Biologic Therapy for Atopic Dermatitis DAYNA LUCUABFEGURGUR PGY
Biologic Therapy for Atopic Dermatitis DAYNA LUCUAB-FEGURGUR PGY 1 9/17/14

Outline �Case Presentation �Introduction/Background Information �Clinical Question �Journal Article �Critical Appraisal �Discussion �Limitations �Conclusion

Case Presentation � 36 y. o. F with a PMH significant for moderate-severe asthma, eczema, chronic sinusitis, and bipolar disorder who presents for follow-up and refill meds. � Pt was recently seen in the ED x 3 in the past month for exacerbations of eczema (pruritis not responsive to betamethasone cream) and given hydroxyzine with no relief. She completed a course of prednisone prescribed by ED (20 mg x 8 days) that helped her rash. � Currently taking cetirizine, hydroxyzine prn and using betamethasone 0. 05%

Case Presentation � Subjective: She complains of persistent itching that is poorly controlled with cream and requesting a prescription for prednisone “just in case”. She also reports nightly asthma symptoms with use of xopenex inhaler 4 x/day in addition to daily advair, montelukast, and qhs albuterol nebulizer tx. � Objective: VS normal. Skin: Slightly erythematous maculopapular rash on flexor surface of arms bilaterally, upper back, and popliteal fossae with areas of significant hypopigmentation. Pulm: Diffuse wheezing in posterior lung fields. � A/P was discussed with attending and pt was continued on betamethasone and antihistamines. She was not provided with steroids as she was scheduled for follow-up with dermatology and pulmonology within a month.

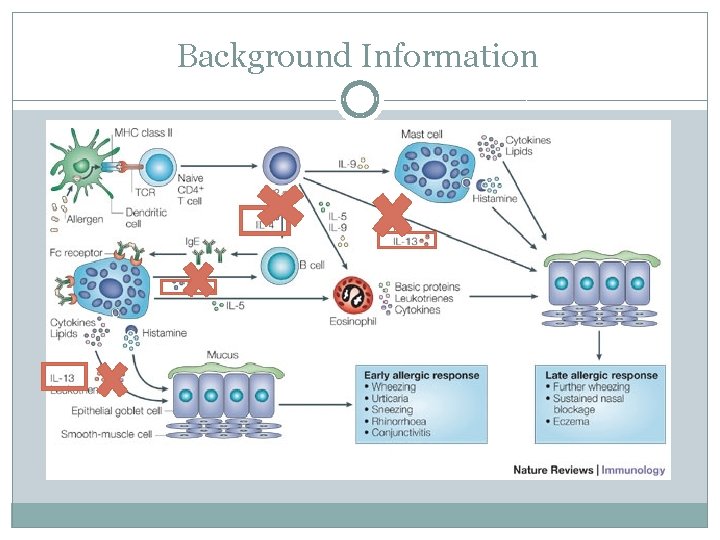
Introduction � Atopic Dermatitis is characterized by disruption of the skin barrier, type 2 helper T-cell-mediated immune responses to environtmental antigens, intractable pruritis, and susceptibility to cutaneous infections � Worldwide prevalence of 1 -20% � ~20% of patients have moderate-to-severe disease � FDA approved tx include topical glucocorticoids and calcineurin inhibitors, which have limited efficacy in moderate-to-severe disease

Background Information Study found that Dupilumab (anti-IL-4) therapy in patients with moderate-to-severe asthma with elevated eosinophils who used inhaled steroids and LABAs, resulted in fewer asthma exacerbations, improved lung function, and reduced levels of TH 2 inflammatory markers compared with placebo when steroids and LABAs were withdrawn.
Background Information

Clinical Question �Is there additional/alternative therapy that can be offered to patients with moderate-to-severe atopic dermatitis that is refractory to topical agents?

