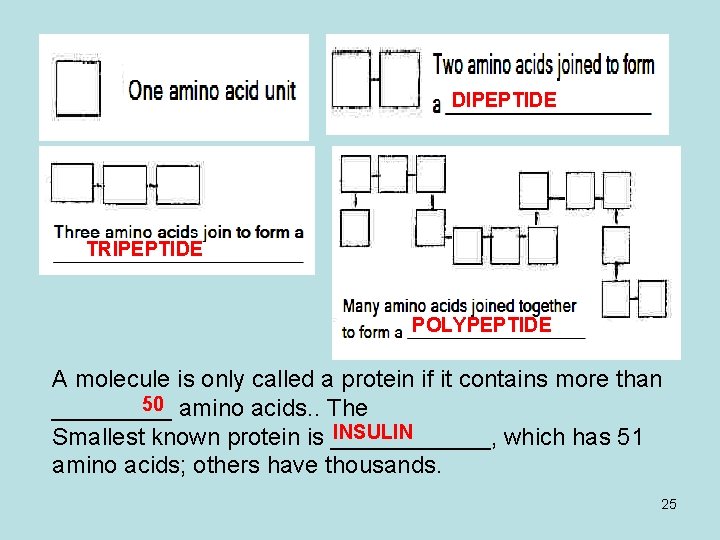
DIPEPTIDE TRIPEPTIDE POLYPEPTIDE A molecule is only called a protein if it contains more than 50 amino acids. . The _____ INSULIN Smallest known protein is ______, which has 51 amino acids; others have thousands. 25

Dipeptides • There are 20 naturally occurring amino acids which we call the essential amino acids for life. • The amino acid MONOMERS are used to build up proteins. • Amino acids join to each other by a CONDENSATION REACTION. WATER is removed in a reaction between the amino group of one amino acid and the carboxyl group of the other. • They are broken down by means of HYDROLYSIS • The bond between the amino acid molecules in a protein molecule is called a PEPTIDE BOND 26
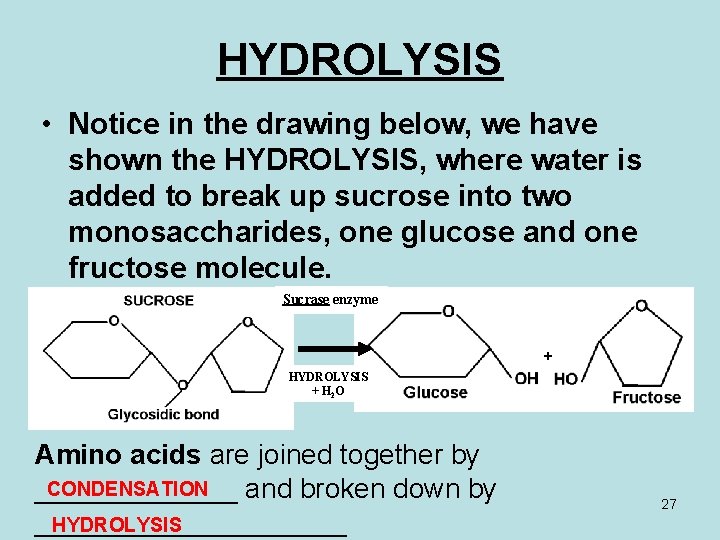
HYDROLYSIS • Notice in the drawing below, we have shown the HYDROLYSIS, where water is added to break up sucrose into two monosaccharides, one glucose and one fructose molecule. Sucrase enzyme + HYDROLYSIS + H 2 O Amino acids are joined together by CONDENSATION _______ and broken down by __________ HYDROLYSIS 27

