BIOL 2401 Biol 2401 Fundamentals of Anatomy and

BIOL 2401 Biol 2401 Fundamentals of Anatomy and Physiology www. biol 2401. weebly. com wgrant 4@alamo. edu (210) 486 -2370 © 2012 Pearson Education, Inc.

2 The Chemical Level of Organization Power. Point® Lecture Presentations prepared by Jason La. Pres Lone Star College—North Harris © 2012 Pearson Education, Inc.

An Introduction to the Chemical Level of Organization Learning Outcomes 2 -1 Describe an atom and how atomic structure affects interactions between atoms. 2 -2 Compare the ways in which atoms combine to form molecules and compounds. 2 -3 Distinguish among the major types of chemical reactions that are important for studying physiology. 2 -4 Describe the crucial role of enzymes in metabolism. 2 -5 Distinguish between organic and inorganic compounds. 2 -6 Explain how the chemical properties of water make life possible. 2 -7 Discuss the importance of p. H and the role of buffers in body fluids. 2 -8 Describe the physiological roles of inorganic compounds. 2 -9 Discuss the structures and functions of carbohydrates. 2 -10 Discuss the structures and functions of lipids. 2 -11 Discuss the structures and functions of proteins. 2 -12 Discuss the structures and functions of nucleic acids. 2 -13 Discuss the structures and functions of high- energy compounds. 2 -14 Explain the relationship between chemicals and cells. © 2012 Pearson Education, Inc.

An Introduction to the Chemical Level of Organization Chemistry Is the science of change. It is the science that deals with the structure of matter (anything that takes up space and has mass, the amount of material in matter). (Mass is not weight). Topics of this chapter include: The structure of atoms The basic chemical building blocks How atoms combine to form increasingly complex structures © 2012 Pearson Education, Inc.

2 -1 Atoms and Atomic Structure Matter is made up of atoms Atoms join together to form chemicals with different characteristics Chemical characteristics determine physiology at the molecular and cellular levels Subatomic Particles of the Atom Proton Positive charge, 1 mass unit Neutron Neutral, 1 mass unit Electron Negative charge, low mass © 2012 Pearson Education, Inc.
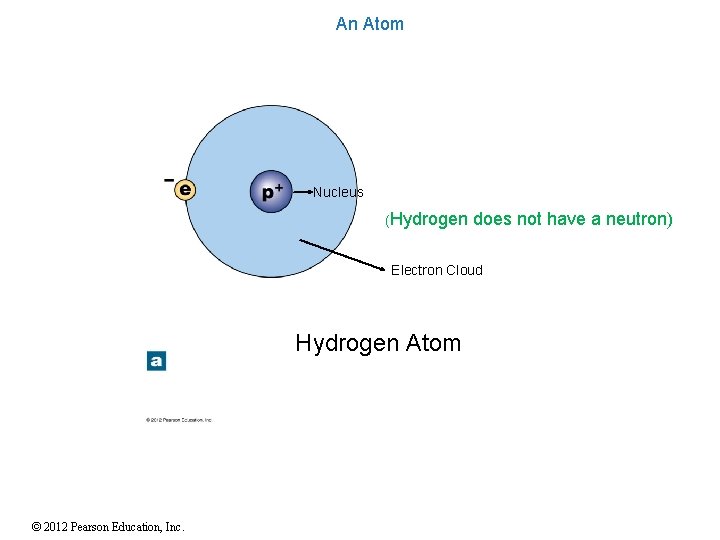
An Atom Nucleus (Hydrogen does not have a neutron) Electron Cloud Hydrogen Atom © 2012 Pearson Education, Inc.

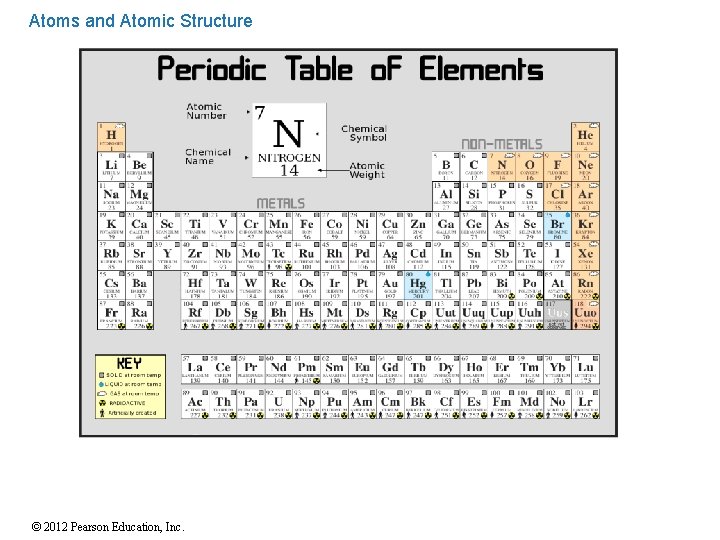
Atoms and Atomic Structure An element is a pure substance composed of atoms of only one kind because atoms are the smallest particles of an element that still retain the characteristics of that element. Only 92 elements exist in nature. All of these elements can be found on the Periodic Table of Elements. © 2012 Pearson Education, Inc.
Atoms and Atomic Structure © 2012 Pearson Education, Inc.

Atoms and Atomic Structure . Sample of one element on Periodic Table of Elements 1 H Hydrogen 1. 00974 Atomic Number Symbol Name of Element Atomic Mass 1 Practice finding information using the Periodic Table of Elements. © 2012 Pearson Education, Inc.
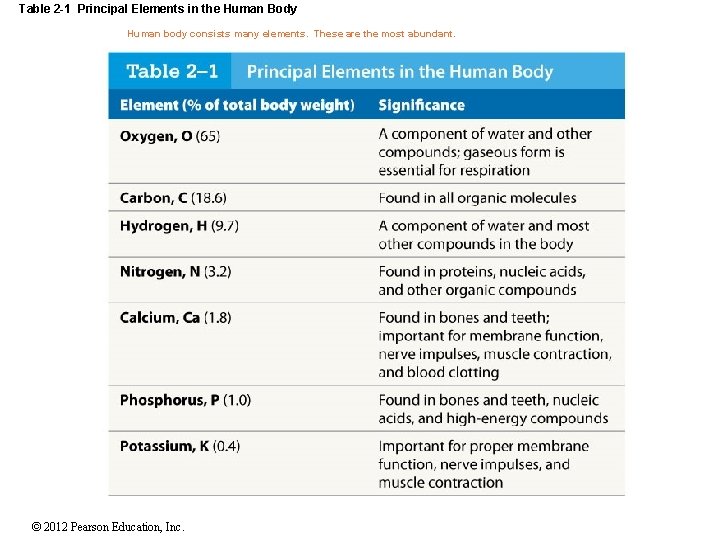
Table 2 -1 Principal Elements in the Human Body Human body consists many elements. These are the most abundant. © 2012 Pearson Education, Inc.
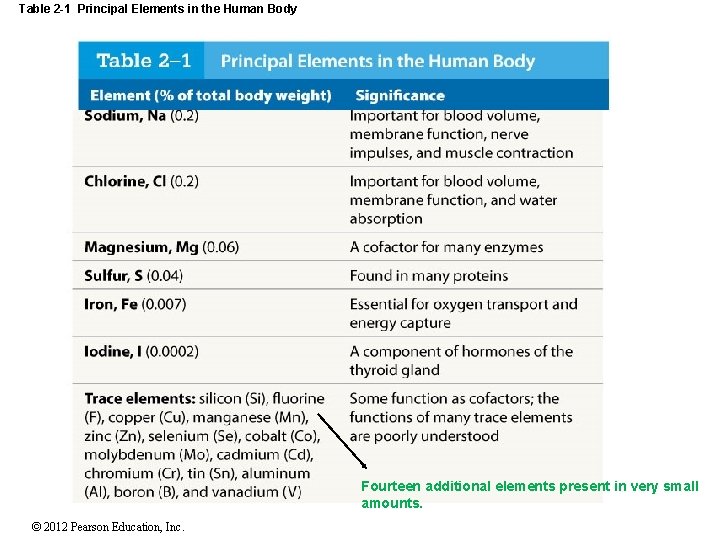
Table 2 -1 Principal Elements in the Human Body Fourteen additional elements present in very small amounts. © 2012 Pearson Education, Inc.

2 -1 Atoms and Atomic Structure Elements and Isotopes Elements are determined by the atomic number of an atom Remember atomic number = number of protons Isotopes are the specific version of an element based on its mass number Mass number = number of protons plus the number of neutrons Only neutrons are different because the number of protons determines the element © 2012 Pearson Education, Inc.

