BIOLOGICAL AND BIOORGANIC CHEMISTRY What is a bioorganic

BIOLOGICAL AND BIOORGANIC CHEMISTRY

What is a bioorganic chemistry? Bioorganic chemistry studies the chemistry of organic biomolecules Bioorganic chemistry is a science that investigates and explains: Ø relationship between the structure of organic compounds and their biological activity Ø relationship between the reactivity of organic compounds and their role in functioning of biological systems
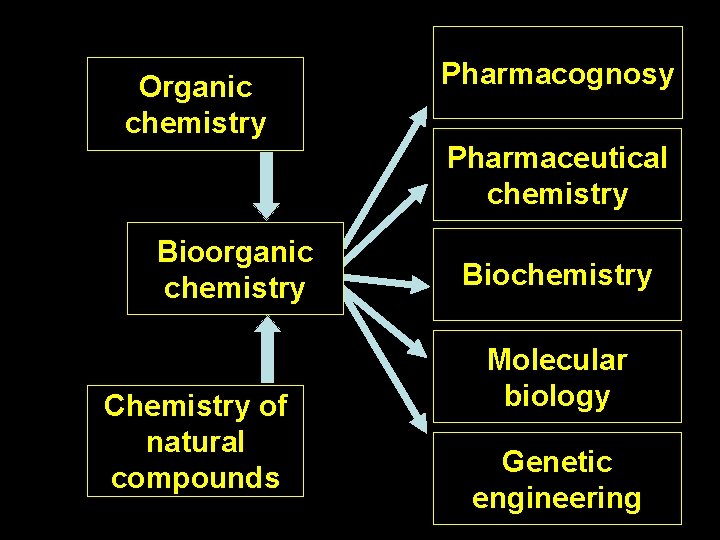
Organic chemistry Pharmacognosy Pharmaceutical chemistry Bioorganic chemistry Chemistry of natural compounds Biochemistry Molecular biology Genetic engineering

What do we have to study? Ø to recall and to repeat bases of organic chemistry (school or lyceum course) Ø to expand the knowledge on structure of organic compounds • isomerism of organic compounds • homology and homological series • nomenclature of organic compounds Ø to study the reactivity of organic compounds • mechanisms of chemical reactions • some reactions typical for important homological series of organic compounds
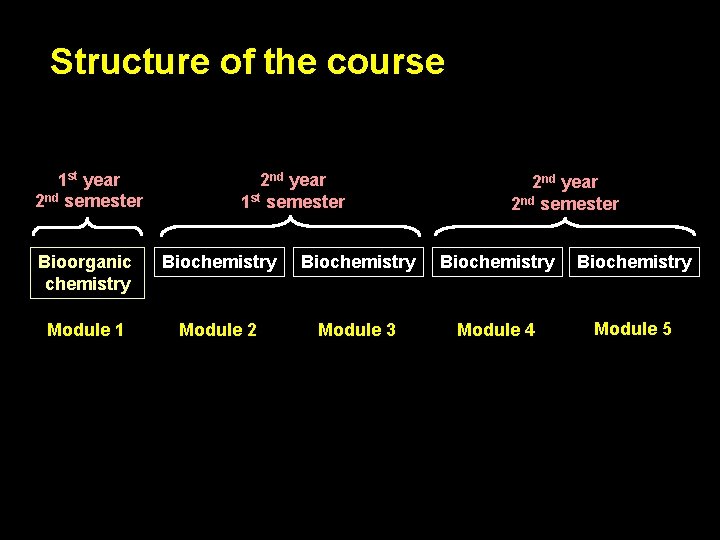
Structure of the course 1 st year 2 nd semester 2 nd year 1 st semester 2 nd year 2 nd semester Bioorganic chemistry Biochemistry Module 1 Module 2 Module 3 Module 4 Module 5

Structure of the course TIMETABLE 1 st year 2 nd semester Bioorganic chemistry Module 401 hours MARKS excellent 5 lectures = 10 hours 9 seminars = 18 hours 5 practices = 10 hours module final = test 180 – 200 points good 150 – 179 points 2 hours satisfactory 120 – 149 points MARKS Semester tests (3) – 40 points max, total 120 points Final test – 40 points max. Practice test (1) – 40 points max Final test – from 10 to 40 points max. unsatisfactory < 120 points