PHYSICAL PROPERTIES - PROTEINS MACRO-MOLECULES • They are __________ (large) p. H • Sensitive to changes in _____ (acidity/alkalinity) • Sensitive to TEMPERATURE _______ (cold does not affect the structure but high temperatures can cause a breakdown in their structure called _________ DENATURATION 28
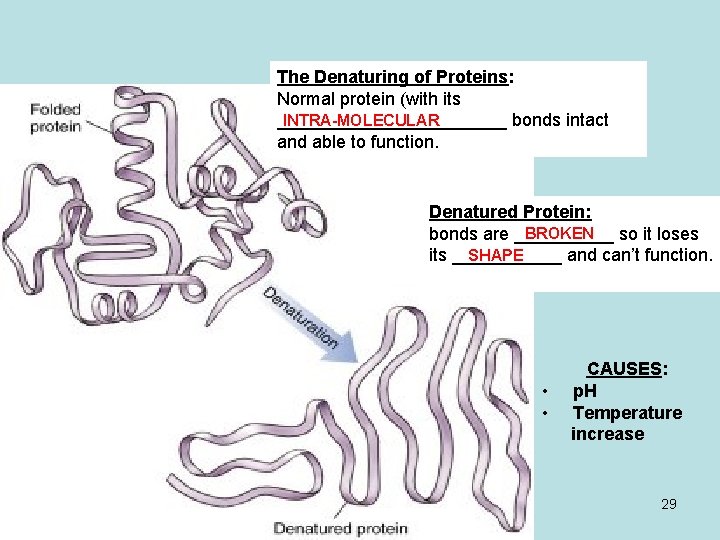
The Denaturing of Proteins: Normal protein (with its INTRA-MOLECULAR ____________ bonds intact and able to function. Denatured Protein: BROKEN bonds are _____ so it loses SHAPE its ______ and can’t function. • • CAUSES: p. H Temperature increase 29

Biological importance of Proteins: FOR ALL ORGANISMS PROTOPLASM 1. Proteins form part of ___________, the living substance in cells. CELL MEMBRANES 2. They form part of all _______, e. g. , nuclear membrane. ENZYMES speeding up biochemical reactions. 3. They act as ______, CHROMOSOMES 4. They form a structural part of _____________, together with DNA. 5. They can be used as a ____________ when carbohydrates and ENERGY SOURCE lipids are in short supply. This is for emergencies only. FOR ANIMALS 1. Some hormones like _____ are proteins (it controls blood glucose level) INSULIN 2. Proteins can be ______ like melanin or haemoglobin. 3. Some proteins act as part of the immune system, e. g. , __________ PIGMENTS in blood. ANTI-BODIES 30

Polymers • All organic compounds are built up into POLYMERS by the process of CONDENSATION ____________ and can be HYDROLYSIS broken down by _________, e. g. , during digestion. • METABOLISM consists of building up processes called _______________ and ANABOLISM breaking down processes called ________________. CATABOLISM 31
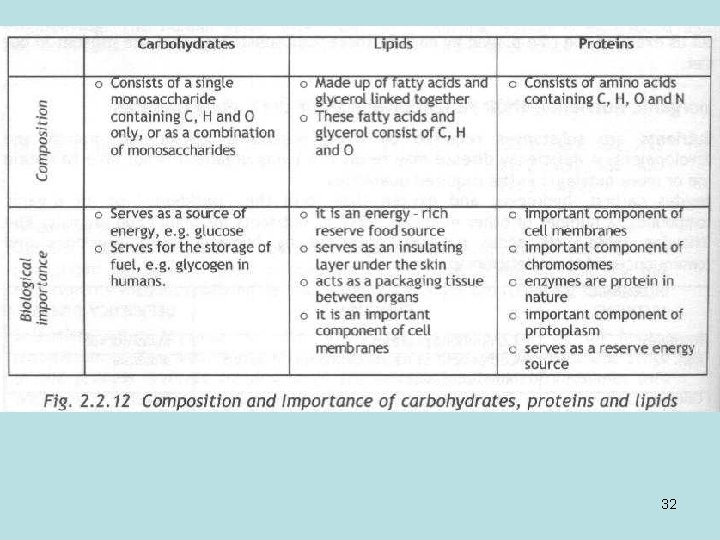
32

FOOD TESTS • All the tests described below are CONTROL TESTS to show what a positive result looks like - we will be using the actual substance we are lasting for with the appropriate method. • This will show you what a positive test looks like, so that further tests on food samples can be COMPARED with these controls. • When you are testing a food sample, DO NOT ADD THE SUBSTANCE TESTED FOR! 33