Atoms and Atomic Structure Radioisotopes are isotopes with unstable nuclei and spontaneously break down and emit subatomic particles or radiation in measurable amounts. The break-down process is called radioactive decay. Strongly radioactive isotopes are dangerous, causing destruction of cells and damage to living tissues. Weakly radioactive isotopes are used in diagnostic procedures. Russian Spy Poisoned with Polonium http: //news. bbc. co. uk/2/hi/uk_news/6180682. stm Yassar Araphat Poisoned with Polonium http: //content. usatoday. com/communities/ondeadline/post/2012/07/aljazeera-tests-hint-yasser-arafat-died-of-polonium-poisoning/1 © 2012 Pearson Education, Inc.
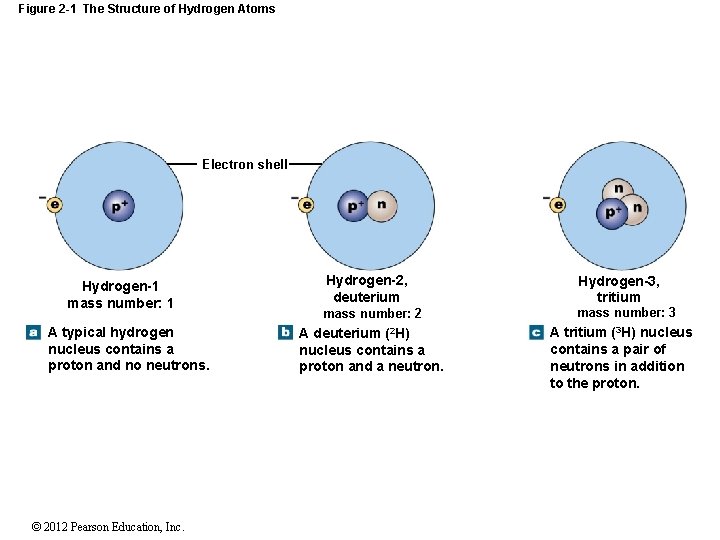
Figure 2 -1 The Structure of Hydrogen Atoms Electron shell Hydrogen-1 mass number: 1 A typical hydrogen nucleus contains a proton and no neutrons. © 2012 Pearson Education, Inc. Hydrogen-2, deuterium Hydrogen-3, tritium mass number: 2 mass number: 3 A deuterium (2 H) nucleus contains a proton and a neutron. A tritium (3 H) nucleus contains a pair of neutrons in addition to the proton.

2 -1 Atoms and Atomic Structure Atomic Weights (expressed in atomic mass units—amu) Exact mass of all particles Measured in moles (mol)—a quantity with a weight in grams equal to the element’s atomic weight. One mole of a given element always contains the same number of atoms as one mole of any other element. That number is called Avogadro’s number— 6. 023 x 1023 or about 600 billion trillion. © 2012 Pearson Education, Inc.

2 -1 Atoms and Atomic Structure Electrons and Energy Levels Electrons in the electron cloud determine the reactivity of an atom The electron cloud contains shells, or energy levels that hold a maximum number of electrons Lower shells fill first Outermost shell is the valence shell, and it determines bonding The number of electrons per shell corresponds to the number of atoms in that row of the Periodic Table. © 2012 Pearson Education, Inc.

Atoms and Atomic Structure Energy Levels Level Maximum Number of Electrons 1 2 2 8 3 8 Practice the energy levels for the element that you had on the Periodic Table of Elements. Example: Element H Nucleus Electron Energy 1 p/0 n 1 2 Write the electron configurations for C, O, Na, Cl, Ar © 2012 Pearson Education, Inc.
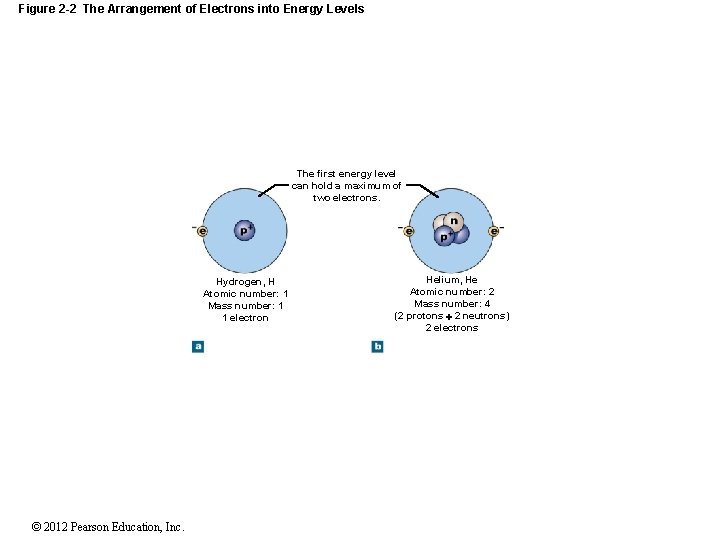
Figure 2 -2 The Arrangement of Electrons into Energy Levels The first energy level can hold a maximum of two electrons. Hydrogen, H Atomic number: 1 Mass number: 1 1 electron © 2012 Pearson Education, Inc. Helium, He Atomic number: 2 Mass number: 4 (2 protons 2 neutrons) 2 electrons
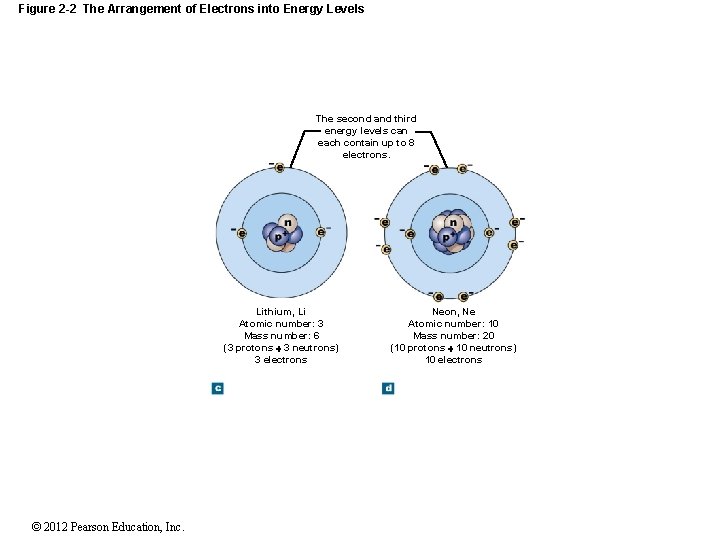
Figure 2 -2 The Arrangement of Electrons into Energy Levels The second and third energy levels can each contain up to 8 electrons. Lithium, Li Atomic number: 3 Mass number: 6 (3 protons 3 neutrons) 3 electrons © 2012 Pearson Education, Inc. Neon, Ne Atomic number: 10 Mass number: 20 (10 protons 10 neutrons) 10 electrons

2 -2 Molecules and Compounds Chemical Bonds Involve the sharing, gaining, and losing of electrons in the valence shell Three major types of chemical bonds Ionic bonds Attraction between cations (electron donor) and anions (electron acceptor) Covalent bonds Strong electron bonds involving shared electrons Hydrogen bonds Weak polar bonds based on partial electrical attractions 3 What is the principal difference between an ionic bond a covalent bond? . © 2012 Pearson Education, Inc.

2 -2 Molecules and Compounds Chemical Bonds Form molecules and/or compounds Molecules Two or more atoms joined by strong bonds Compounds Two or more atoms OF DIFFERENT ELEMENTS joined by strong or weak bonds Compounds are all molecules, but not all molecules are compounds H 2 = molecule only © 2012 Pearson Education, Inc. H 2 O = molecule and compound

2 -2 Molecules and Compounds Ionic Bonds One atom—the electron donor—loses one or more electrons and becomes a cation, with a positive charge Another atom—the electron acceptor—gains those same electrons and becomes an anion, with a negative charge Attraction between the opposite charges then draws the two ions together 4 What are cations and anions? Cations are positively charged ions and anions are negatively charged ions. © 2012 Pearson Education, Inc.
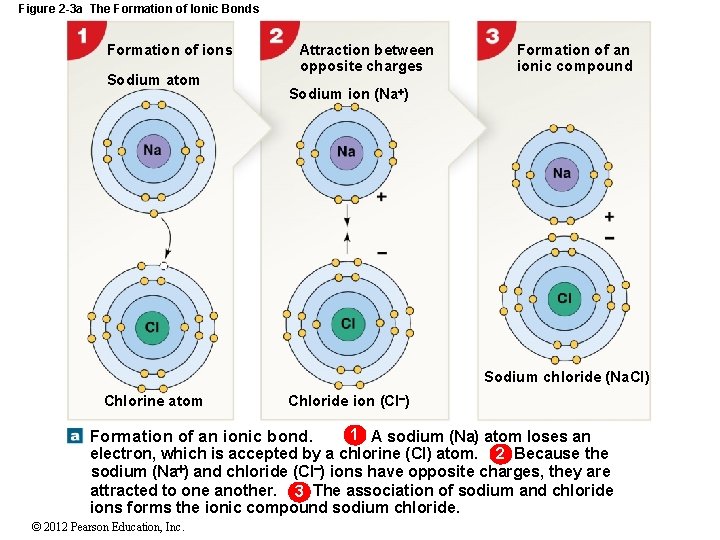
Figure 2 -3 a The Formation of Ionic Bonds Formation of ions Sodium atom Attraction between opposite charges Formation of an ionic compound Sodium ion (Na ) Sodium chloride (Na. Cl) Chlorine atom Chloride ion (Cl ) 1 A sodium (Na) atom loses an electron, which is accepted by a chlorine (Cl) atom. 2 Because the sodium (Na ) and chloride (Cl ) ions have opposite charges, they are attracted to one another. 3 The association of sodium and chloride ions forms the ionic compound sodium chloride. Formation of an ionic bond. © 2012 Pearson Education, Inc.
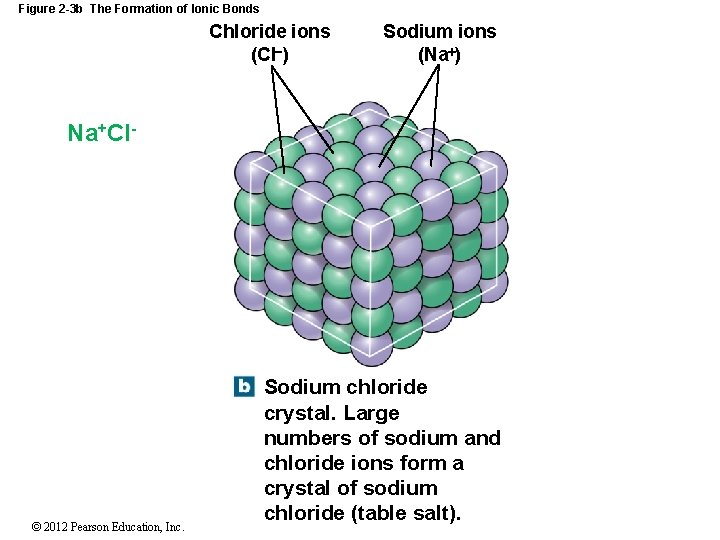
Figure 2 -3 b The Formation of Ionic Bonds Chloride ions (Cl ) Sodium ions (Na ) Na+Cl- © 2012 Pearson Education, Inc. Sodium chloride crystal. Large numbers of sodium and chloride ions form a crystal of sodium chloride (table salt).
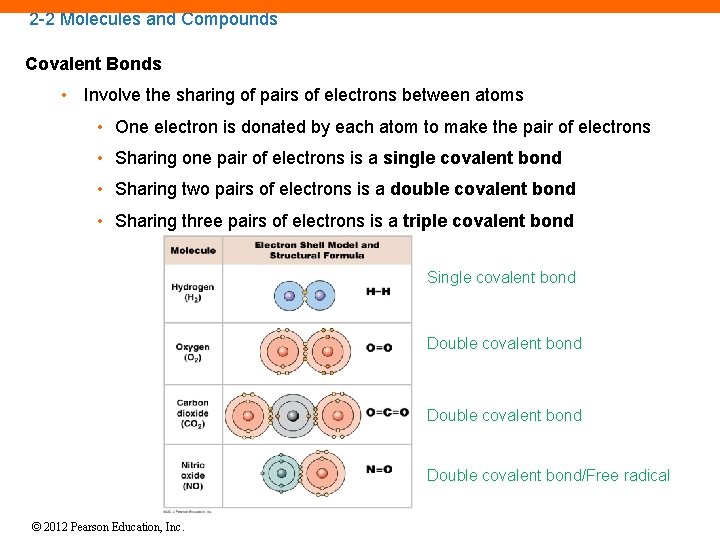
2 -2 Molecules and Compounds Covalent Bonds • Involve the sharing of pairs of electrons between atoms • One electron is donated by each atom to make the pair of electrons • Sharing one pair of electrons is a single covalent bond • Sharing two pairs of electrons is a double covalent bond • Sharing three pairs of electrons is a triple covalent bond Single covalent bond Double covalent bond/Free radical © 2012 Pearson Education, Inc.