Structure of the course v Lectures Lecture 1 Introduction to organic and biological chemistry. Physicochemical bases of organic and bioorganic chemistry. Mechanisms of formation and breaking of chemical bonds. Homolysis and heterolysis. Lecture 2 Basic terms, statements and rules in organic and bioorganic chemistry. Isomerism of organic molecule. Lecture 3 Chemical nomenclature. Naming compounds in the organic and bioorganic chemistry (I). Lecture 4 Chemical nomenclature. Naming compounds in the organic and bioorganic chemistry (II). Mechanisms of chemical reactions (I). Lecture 5 Mechanisms of chemical reactions (II and III).

Structure of the course v Seminars Seminar 0 Introduction to the Course. Rules and grading criteria. History of organic and bioorganic chemistry. Nature of chemical bonds. Seminar 1 Classification and nomenclature of organic compounds. Saturated hydrocarbons – alkanes and cycloalkanes. Seminar 2 Unsaturated aliphatic hydrocarbons – alkenes and alkynes. Polymerization. Seminar 3 Arenes and their derivatives. Seminar 4 Alcohols and ethers. Seminar 5 Carbonyl compounds and reactions of carbonyl groups. Aldehydes and ketones. Carbohydrates. Seminar 6 Carboxylic acids and esters. Seminar 7 Amines and nitro derivatives. Aminoacids. Рeptides. Seminar 8 Heterocyclic compounds.

Teachers Instructors Full and assistant professors Alexander Roshal (lecturer) Ludmila Chepeleva Alexander Nikishin Anton Zakharov Oleg Alexey Tkachenko Kolomoitsev Lesya Kulyk References F. Bettelheim, W. H. Brown, M. K. Campbell, Sh. O. Farrell, O. Torres. Introduction to General, Organic and Biochemistry, 10 th Edition. – 2013 (or previous years of the edition – 2010, 2007) Maxim Kolosov

Our site http: //chemo. univer. kharkov. ua/medical/index. htm

Chemical bonds in organic compounds

Chemical bonds in organic compounds Ø covalent bond coordinative Ø ionic bond (dative, donor-acceptor) bond coordinative Ø metallic bond Ø hydrogen bond
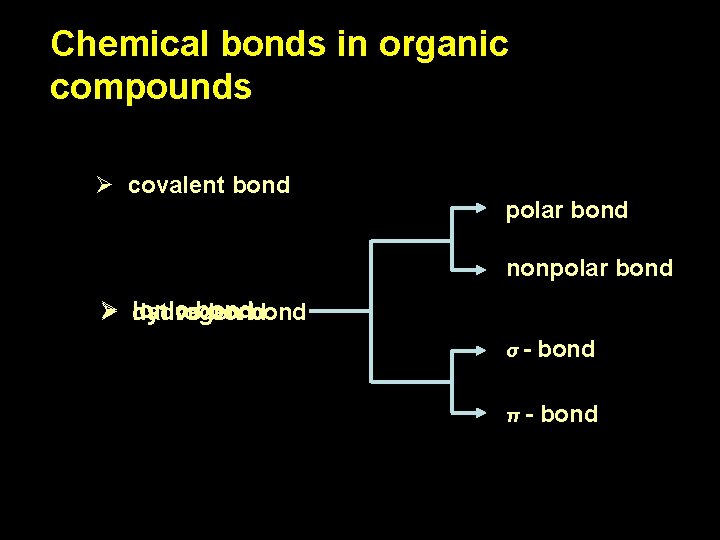
Chemical bonds in organic compounds Ø covalent bond polar bond nonpolar bond Ø ionic dativebond hydrogen bond σπ bond - bond