• Two procedures to be followed when testing food samples for various compounds: • • CRUSH IT OR CUT IT FINELY TO INCREASE THE SURFACE AREA OF THE REAGENT TO REACT __________________ WITH ADD WATER TO CREATE A LIQUID MEDIUM FOR THE CHEMICAL RECTIONS TO WORK IN __________________ 34

GLUCOSE TEST • • • Place a small amount of glucose powder (or food) into a test tube. Addwater & shake it. Light a Bunsen burner. Add a few drops of BENEDICTS SOLUTION to the mixture and shake. Use a test tube holder to heat the solution over the flame, moving it constantly and keeping the open end of the test tube away from people. (Heat Is used here to SPEED UP THE RATE OF THE REACTION __________________________ Record colour changes. DO NOT overheat the mixture - stop when it boils, RESULT: No glucose BLUE some glucose GREEN more glucose YELLOW lots of glucose REDDISH-ORANGE CONCLUSION IF A SUBSTANCE TURNS ORANGE IN THE PRESENCE OF BENEDICT’S SOLUTION AND HEAT, IT CONTAINS GLUCOSE 35

TO TEST FOR THE PRESENCE OF STARCH: • Place a small amount of starch powder (or food) Into a test tube and add a little water. Shake it. No heat is needed - why? THE REACTION IS FAST ENOUGH ______________ • Add a few drops of IODINE SOLUTION and shake it. • Record colour changes. • RESULT: BROWN IODINE CHANGES TO BLUE-BLACK • ___________________________ • CONCLUSION: ANY FOOD THAT TURNS FROM BROWN TO BLUE-BLACK IN THE OF IODINE CONTAINS STARCH. • PRESENCE ___________________________ 36

Biological Compounds Page 9 • All organic compounds are built up into POLYMERS by the process of CONDENSATION ____________ and can be HYDROLYSIS broken down by _________, e. g. , during digestion. • METABOLISM consists of building up processes called _______________ and ANABOLISM breaking down processes called CATABOLISM ________________. 37
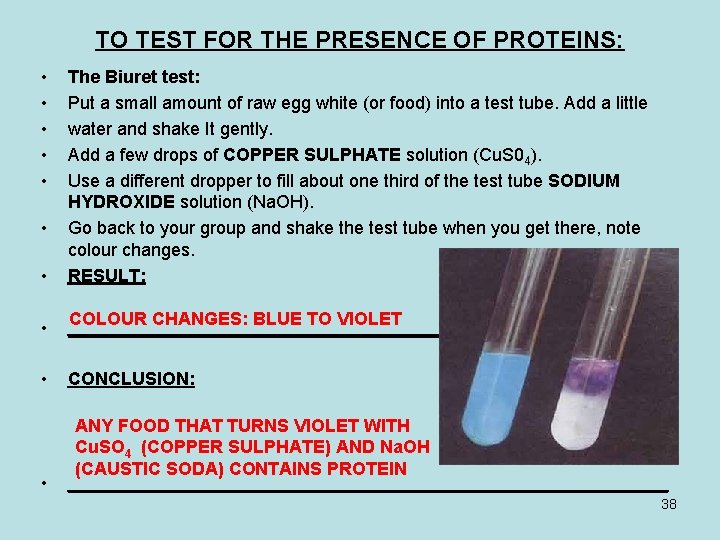
TO TEST FOR THE PRESENCE OF PROTEINS: • • • The Biuret test: Put a small amount of raw egg white (or food) into a test tube. Add a little water and shake It gently. Add a few drops of COPPER SULPHATE solution (Cu. S 04). Use a different dropper to fill about one third of the test tube SODIUM HYDROXIDE solution (Na. OH). Go back to your group and shake the test tube when you get there, note colour changes. RESULT: • COLOUR CHANGES: BLUE TO VIOLET ______________________________ • CONCLUSION: • ANY FOOD THAT TURNS VIOLET WITH Cu. SO 4 (COPPER SULPHATE) AND Na. OH (CAUSTIC SODA) CONTAINS PROTEIN ______________________________ • 38

Page 11 FOOD TEST Glucose Starch Proteins Biuret test REAGEN T HEAT? Positive test: Colour? Negative test: Colour? BLUE BENEDICTS YES GR/YE/OR IODINE NO BLUE-BLACK BROWN NO MAUVE/ PURPLE Cu. SO 4 + Na. OH NONE 39