2 -2 Molecules and Compounds Covalent Bonds Nonpolar covalent bonds Involve equal sharing of electrons because atoms involved in the bond have equal pull for the electrons Polar covalent bonds Involve the unequal sharing of electrons because one of the atoms involved in the bond has a disproportionately strong pull on the electrons Form polar molecules — like water © 2012 Pearson Education, Inc.
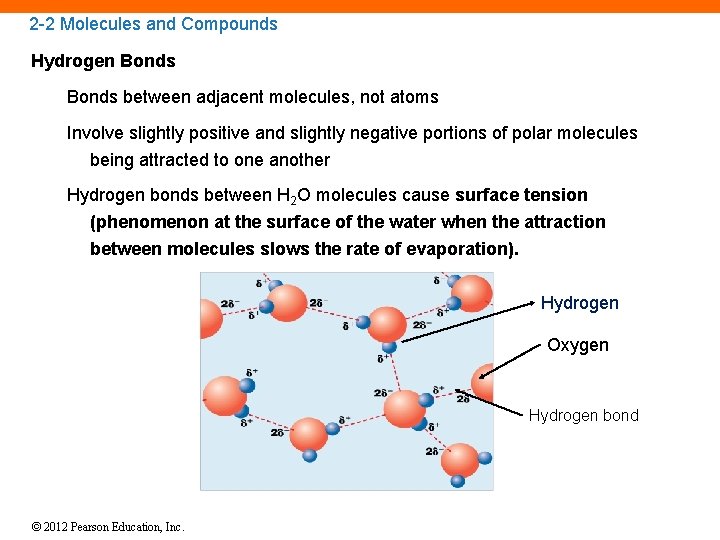
2 -2 Molecules and Compounds Hydrogen Bonds between adjacent molecules, not atoms Involve slightly positive and slightly negative portions of polar molecules being attracted to one another Hydrogen bonds between H 2 O molecules cause surface tension (phenomenon at the surface of the water when the attraction between molecules slows the rate of evaporation). Hydrogen Oxygen Hydrogen bond © 2012 Pearson Education, Inc.

2 -2 Molecules and Compounds States of Matter Solid Constant volume and shape Liquid Constant volume but changes shape Gas Changes volume and shape Water is the only substance that occurs as a solid (ice), a liquid (water), and a gas (water vapor at temperatures compatible with life. © 2012 Pearson Education, Inc.

2 -2 Molecules and Compounds Molecular Weights The molecular weight of a molecule is the sum of the atomic weights of its component atoms H = approximately 1 O = approximately 16 H 2 = approximately 2 H 2 O = approximately 18 What is the molecular weight of these compounds? C 6 H 12 O 6 H 2 SO 4 © 2012 Pearson Education, Inc. 72+12+96 = 180 g 2+32+64 = 98 g

2 -3 Chemical Reactions In a Chemical Reaction Either new bonds are formed or existing bonds are broken Reactants Materials going into a reaction Products Materials coming out of a reaction Metabolism All of the reactions that are occurring at one time © 2012 Pearson Education, Inc.
5 Write the chemical notation for MOLECULES of water. © 2012 Pearson Education, Inc.

Molecules and Compounds First Law of Thermodynamics is an expression of the principle of conservation of energy. The law expresses that energy can be transformed, i. e. changed from one form to another but cannot be created or destroyed. How does this equation demonstrate the law? 2 H 2 + O 2 → 2 H 2 O Balance the following equation. ___CO 2 + ___H 2 O + Energy → C 6 H 12 O 6 + ___ O 2 6 CO 2 + 6 H 20 + Energy → C 6 H 12 O 6 + 6 O 2 © 2012 Pearson Education, Inc.
© 2012 Pearson Education, Inc.
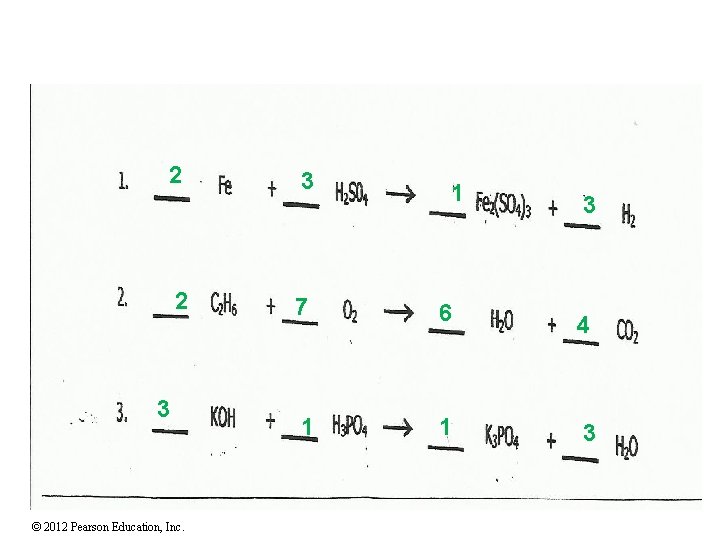
2 2 3 © 2012 Pearson Education, Inc. 3 1 3 7 6 4 1 1 3

2 -3 Chemical Reactions Basic Energy Concepts Energy The power to do work Work A change in mass or distance Kinetic energy Energy of motion Potential energy Stored energy Chemical energy Potential energy stored in chemical bonds © 2012 Pearson Education, Inc.

2 -3 Chemical Reactions Types of Chemical Reactions Decomposition reaction (catabolism) Synthesis reaction (anabolism) Exchange reaction Reversible reaction © 2012 Pearson Education, Inc.

2 -3 Chemical Reactions Decomposition Reaction (Catabolism) Breaks chemical bonds AB A + B Hydrolysis A-B + H 2 O A-H + HO-B Synthesis Reaction (Anabolism) Forms chemical bonds A + B AB Dehydration synthesis (condensation reaction) A-H + HO-B A-B + H 2 O © 2012 Pearson Education, Inc.

2 -3 Chemical Reactions Exchange Reaction Involves decomposition first, then synthesis AB + CD AD + CB Reversible Reaction A + B AB At equilibrium the amounts of chemicals do not change even though the reactions are still occurring Reversible reactions seek equilibrium, balancing opposing reaction rates Add or remove reactants Reaction rates adjust to reach a new equilibrium © 2012 Pearson Education, Inc.

2 -4 Enzymes Chemical Reactions Cells cannot start without help Activation energy is the amount of energy needed to get a reaction started Enzymes are protein catalysts that lower the activation energy of reactions. It only affects the rate of the reaction, not the direction or products. Without enzyme Reactants With enzyme Stable product © 2012 Pearson Education, Inc.

2 -4 Enzymes Exergonic (Exothermic) Reactions Produce more energy than they use Endergonic (Endothermic) Reactions Use more energy than they produce © 2012 Pearson Education, Inc.

2 -5 Organic and Inorganic Compounds Major Classes or Chemical Compounds Nutrients Essential molecules obtained from food Metabolites Molecules made or broken down in the body Inorganic Compounds Molecules not based on carbon and hydrogen Carbon dioxide, oxygen, water, and inorganic acids, bases, and salts Organic Compounds Molecules based on carbon and hydrogen Carbohydrates, proteins, lipids, and nucleic acids © 2012 Pearson Education, Inc.

2 -6 Properties of Water Accounts for up to two-thirds of your total body weight A solution is a uniform mixture of two or more substances It consists of a solvent, or medium, in which atoms, ions, or molecules of another substance, called a solute, are individually dispersed. © 2012 Pearson Education, Inc.