Journal Article

Objective �Beck, et. al conducted a study to determine if Dupilumab, a fully human monoclonal antibody against IL-4 and IL-13, could potentially help in the treatment of atopic dermatitis by blocking key drivers of Th 2 -mediated inflammation in comparison to placebo.
Study Design � Randomized, Placebo-controlled Trials � Double-blinded � Four Separate Studies 3 early-phase studies designed primarily to assess safety, but all trials included clinical endpoints as part of study design 2 with dose-escalation designs, two with parallel-group design � Monotherapy: 4 weeks, phase 1, sequential, dose-escalation cohorts (M 4 A in US and M 4 B Multi-national study) 22 and 37 participants, respectively � Monotherapy: 12 weeks, parallel-group cohort (M 12 study in Europe) 31 participants � Combination Therapy: 4 weeks, phase 2 a parallel-group cohort (C 4 study in Europe) 109 participants

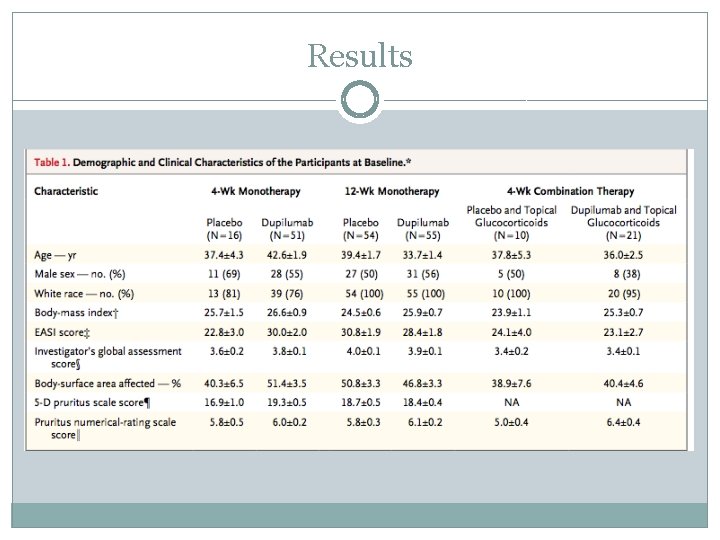
Study Sample Inclusion Criteria Exclusion Criteria 18 years or older Prior tx with Dupilumab (M 4 B, C 4) Moderate-to-severe atopic dermatitis defined by GAS of ≥ 3 Various lab values (e. g. , Hgb <11, Plt <125, WBC <3. 5) Proportion of involved BSA (variable: ≥ 15% in M 4 A, ≥ 10% in M 4 B, M 12, and C 4) Tx with systemic glucocorticoids within 4 weeks before baseline visit SCORAD >20 in C 4 Tx with allergen immunotherapy within 6 months EASI ≥ 12 in M 4 A and M 4 B Tx with topical agents within 1 week for monotherapy studies EASI ≥ 16 in M 12 Systemic tx w/ cyclosporine, Mycophenolate, IFN-γ, UVB/UVA, azathioprine, or MTX within 4 weeks ≥ 3 years duration of disease for monotherapy, ≥ 2 years for combination study Tx with leukotriene inhibitors within 4 weeks Hx of inadequate response to stable regimen of TCS or calcineurin inhbitors

Interventions �M 4 A Study: Sub. Q Dupilumab injections, once a week at either 75, 150, or 300 mg x 4 weeks (1: 4) �M 4 B Study Sub. Q Dupilumab injections, once a week at either 150 or 300 mg x 4 weeks (1: 3) �M 12 Study: Sub. Q Dupilumab injections, once a week at 300 mg x 12 weeks (1: 1) �C 4 Study: Sub. Q Dupilumab injections 4 x per week at 300 mg in combination with standardized regimen of topical glucocorticoids (1: 1)