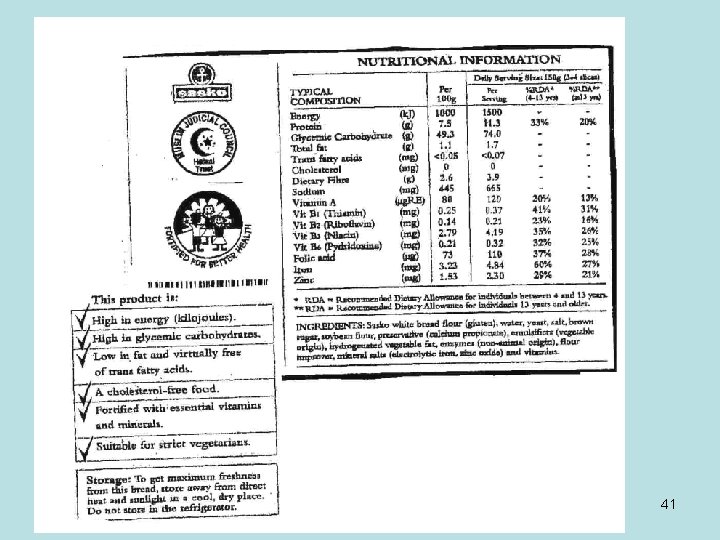
Food labels • To compare different foods, use the Nutritional content per 100 g (this is also easily converted into % (30 g per 100 g = 30% • Keep fat content to a minimum • Water is seldom included (if it adds up to less than 100 g, the remainder is water). • Carbohydrates include only digestible (starch, sugar), not fibre, cellulose • Go for fewer refined carbs (like sugars) 40
41
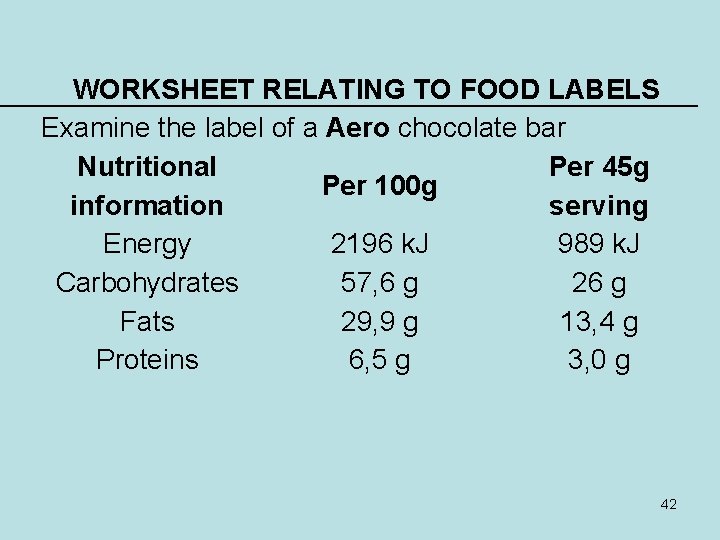
WORKSHEET RELATING TO FOOD LABELS Examine the label of a Aero chocolate bar Nutritional Per 45 g Per 100 g information serving Energy 2196 k. J 989 k. J Carbohydrates 57, 6 g 26 g Fats 29, 9 g 13, 4 g Proteins 6, 5 g 3, 0 g 42

1. Which compound forms the majority of this food item? _________ (1) CARBOHYDRATES 2. What is the main specific carbohydrate in an Aero bar likely to be? AERO IS A CHOCOLATE AND IS MADE (2) • LACTOSE. ___________________ OF DAIRY AND ADDED SUGAR (SUCROSE) 3 Use the serving size to calculate the percentage fats in this food item. Show your working. 29, 9/100 X 100 = 0, 299 X 100 = 29, 9% (OR 29, 9 G OF 100 G = 29, 9%) (3) 4 Do you think that these fats are likely to be saturated or unsaturated? Give a reason for your answer. SATURATED: CHOCOLATE CONTAINS CREAM WHICH CONTAINS SATURATED FATS 43 (2)