2 -6 Properties of Water Unusual Properties of Water Solubility Water’s ability to dissolve a solute in a solvent to make a solution Reactivity Most body chemistry occurs in water High Heat Capacity Water’s ability to absorb and retain heat Lubrication To moisten and reduce friction © 2012 Pearson Education, Inc.

2 -6 Properties of Water The Properties of Aqueous Solutions Ions and polar compounds undergo ionization, or dissociation in water Polar water molecules form hydration spheres around ions and small polar molecules to keep them in solution © 2012 Pearson Education, Inc.
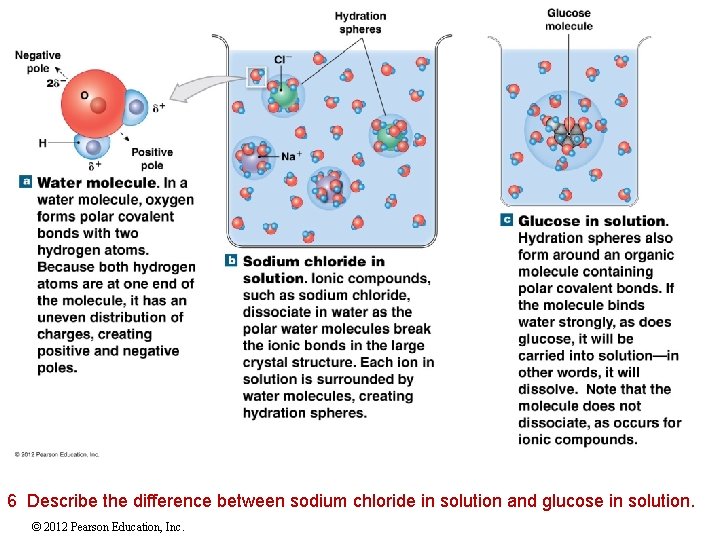
6 Describe the difference between sodium chloride in solution and glucose in solution. © 2012 Pearson Education, Inc.

2 -6 Properties of Water The Properties of Aqueous Solutions Electrolytes and body fluids Electrolytes are inorganic ions that conduct electricity in solution Electrolyte imbalance seriously disturbs vital body functions © 2012 Pearson Education, Inc.

Table 2 -2 Important Electrolytes that Dissociate in Body Fluids An electrolyte is a soluble inorganic molecules whose ions will conduct an electrical current © 2012 Pearson Education, Inc.

2 -6 Properties of Water The Properties of Aqueous Solutions Hydrophilic and hydrophobic compounds Hydrophilic hydro- = water, philos = loving Interacts with water Includes ions and polar molecules Hydrophobic phobos = fear What is hydrophobia? RABIES – an acute deadly virus disease of the nervous system transmitted by the bite of an affected animal. Does NOT interact with water Includes nonpolar molecules, fats, and oils © 2012 Pearson Education, Inc.

2 -6 Properties of Water Colloids and Suspensions Colloid A solution of very large organic molecules For example, blood plasma Suspension A solution in which particles settle (sediment) For example, whole blood Concentration The amount of solute in a solvent (mol/L, mg/m. L) © 2012 Pearson Education, Inc.

2 -7 p. H and Homeostasis Hydrogen ions are extremely reactive in solution, they will break chemical bonds, change the shapes of complex molecules, and disrupt cell and tissue functions. The concentration of hydrogen ions in body fluids must be regulated precisely. p. H The concentration of hydrogen ions (H+) in a solution Neutral p. H A balance of H+ and OH Pure water = 7. 0 (1 x 10 -7 mol/L. -(-7) or 7. © 2012 Pearson Education, Inc.

2 -7 p. H and Homeostasis Acidic p. H Lower Than 7. 0 High H+ concentration Low OH concentration Basic (or alkaline) p. H Higher Than 7. 0 Low H+ concentration High OH concentration p. H of Human Blood Ranges from 7. 35 to 7. 45 © 2012 Pearson Education, Inc.
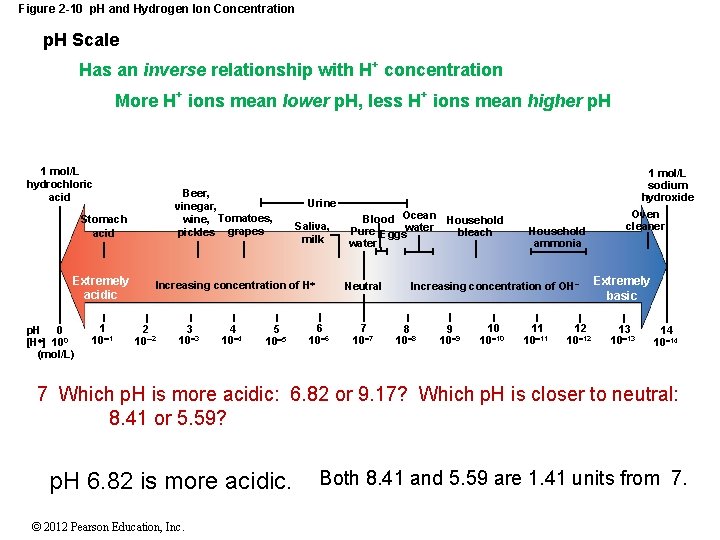
Figure 2 -10 p. H and Hydrogen Ion Concentration p. H Scale Has an inverse relationship with H+ concentration More H+ ions mean lower p. H, less H+ ions mean higher p. H 1 mol/L hydrochloric acid Beer, vinegar, wine, Tomatoes, pickles grapes Stomach acid Extremely acidic p. H 0 [H ] 100 (mol/L) 1 10 1 Urine Saliva, milk Increasing concentration of H 2 10 2 3 10 3 4 10 4 5 10 5 1 mol/L sodium hydroxide Blood Ocean Household Pure Eggswater bleach water Neutral 6 10 6 7 10 7 Household ammonia Increasing concentration of OH 8 10 8 9 10 10 10 11 12 10 12 Oven cleaner Extremely basic 13 10 13 14 10 14 7 Which p. H is more acidic: 6. 82 or 9. 17? Which p. H is closer to neutral: 8. 41 or 5. 59? p. H 6. 82 is more acidic. © 2012 Pearson Education, Inc. Both 8. 41 and 5. 59 are 1. 41 units from 7.

2 -8 Inorganic Compounds Acid A solute that adds hydrogen ions to a solution—proton donor Strong acids dissociate completely in solution Base A solute that removes hydrogen ions from a solution—proton acceptor Strong bases dissociate completely in solution Weak Acids and Weak Bases Fail to dissociate completely Help to balance the p. H Salts Solutes that dissociate into cations and anions other than hydrogen ions and hydroxide ions Example: Table salt Na. Cl → Na+ and Cl- © 2012 Pearson Education, Inc.

2 -8 Inorganic Compounds Buffers and p. H Control Buffers Weak acid/salt compounds Neutralize either strong acid or strong base Sodium bicarbonate (Na. HCO 3) is very important in humans Antacids Basic compounds that neutralize acid and form a salt Alka-Seltzer, Tums, Rolaids, etc. © 2012 Pearson Education, Inc.

BIOL 2401 -094 • Fundamentals of Anatomy and Physiology • Chapter 2 • wgrant 4@alamo. edu • (210) 486 -2370 © 2012 Pearson Education, Inc.

2 -9 Carbohydrates Organic Molecules Contain H, C, and usually O Are covalently bonded Contain functional groups that determine chemistry Carbohydrates Lipids Proteins (or amino acids) Nucleic acids Grouping of atoms occurring repeatedly that influence the properties of any molecule they are in. © 2012 Pearson Education, Inc.

2 -9 Carbohydrates Contain carbon, hydrogen, and oxygen in a 1: 2: 1 ratio Monosaccharide — simple sugar Disaccharide — two sugars Polysaccharide — many sugars © 2012 Pearson Education, Inc.