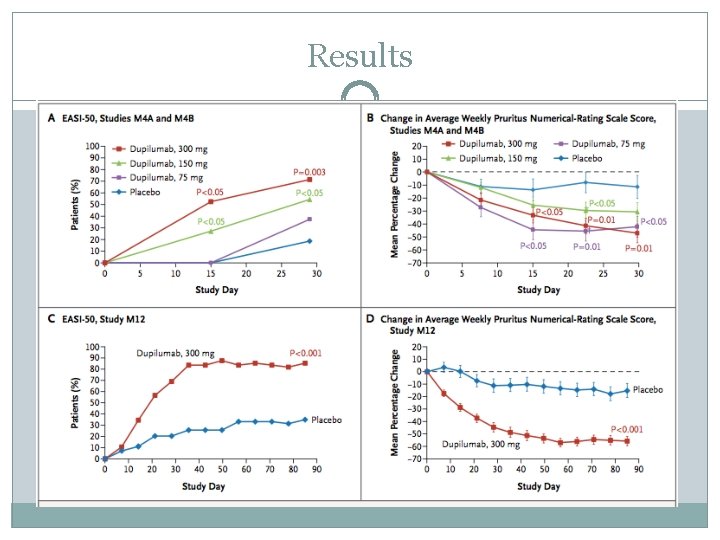
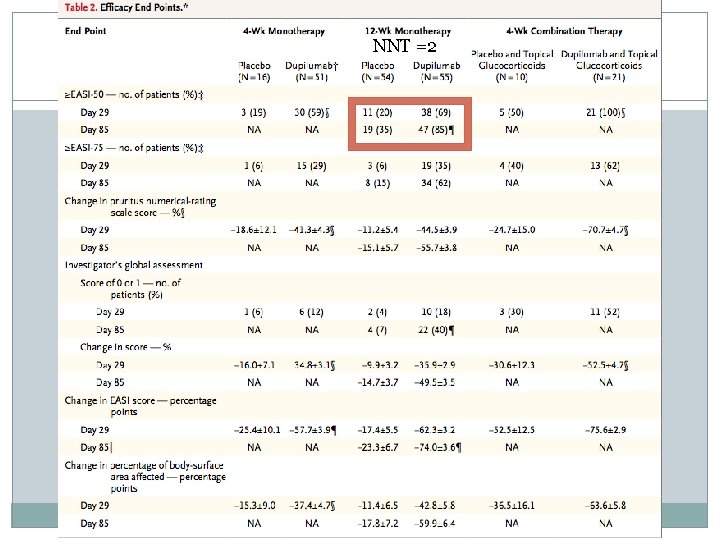
End Points � C 4 Study Primary endpoints: Incidence and severity of adverse events Efficacy endpoints: % change in EASI score, Global assessment score, SCORAD score, Pruritis numerical-rating scale, proportions of pt with GAS of 0 or 1 and EASI-50 at day 29 � M 4 A and M 4 B Studies Primary endpoint is safety (incidence of adverse events, VS, PE, clinical lab testing, ECG) Exploratory efficacy endpoints: Investigator’s global assessment score of 0 or 1, % reduction in affected BSA, EASI score, 5 D pruritis score, Pruritis numerical rating scale, and levels of TARC and Ig. E � M 12 Study Primary endpoint: assess clinical efficacy based on percent change in EASI score, Secondary endpoint: assess safety Other Efficacy endpoints: % in affected BSA, SCORAD score, pruritis numericalrating scale, 5 -D pruritis scale, proportion of patients with of ≥ 50% in EASI score (EASI-50), global assessment score of 0 or 1

Statistical Analysis � Observations from M 4 A and M 4 B were pooled due to similar study design and patient populations Fisher’s exact test for categorical variables ANCOVA for continuous variables (Tx as main factor and baseline value as covariate) � M 12 Study 50 patients/grp to provide study with 97% power to detect between-group difference of 40% in percentage change from baseline EASI scores (assuming SD of 50% for %change from baseline to week 12; use of Two-sided test with 0. 049 significance level Analysis of continuous variables with ANCOVA (tx and baseline Ig. E as fixed effects, relevant baseline value as covariate) � C 4 Study Analysis of categorical variables with Fisher’s exact test Continuous variables analyzed with ANCOVA with tx as main factor and baseline values as covariates, and time-to-event variables with the use of log rank test Nominal P values were estimated
Results
Results
NNT =2 Results
Results