Mechanism of chemical bonds formation Chemical bonds form in two stages: v Impact of atoms v Redistribution of electronic density
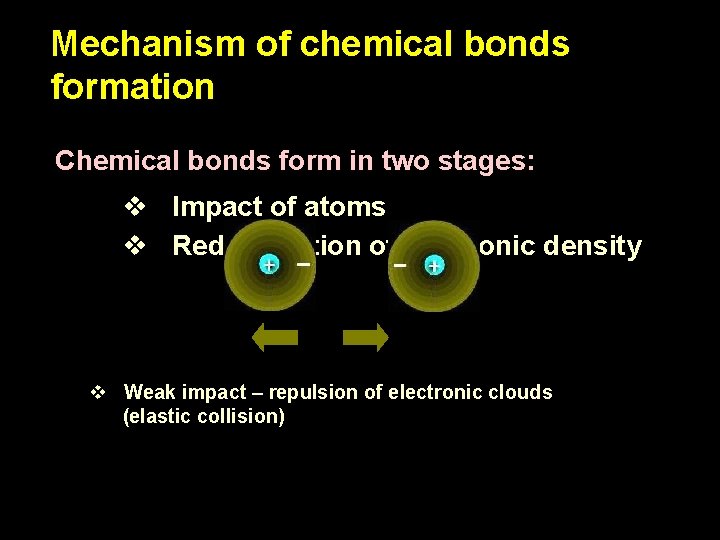
Mechanism of chemical bonds formation Chemical bonds form in two stages: v Impact of atoms v Redistribution of electronic density v Weak impact – repulsion of electronic clouds (elastic collision)
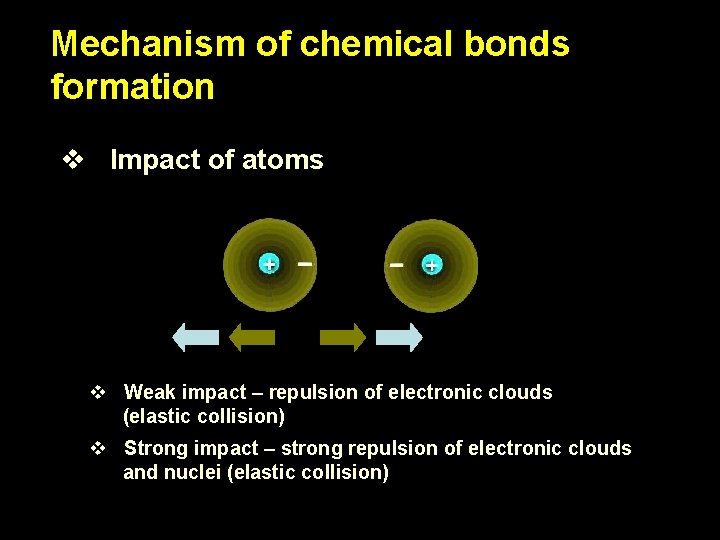
Mechanism of chemical bonds formation v Impact of atoms v Weak impact – repulsion of electronic clouds (elastic collision) v Strong impact – strong repulsion of electronic clouds and nuclei (elastic collision)
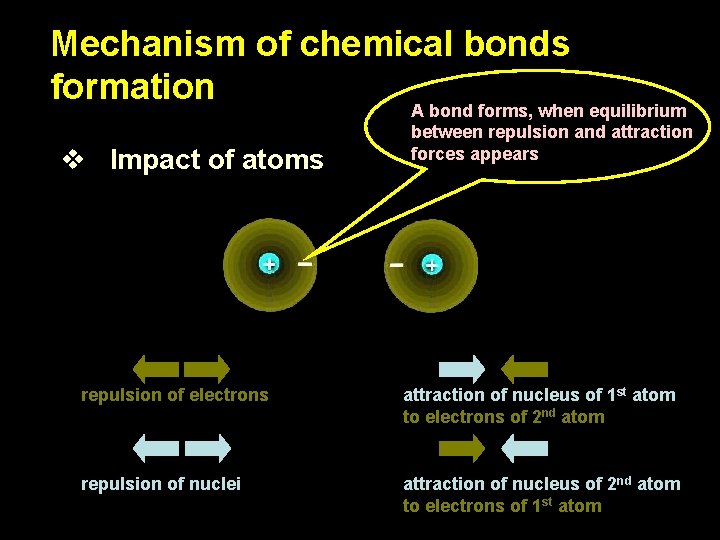
Mechanism of chemical bonds formation A bond forms, when equilibrium v Impact of atoms between repulsion and attraction forces appears repulsion of electrons attraction of nucleus of 1 st atom to electrons of 2 nd atom repulsion of nuclei attraction of nucleus of 2 nd atom to electrons of 1 st atom