2 -9 Carbohydrates Monosaccharides Simple sugars with 3 to 7 carbon atoms Glucose, fructose, galactose Disaccharides Two simple sugars condensed by dehydration synthesis Sucrose, maltose Polysaccharides Many monosaccharides condensed by dehydration synthesis Glycogen, starch, cellulose © 2012 Pearson Education, Inc.
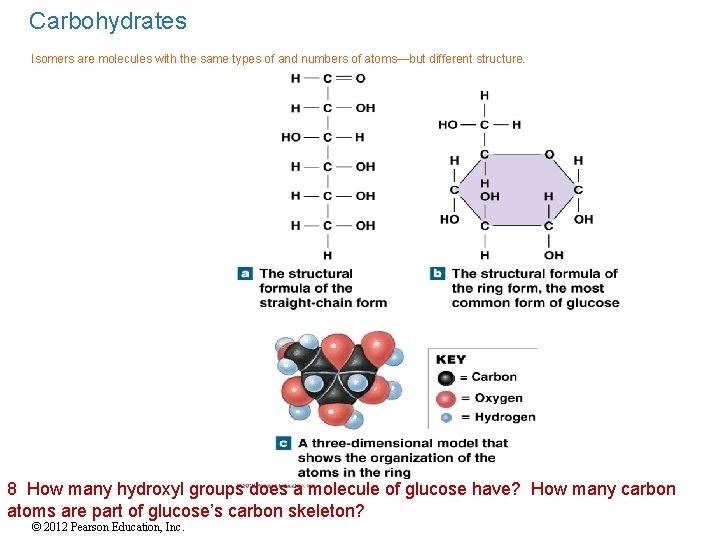
Carbohydrates Isomers are molecules with the same types of and numbers of atoms—but different structure. 8 How many hydroxyl groups does a molecule of glucose have? How many carbon atoms are part of glucose’s carbon skeleton? © 2012 Pearson Education, Inc.
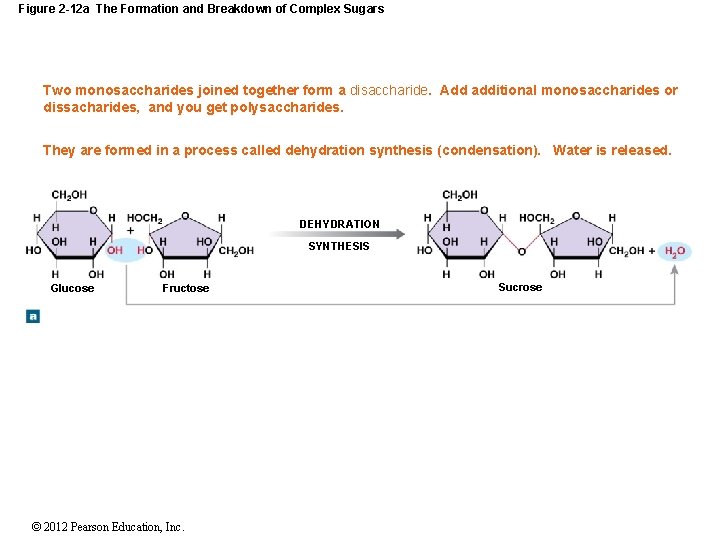
Figure 2 -12 a The Formation and Breakdown of Complex Sugars Two monosaccharides joined together form a disaccharide. Add additional monosaccharides or dissacharides, and you get polysaccharides. They are formed in a process called dehydration synthesis (condensation). Water is released. DEHYDRATION SYNTHESIS Glucose Fructose © 2012 Pearson Education, Inc. Sucrose
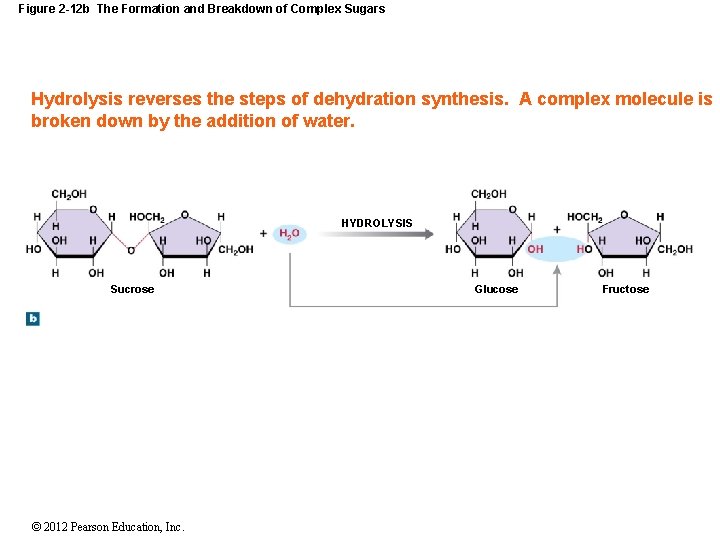
Figure 2 -12 b The Formation and Breakdown of Complex Sugars Hydrolysis reverses the steps of dehydration synthesis. A complex molecule is broken down by the addition of water. HYDROLYSIS Sucrose © 2012 Pearson Education, Inc. Glucose Fructose
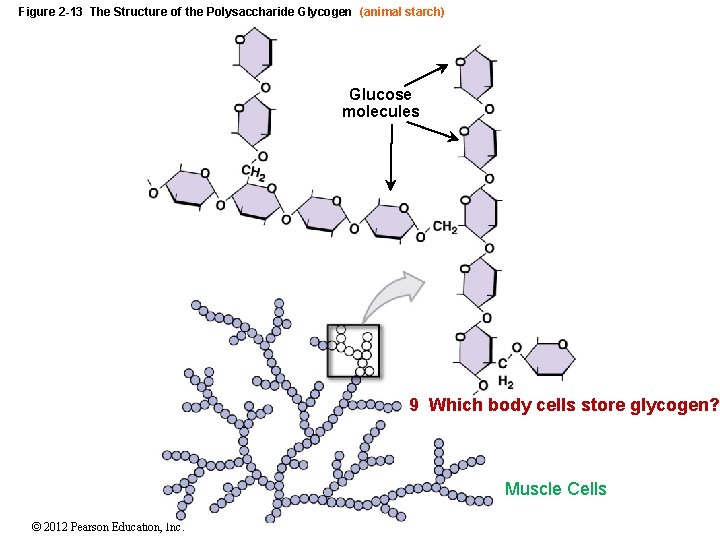
Figure 2 -13 The Structure of the Polysaccharide Glycogen (animal starch) Glucose molecules 9 Which body cells store glycogen? Muscle Cells © 2012 Pearson Education, Inc.
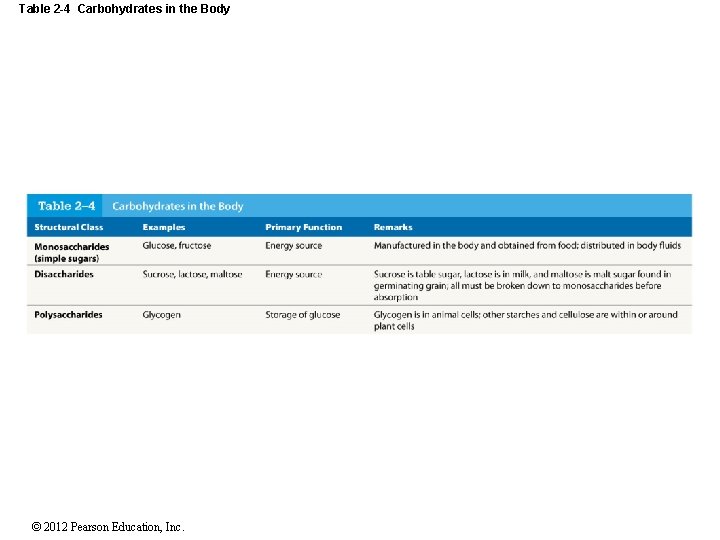
Table 2 -4 Carbohydrates in the Body © 2012 Pearson Education, Inc.

2 -10 Lipids Mainly hydrophobic molecules such as fats, oils, and waxes Made mostly of carbon and hydrogen atoms Include: Fatty acids Eicosanoids Glycerides Steroids Phospholipids and glycolipids © 2012 Pearson Education, Inc.

2 -10 Lipids Fatty Acids Long chains of carbon and hydrogen with a carboxyl group (COOH) at one end Are relatively nonpolar, except the carboxyl group Fatty acids may be: Saturated with hydrogen (no covalent bonds) Unsaturated (one or more double bonds) Monounsaturated = one double bond Polyunsaturated = two or more double bonds © 2012 Pearson Education, Inc.
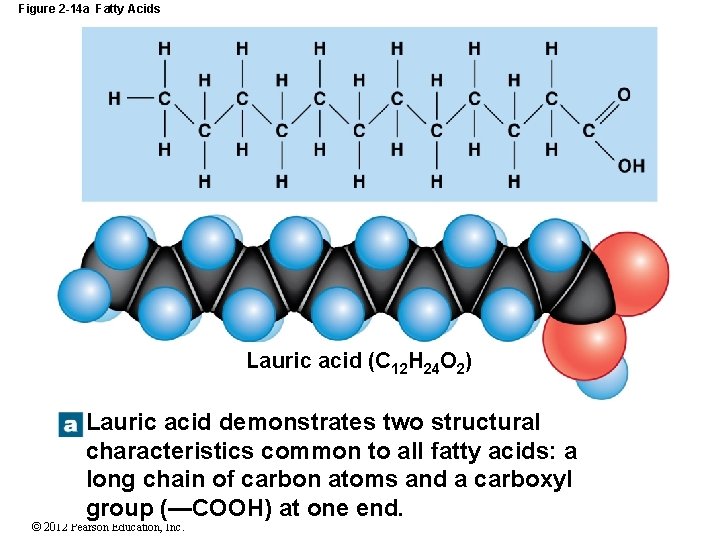
Figure 2 -14 a Fatty Acids Lauric acid (C 12 H 24 O 2) Lauric acid demonstrates two structural characteristics common to all fatty acids: a long chain of carbon atoms and a carboxyl group (—COOH) at one end. © 2012 Pearson Education, Inc.
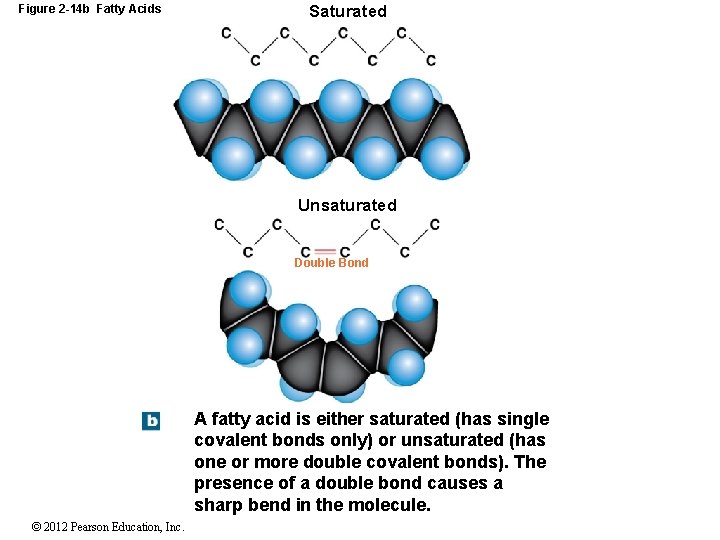
Figure 2 -14 b Fatty Acids Saturated Unsaturated Double Bond A fatty acid is either saturated (has single covalent bonds only) or unsaturated (has one or more double covalent bonds). The presence of a double bond causes a sharp bend in the molecule. © 2012 Pearson Education, Inc.