• If a person needs 150 g of protein in a day to maintain her body, how many of these bars should she consume daily if she eats nothing else? Give your answer to the nearest whole bar. 150 G/3 G = 50 BARS. _______________________ (3) • Would you suggest that she eats Aero bars in order to obtain the correct protein content in her diet? Give a reason for your answer. NO WAY! ALTHOUGHS HE GETS THE REQUIRED PROTEINS SHE WILL EAT TOO MUCH FAT AND CARBOHYDRATES. • ______________________(3) • Calculate the percentage water in this food item 100 – (57, 6 + 29, 9 + 6, 5) = 6/100 = 6% • _____________________ (2) 44

Vitamins: Group of complex organic compounds, needed in small quantities that help your body maintain normal metabolism, growth, and development. Vitamins mainly made by plants and animals obtain theirs from the food they eat.

• They have no energy value - taking a vitamin supplement can't "give you energy", but can optimise certain functions like energy release from food. • They act as important REGULATORS in body processes and are essential in the maintenance of good health. 46

Vitamins Two Groups of Vitamins: • Water-Soluble • Fat-Soluble Water-Soluble Vitamins: • Vitamins that dissolve in water and are NOT stored in your body for future use Vitamin B and Vitamin C

Vitamins Fat-Soluble Vitamins: • Vitamins that dissolve into and are transported by fat • They can be stored in fat tissue, the liver, and the kidneys. • Vitamins A, D, E, and K

Examples of Fat-Soluble Vitamins: • Vitamin A: • Essential forming of a pigment in the rods of the eye (retina) to see in dim light. Maintains good vision, promotes body cell growth, helps protect teeth – Food sources: green vegetables (carrots, spinach, gem squash), dairy products – Deficiency diseases: Nightblindness

Vitamin B 3: Acts as a co-enzyme in cell respiration, assisting energy release from food. – Food sources: Red meat, unrefined cereals, Marmite, Bovril – Deficiency disease: Pellagra – skin rashes, redness, swollen red tongue, diarrhea
Examples of Water-Soluble Vitamins: • Vitamin C: Essential formation of collagen and plays a role in immunity (fights against infection, maintains healthy gums, strengthens and maintains blood vessel structure) – Food sources: citrus fruits, tomatoes, strawberry, pineapple, guava, leafy vegetables Deficiency disease: Scurvy & anaemia Bleeding gums, poor wound healing, frequent infections

• Vitamin D: Essential for absorption of calcium and phosphorus and thus hardening bones and teeth Food sources: eggs, cod liver oil, salmon, butter, margarine, fortified breakfast cereal. Other sources include sunlight Deficiency disease: Rickets (children) - soft bones causing bow-legs or knock knees

• Vitamins found in all the major food groups but, NO ONE FOOD CONTAINS ALL THE REQUIRED VITAMINS. • A balanced diet is the best way to ensure that one gets all the necessary vitamins. 53

IMPORTANCE OF WATER IN NUTRITION PHOTOSYNTHESIS 1. RAW MATERIAL FOR ____________ (and a waste product of cellular respiration) 2. CHEMICAL REACTIONS IN CELLS ALWAYS TAKE PLACE IN THE MEDIUM OF WATER. Water acts as a SOLVENT ___________. Ions, atoms and molecules are SOLUTES which dissolve in water before being able to react. 3. WATER CAN BE INVOLVED IN A CHEMICAL HYDROLYSIS REACTION CALLED ________ ("hydro" = water; "lysis" = to dissolve or break down Water actually is involved in the reaction (usually digestion) and it splits a larger molecule into smaller molecules. e. g. Maltose + water = glucose + glucose maltase (enzyme) 54

A TRANSPORT 4. WATER IS _____________ MEDIUM FOR SUBSTANCES, e. g. foods, gases and waste products in the blood circulation of animals or in the phloem of plants. 5. WATER EVAPORATES FROM THE SKIN AND THEREFORE COOLS THE BODY (IT HELPS THE BODY TO REGULATE ITS TEMPERATURE) 6. WATER ACTS AS A LUBRICANT ____________, REDUCING FRICTION, e. g. in the joints of the body, allowing freer movement. 55