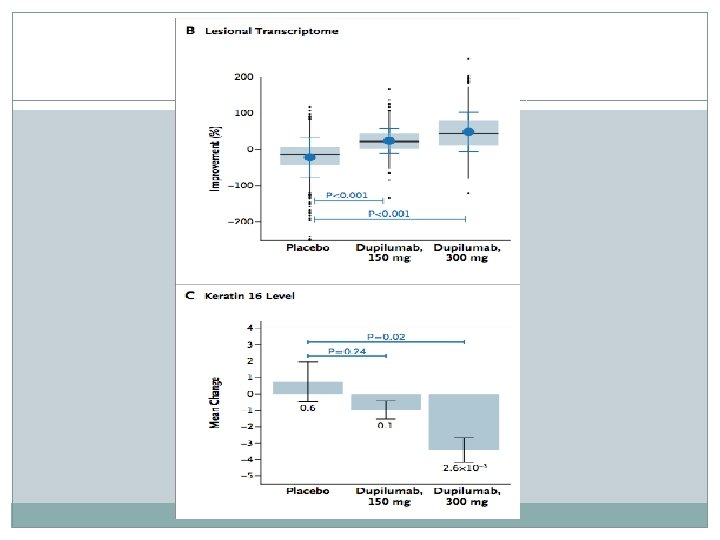
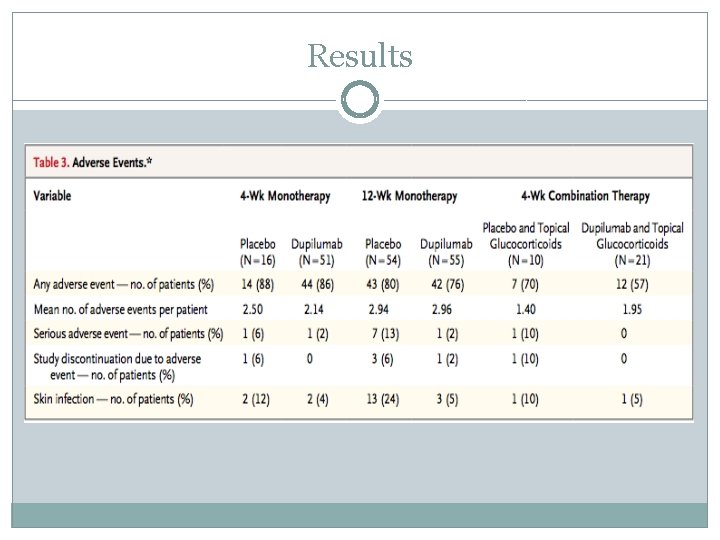
Discussion �Dupilumab treatment for adults with moderate-to-severe atopic dermatitis resulted in marked reductions in signs, symptoms, and associated biomarker levels across four separate studies. �Strong support for pathophys importance of Th 2 cytokines in atopic dermatitis �Monotherapy or combo therapy with Dupilumab was associated with both improved skin lesions and rapid and substantial in pruritis � Side effects and adverse events were generally mild and balanced between drug and placebo, with placebo groups having rates of skin infection

Are the Results Valid? �Randomized: Yes �Double-Blinded: Yes �Multi-Center: Yes �Similar Baseline Characteristics: Yes �Groups Treated Equally: Within sub-studies, but not as a whole �Were all the patients accounted for? No

Limitations �Small sample sizes with significant dropout rates in monotherapy studies �Variable inclusion and exclusion criteria �Data analysis by pharmaceutical company (Regeneron and Sanofi) �Population studied was limited to middle-aged Caucasian males

Conclusion �Patients treated with dupilumab had marked and rapid improvement in all evaluated measures of atopic dermatitis disease activity. Side effect profiles were not dose-limiting

Will the Results Help Me in Caring for My Patient? �Can the results be applied to my patient? While my patient has moderate-to-severe atopic dermatitis despite treatment with topical steroids, she is not a middleaged Caucasian male Studies were not performed in patients with concurrent uncontrolled asthma �Were Clinically Important Outcomes Measured? Yes �Are the likely benefits worth the cost? Cost was not studied

References � Middleton's Allergy: Principles and Practice , Eighth Edition N. Franklin Adkinson, Bruce S. Bochner, A. Wesley Burks, William W. Busse, Stephen T. Holgate, Robert F. Lemanske, and Robyn E. O'Hehir
- Slides: 26