BIOL 2401 Something to Consider: A diet containing large amounts of saturated fatty acids has been shown to increase the risk of heart disease and other cardiovascular problems. The healthiest choices of unsaturated oils to use are canola oils and olive oil. Be careful of trans fatty acids (compounds from polyunsaturated oils such are margarine) seem to increase the risk of heart disease. Foods high in omega-3 fatty acids (fish flesh and fish oils) seem to reduce the risk of heart disease and other inflammatory diseases. (rheumatoid arthritis). © 2012 Pearson Education, Inc.
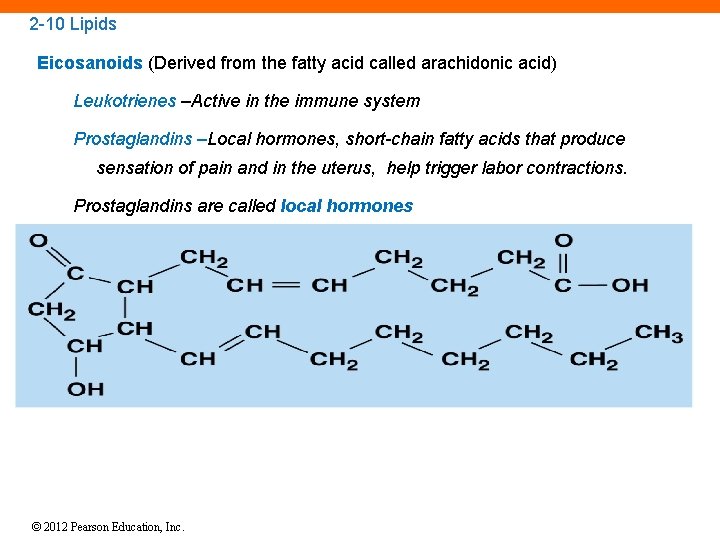
2 -10 Lipids Eicosanoids (Derived from the fatty acid called arachidonic acid) Leukotrienes –Active in the immune system Prostaglandins –Local hormones, short-chain fatty acids that produce sensation of pain and in the uterus, help trigger labor contractions. Prostaglandins are called local hormones © 2012 Pearson Education, Inc.

2 -10 Lipids Glycerides are fatty acids attached to a glycerol molecule Monoglyceride (glycerol + one fatty acid) Diglyceride (glycerol + two fatty acids) Triglycerides are three fatty-acid tails Also called triacylglycerols or neutral fats Have three important functions Energy source Insulation Protection © 2012 Pearson Education, Inc.
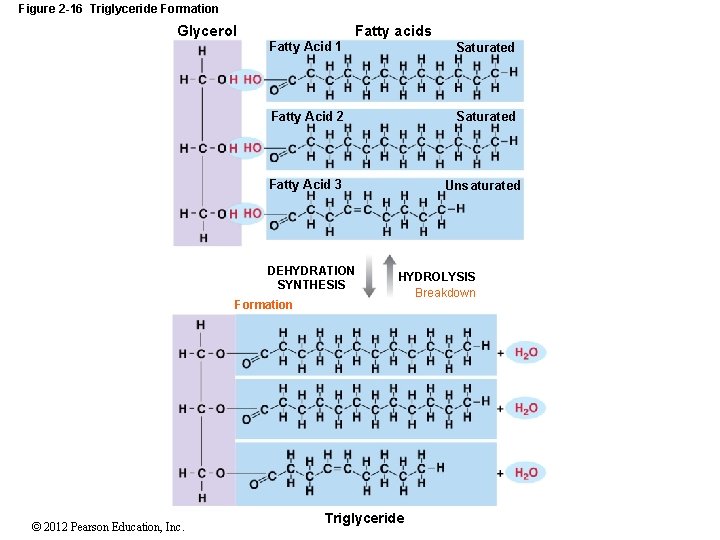
Figure 2 -16 Triglyceride Formation Glycerol Fatty acids Fatty Acid 1 Saturated Fatty Acid 2 Saturated Fatty Acid 3 Unsaturated DEHYDRATION SYNTHESIS Formation © 2012 Pearson Education, Inc. HYDROLYSIS Breakdown Triglyceride

2 -10 Lipids Steroids (large lipid molecules with a distinctive carbon framework) Four rings of carbon and hydrogen with an assortment of functional groups Types of steroids: Cholesterol Component of plasma (cell) membranes Estrogens and testosterone Sex hormones Corticosteroids and calcitriol Metabolic regulation Bile salts Derived from steroids 10 What is the danger of a diet high in cholesterol? The development of heart disease is the danger of high cholesterol blood levels. © 2012 Pearson Education, Inc.
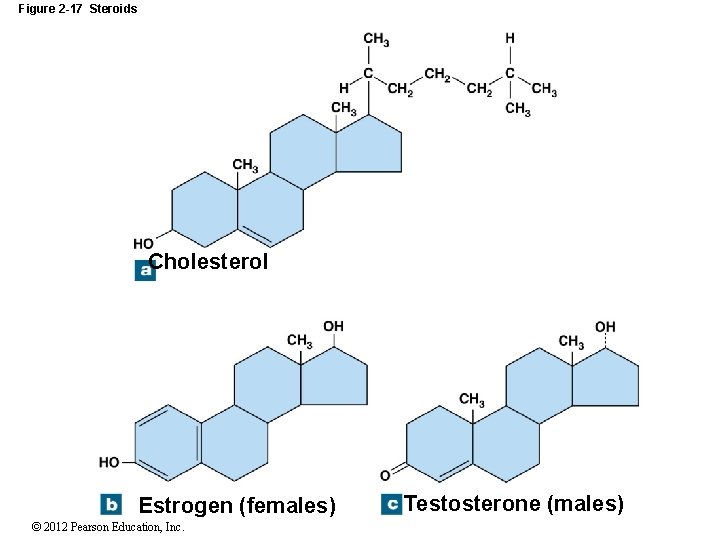
Figure 2 -17 Steroids Cholesterol Estrogen (females) © 2012 Pearson Education, Inc. Testosterone (males)

2 -10 Lipids Phospholipids and Glycolipids Diglycerides attached to either a phosphate group (phospholipid) or a sugar (glycolipid) Generally, both have hydrophilic heads and hydrophobic tails and are structural lipids, components of plasma (cell) membranes © 2012 Pearson Education, Inc.
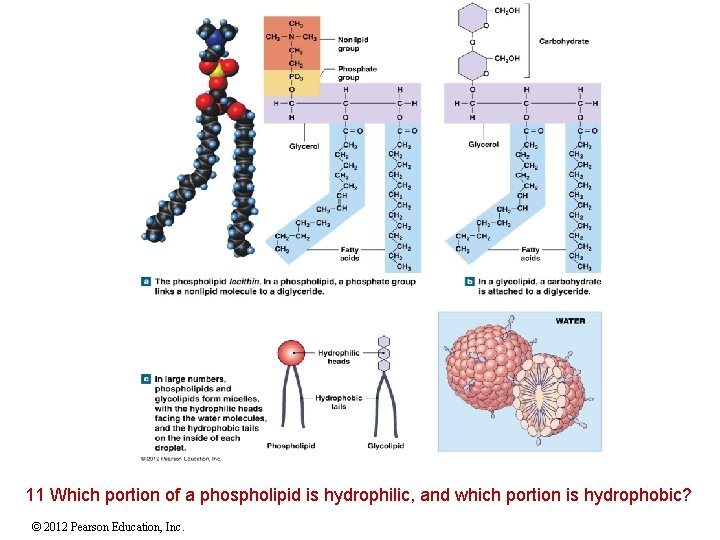
11 Which portion of a phospholipid is hydrophilic, and which portion is hydrophobic? © 2012 Pearson Education, Inc.
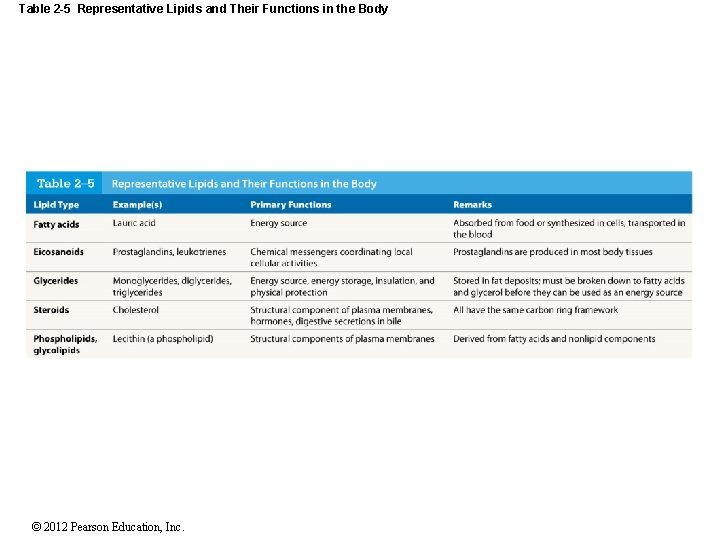
Table 2 -5 Representative Lipids and Their Functions in the Body © 2012 Pearson Education, Inc.

2 -11 Proteins Are the most abundant and important organic molecules Contain basic elements Carbon (C), hydrogen (H), oxygen (O), and nitrogen (N) Basic building blocks 20 amino acids © 2012 Pearson Education, Inc.