MINERALS • 20, inorganic, NOT MADE BY PLANTS OR ANIMALS • Macro-elements are elements required in LARGE QUANTITIES BY AN ORGANISM. • Micro-elements are elements required in SMALL QUANTITIES BY AN ORGANISM (they are also known as TRACE ELEMENTS). 56
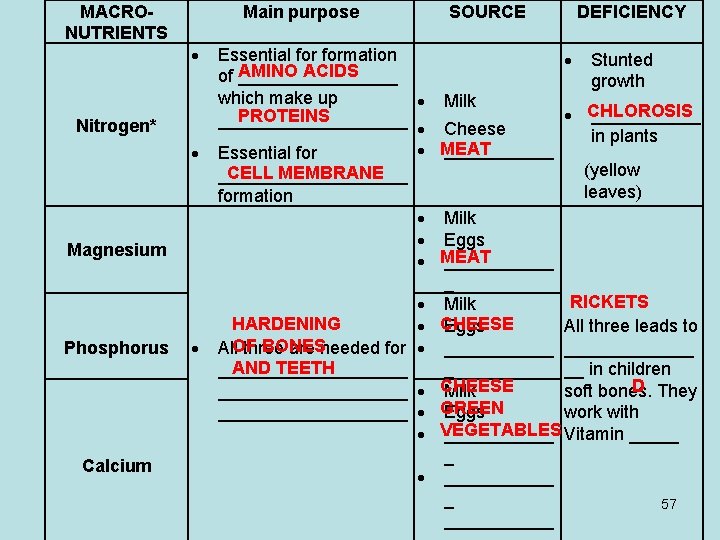
MACRONUTRIENTS Main purpose Nitrogen* Magnesium Phosphorus Calcium SOURCE DEFICIENCY Essential formation Stunted AMINO ACIDS of ________ growth which make up Milk CHLOROSIS ______ PROTEINS __________ Cheese in plants MEAT ______ Essential for (yellow CELL MEMBRANE __________ leaves) formation Milk Eggs MEAT ______ _ RICKETS Milk HARDENING All three leads to CHEESE Eggs BONES All. OF three are needed for _____________ AND TEETH __________ __ in children _ D They __________ CHEESE soft bones. Milk __________ GREEN Eggs work with VEGETABLES ______ Vitamin _____ _ ______ _ 57 ______
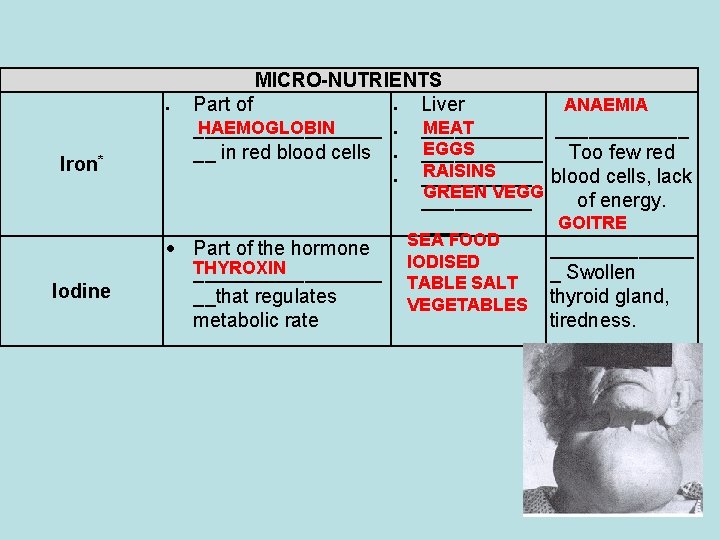
MICRO-NUTRIENTS Part of Liver ANAEMIA HAEMOGLOBIN MEAT ____________ EGGS __ in red blood cells ______ Too few red RAISINS _____ blood cells, lack GREEN VEGG _____ of energy. GOITRE ____ SEA FOOD Part of the hormone _______ IODISED THYROXIN _________ _ Swollen TABLE SALT __that regulates VEGETABLES thyroid gland, metabolic rate tiredness. Iron* Iodine 58
- Slides: 58