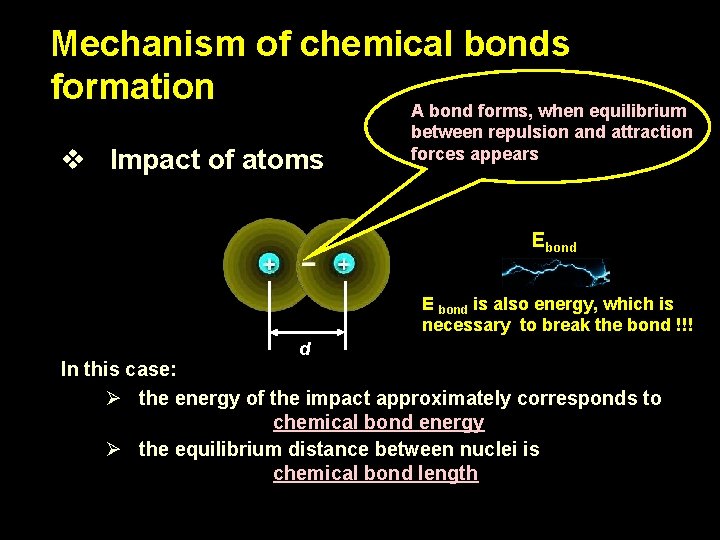
Mechanism of chemical bonds formation A bond forms, when equilibrium v Impact of atoms between repulsion and attraction forces appears Ebond E bond is also energy, which is necessary to break the bond !!! d In this case: Ø the energy of the impact approximately corresponds to chemical bond energy Ø the equilibrium distance between nuclei is chemical bond length
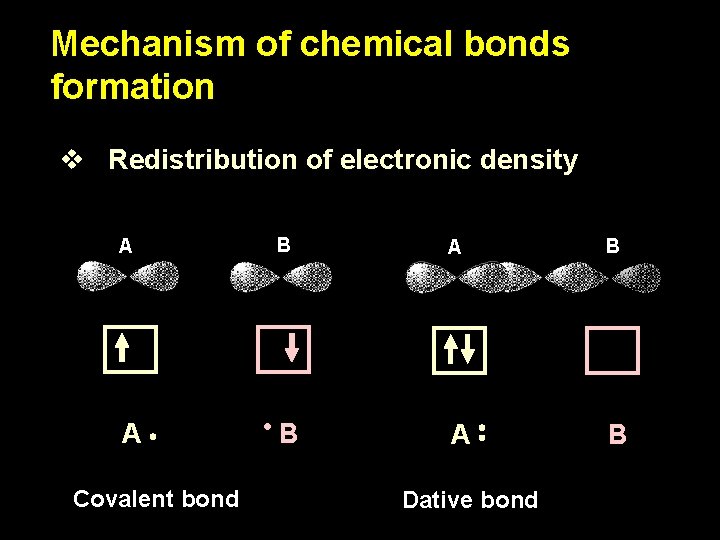
Mechanism of chemical bonds formation v Redistribution of electronic density A B A B Covalent bond Dative bond
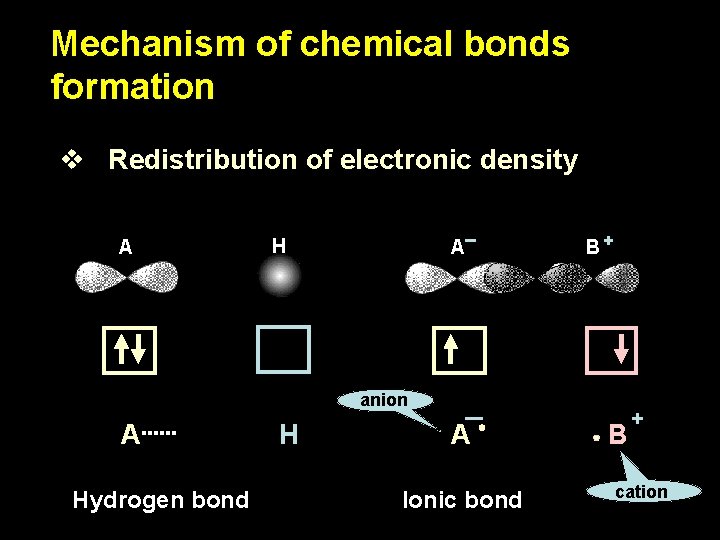
Mechanism of chemical bonds formation v Redistribution of electronic density A H A B+ anion A Hydrogen bond H A Ionic bond B + cation