2 -11 Proteins Seven Major Protein Functions Support Structural proteins Movement Contractile proteins Transport (carrier) proteins Buffering Regulation of p. H Metabolic Regulation Enzymes Coordination and Control Hormones Defense Antibodies © 2012 Pearson Education, Inc.
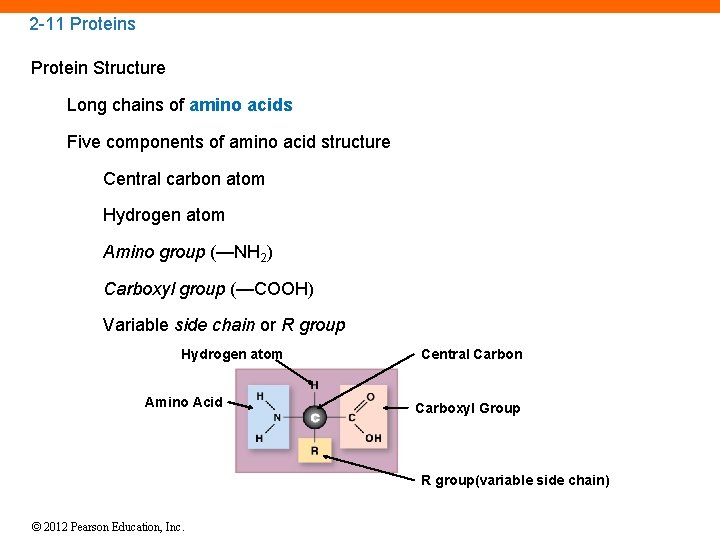
2 -11 Proteins Protein Structure Long chains of amino acids Five components of amino acid structure Central carbon atom Hydrogen atom Amino group (—NH 2) Carboxyl group (—COOH) Variable side chain or R group Hydrogen atom Amino Acid Central Carbon Carboxyl Group R group(variable side chain) © 2012 Pearson Education, Inc.

2 -11 Proteins Hooking Amino Acids Together Requires a dehydration synthesis between: The amino group of one amino acid and the carboxyl group of another amino acid Forms a peptide bond resulting in a peptide Two amino acids – dipeptide Three amino acids – tripeptide Chain of amino acids - polypeptide © 2012 Pearson Education, Inc.
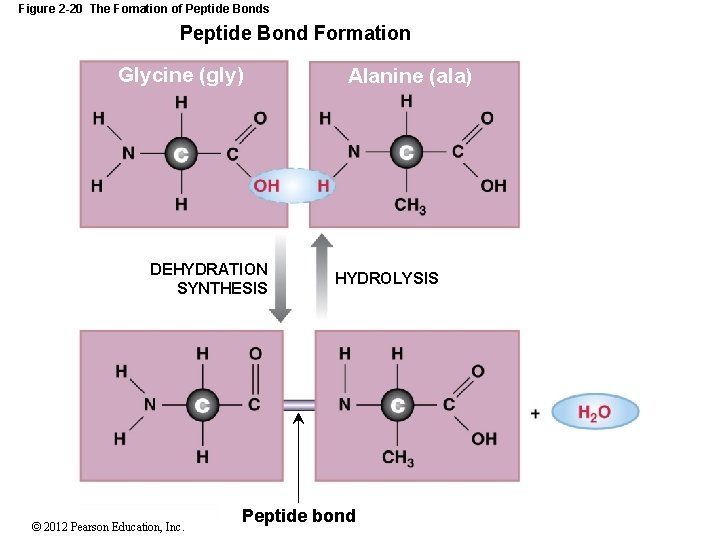
Figure 2 -20 The Fomation of Peptide Bonds Peptide Bond Formation Glycine (gly) DEHYDRATION SYNTHESIS © 2012 Pearson Education, Inc. Alanine (ala) HYDROLYSIS Peptide bond

BIOL 2401 Essential Amino Acids: Arginine Isoleucine Histidine Leucine Methionine Lysine Phenylalanine Tryptophan Threonine Valine These cannot be made by the body and must be obtained in the foods we eat. Non-Essential Amino Acids: Alanine Asparagine Aspartic Acid Cysteine Glutamine Glycine These are made by the body. 12 Differentiate between essential amino acids and non-essential amino acids. © 2012 Pearson Education, Inc.

2 -11 Proteins Protein Shape Primary structure The sequence of amino acids along a polypeptide Secondary structure Hydrogen bonds form spirals or pleats Tertiary structure Secondary structure folds into a unique shape Quaternary structure Final protein shape — several tertiary structures together © 2012 Pearson Education, Inc.
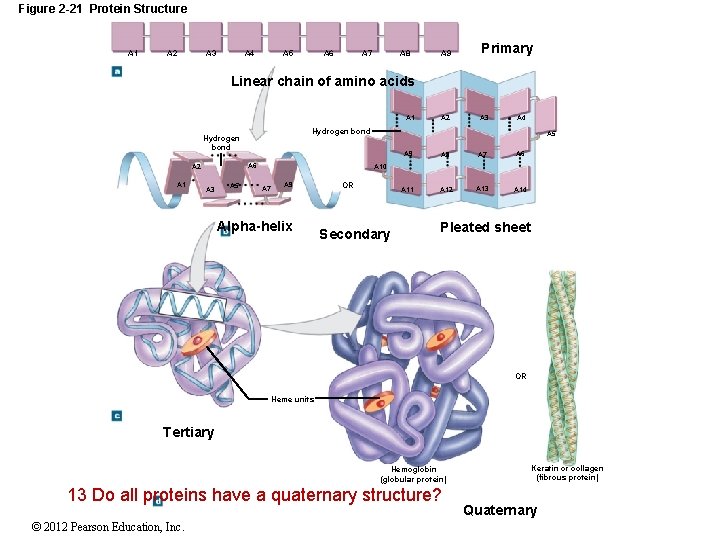
Figure 2 -21 Protein Structure A 1 A 3 A 2 A 5 A 4 A 7 A 6 A 8 A 9 Primary A 2 A 3 Linear chain of amino acids A 1 Hydrogen bond A 6 A 2 A 1 A 3 A 5 A 4 A 5 A 9 A 8 A 7 A 6 A 11 A 12 A 13 A 14 A 10 A 7 A 9 Alpha-helix OR Secondary Pleated sheet OR Heme units Tertiary Hemoglobin (globular protein) 13 Do all proteins have a quaternary structure? © 2012 Pearson Education, Inc. Keratin or collagen (fibrous protein) Quaternary

2 -11 Proteins Classes of proteins are based on overall shape and properties. Fibrous Proteins Structural sheets or strands Globular Proteins Soluble spheres with active functions Protein function is based on shape Shape is based on sequence of amino acids 20 amino acids can be linked in many combinations creating many proteins of varied shape and function. © 2012 Pearson Education, Inc.

2 -11 Proteins Enzyme Function Enzymes are catalysts Proteins that lower the activation energy of a chemical reaction Are not changed or used up in the reaction Enzymes also exhibit: Specificity — will only work on limited types of substrates Saturation Limits — by their concentration Regulation — by other cellular chemicals © 2012 Pearson Education, Inc.
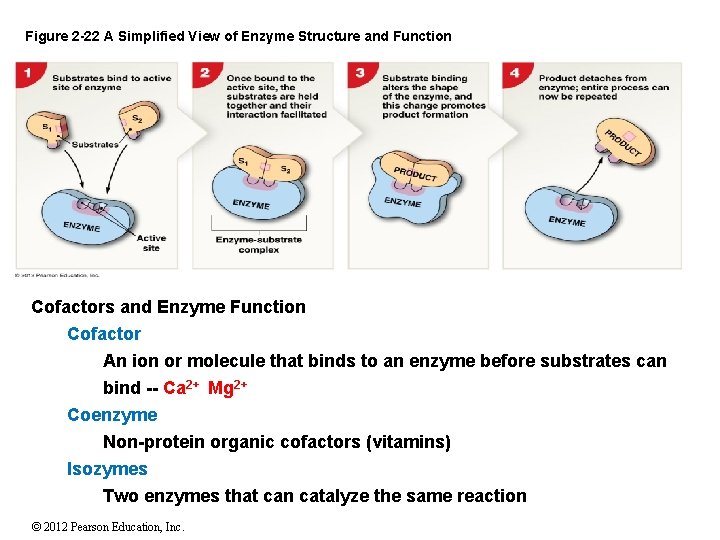
Figure 2 -22 A Simplified View of Enzyme Structure and Function Cofactors and Enzyme Function Cofactor An ion or molecule that binds to an enzyme before substrates can bind -- Ca 2+ Mg 2+ Coenzyme Non-protein organic cofactors (vitamins) Isozymes Two enzymes that can catalyze the same reaction © 2012 Pearson Education, Inc.

2 -11 Proteins Effects of Temperature and p. H on Enzyme Function Denaturation Loss of shape (tertiary or quaternary) and function (becomes non-functional) Due to heat (temperatures above 1100) p. H (acidic or basic depending on enzyme) © 2012 Pearson Education, Inc.

2 -11 Proteins Glycoproteins and Proteoglycans Glycoproteins Large protein + small carbohydrate Includes enzymes, antibodies, hormones, and mucus production Proteoglycans Large polysaccharides + polypeptides Promote viscosity © 2012 Pearson Education, Inc.