Mechanism of chemical bonds formation Type and polarity of the bonds

Mechanism of chemical bonds formation Type and polarity of the bonds If the difference of atom negativities is: equal to zero One obtains: Atoms of high electronegativity covalent nonpolar bond Atoms of low electronegativity metallic bond dative bond less than 2. 0 covalent polar bond hydrogen bond (between H and N, O, F) more than 2. 0 ionic bond
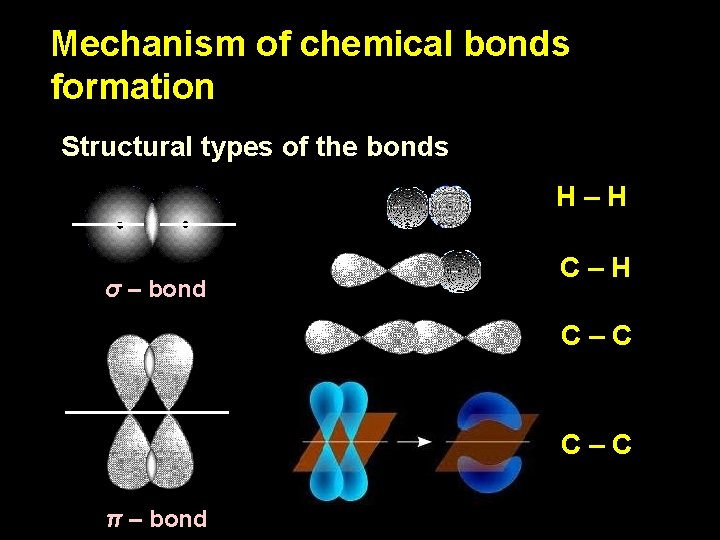
Mechanism of chemical bonds formation Structural types of the bonds H–H σ – bond C–H C–C π – bond

Mechanism of chemical bonds formation Structural types of the bonds The stability and rigidity of a bond depends on the volume of overlapping space between orbitals The greater overlapping space between orbitals, the stronger is a bond σ – bond has one big overlapping space, π- – bond has two little overlapping spaces. σ – bonds are substantially stronger than π – bonds

Mechanism of chemical bonds formation σ – bonds are substantially stronger than π – bonds 1. All the atomic chains in organic molecules (molecular “skeleton”) are constructed with σ – bonds. 2. π – bonds can be “in addition” to σ – bonds, can accompany them. 3. In chemical reactions, weak π – bonds are breaking always first. a. Substances with π – bonds are more chemically active, than substances without π – bonds. b. The greater number of π – bonds in a molecule, the greater chemical activity of corresponding substance. 3. Since π – bond-containing compounds have higher reactivity, their biological activity is also higher.
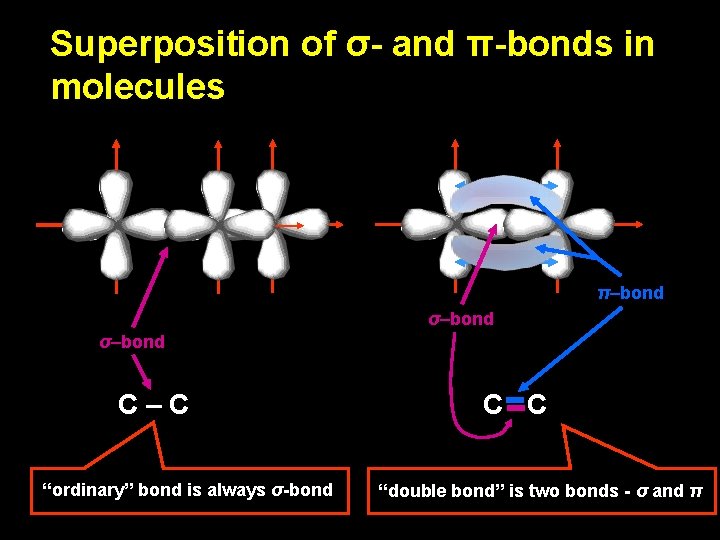
Superposition of σ- and π-bonds in molecules π–bond σ–bond C–C “ordinary” bond is always σ-bond C C “double bond” is two bonds - σ and π
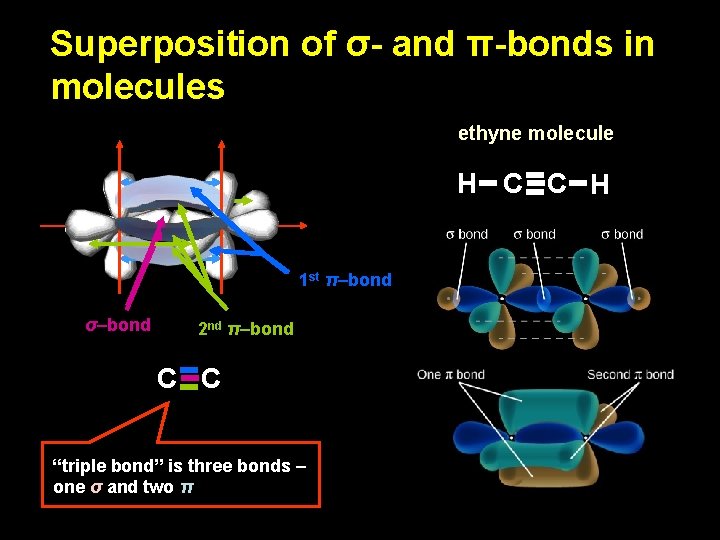
Superposition of σ- and π-bonds in molecules ethyne molecule H C C H 1 st π–bond σ–bond 2 nd π–bond C C “triple bond” is three bonds – one σ and two π

“Aromatic” π-bonds Covalent π – bonds can be formed not only by 2 electrons, but also by a group of electrons which number corresponds to Huckel rule - 4 n+2 Multielectron π – bonds are called “aromatic” bonds The greater number of electrons constructing a π – bond, the stronger this π – bonds. That is why, aromatic 6 -electron π – bonds are stronger than usual 2 -electron π – bonds.
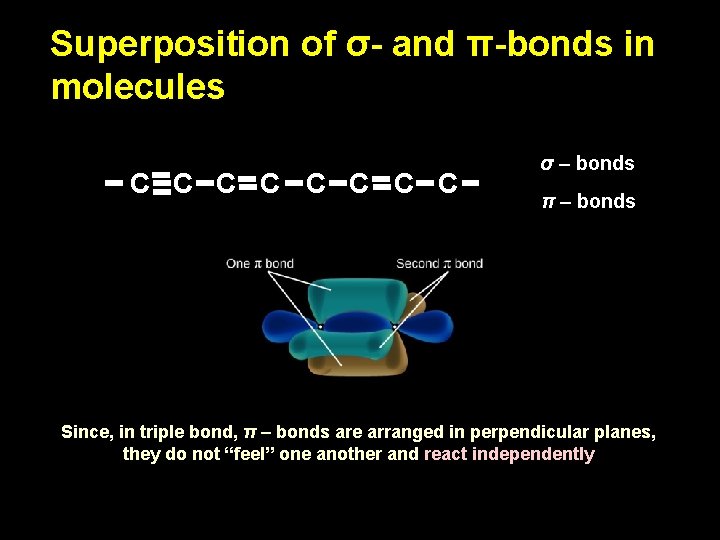
Superposition of σ- and π-bonds in molecules C C C C σ – bonds π – bonds Since, in triple bond, π – bonds are arranged in perpendicular planes, they do not “feel” one another and react independently

Chemical bond breaking Homolysis A B A + B free radicals Homolysis is typical for nonpolar bonds. To initiate homolysis in the case of polar bonds, one needs a special reagent – initiator, which helps to generate free radicals. Reactions with a homolysis stage are called radical reactions.

Chemical bond breaking Heterolysis A B A - + B + ions Heterolysis is typical for polar bonds. To initiate heterolysis in the case of nonpolar bonds, one needs a special reagent – catalyst, which helps to generate ions. Reactions with a heterolysis stage are called ionic reactions.

Thank you for your attention! http: //chemo. univer. kharkov. ua/medical/index. htm
- Slides: 33