2 -12 Nucleic Acids Are large organic molecules, found in the nucleus, which store and process information at the molecular level Deoxyribonucleic acid (DNA) Determines inherited characteristics Directs protein synthesis Controls enzyme production Controls metabolism Ribonucleic acid (RNA) Controls intermediate steps in protein synthesis © 2012 Pearson Education, Inc.
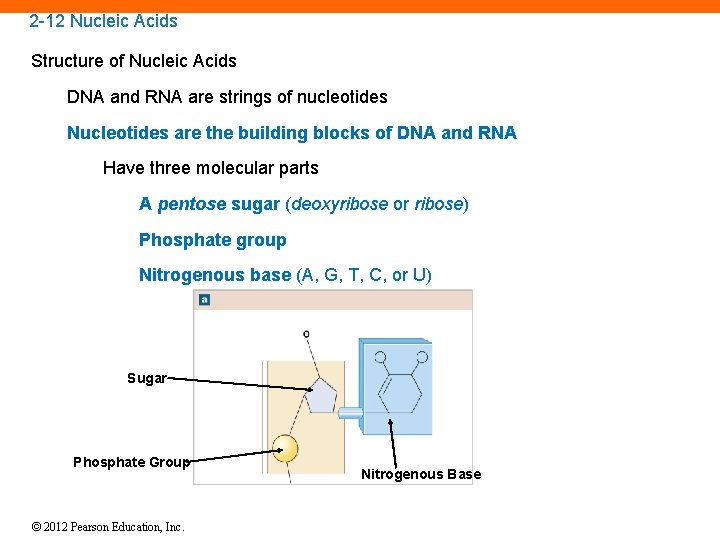
2 -12 Nucleic Acids Structure of Nucleic Acids DNA and RNA are strings of nucleotides Nucleotides are the building blocks of DNA and RNA Have three molecular parts A pentose sugar (deoxyribose or ribose) Phosphate group Nitrogenous base (A, G, T, C, or U) Sugar Phosphate Group © 2012 Pearson Education, Inc. Nitrogenous Base
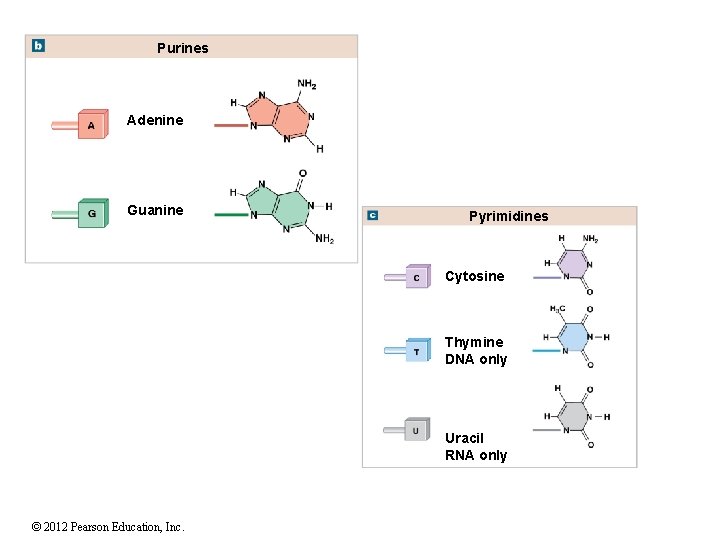
Purines Adenine Guanine Pyrimidines Cytosine Thymine DNA only Uracil RNA only © 2012 Pearson Education, Inc.

2 -12 Nucleic Acids DNA and RNA DNA is double stranded, and the bases form hydrogen bonds to hold the DNA together Sometimes RNA can bind to itself but is usually a single strand DNA forms a twisting double helix Complementary base pairs Purines pair with pyrimidines DNA Adenine (A) and thymine (T) Cytosine (C) and guanine (G) RNA Uracil (U) replaces thymine (T) Types of RNA Messenger RNA (m. RNA) Transfer RNA (t. RNA) Ribosomal RNA (r. RNA) © 2012 Pearson Education, Inc.
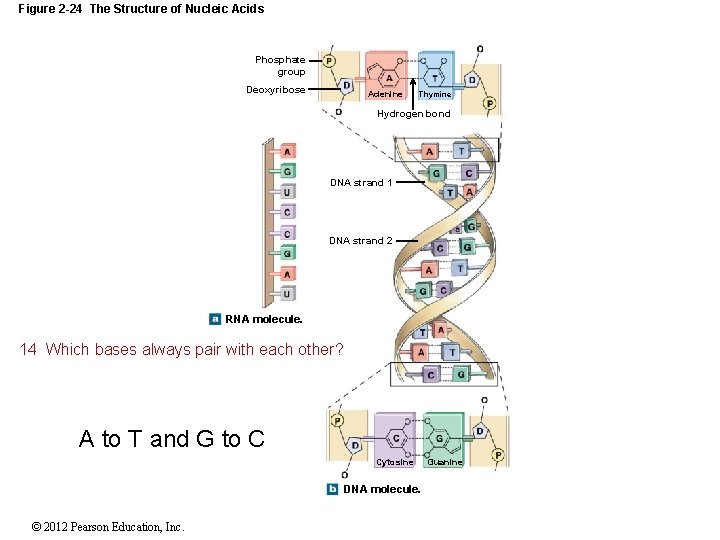
Figure 2 -24 The Structure of Nucleic Acids Phosphate group Deoxyribose Adenine Thymine Hydrogen bond DNA strand 1 DNA strand 2 RNA molecule. 14 Which bases always pair with each other? A to T and G to C Cytosine DNA molecule. © 2012 Pearson Education, Inc. Guanine
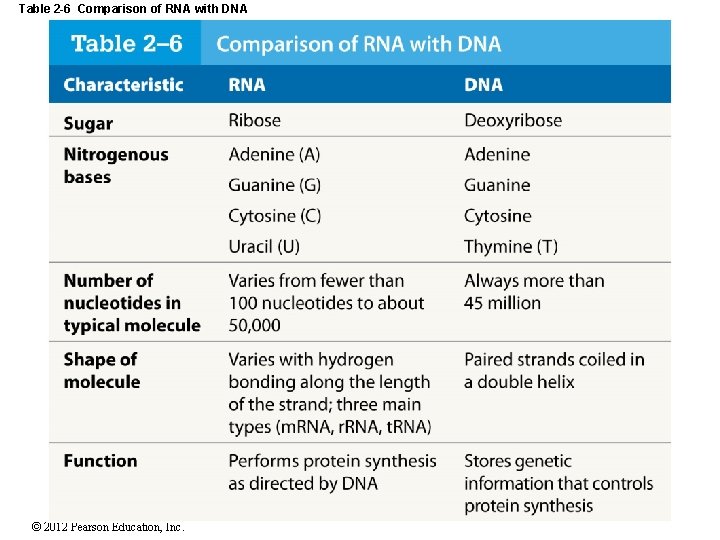
Table 2 -6 Comparison of RNA with DNA © 2012 Pearson Education, Inc.

2 -13 High-Energy Compounds Nucleotides Can Be Used to Store Energy Adenosine diphosphate (ADP) Two phosphate groups; di- = 2 Adenosine triphosphate (ATP) Three phosphate groups; tri- = 3 Phosphorylation Adding a phosphate group to ADP with a high-energy bond to form the high-energy compound ATP Adenosine triphosphatase (ATPase) The enzyme that catalyzes the conversion of ATP to ADP © 2012 Pearson Education, Inc.
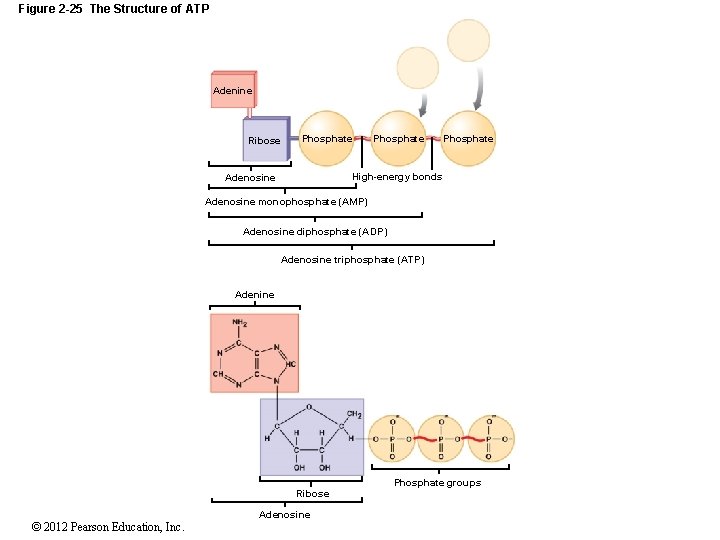
Figure 2 -25 The Structure of ATP Adenine Ribose Phosphate High-energy bonds Adenosine monophosphate (AMP) Adenosine diphosphate (ADP) Adenosine triphosphate (ATP) Adenine Phosphate groups Ribose © 2012 Pearson Education, Inc. Adenosine

2 -14 Chemicals and Cells Biochemical building blocks form functional units called cells Metabolic turnover lets your body grow, change, and adapt to new conditions and activities Your body recycles and renews all of its chemical components at intervals ranging from minutes to years © 2012 Pearson Education, Inc.
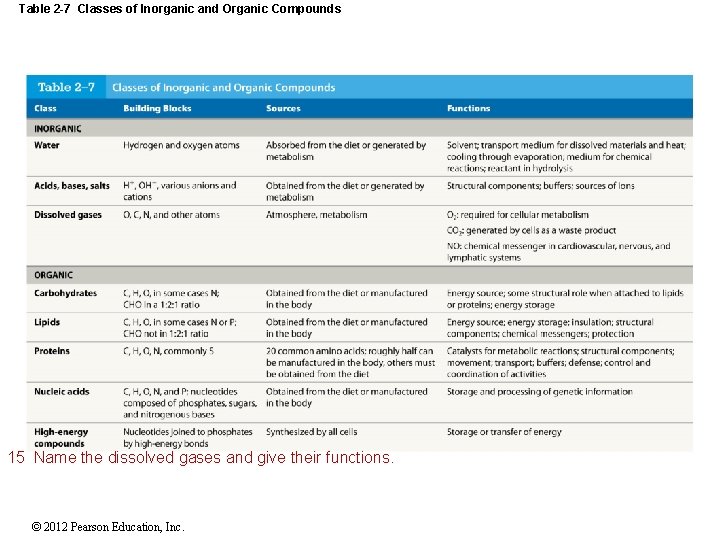
Table 2 -7 Classes of Inorganic and Organic Compounds 15 Name the dissolved gases and give their functions. © 2012 Pearson Education, Inc.
- Slides: 99