BIOL 2207 Clinical Chemistry Introduction to Enzyme Assays

BIOL 2207 Clinical Chemistry Introduction to Enzyme Assays and Clinical Enzymology

Learning Outcomes • After this lecture you should be able to: – Describe how enzyme activity can be measured – Explain how enzymes can be used as diagnostic reagents to measure particular analytes – Discuss the importance of enzyme assay standardisation – Define the units of enzyme activity – List examples of commonly measured serum enzymes – Discuss some of the problems associated with measuring enzyme activity

What I’m going to assume! • Familiarity with, – Basic protein structure • Primary, secondary, tertiary, quaternary structure – Enzyme function • Catalysts – Enzyme kinetics • E + S ↔ ES → E + P

Measuring enzyme activity • The activity of an enzyme can be monitored by following the concentration of substrate or product change with time – Following the progress of the reaction • Substrate will decrease • Product will build up • As enzymes work so fast we generally use an artificial substrate – Slows the reaction down so we can easily measure the rate
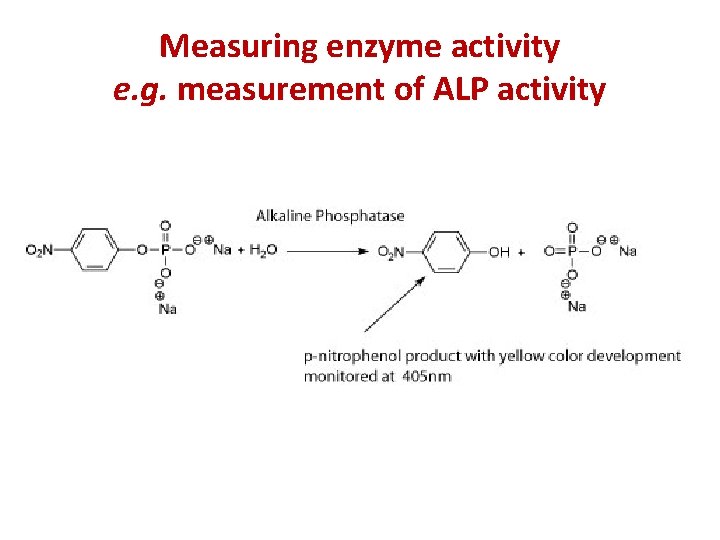
Measuring enzyme activity e. g. measurement of ALP activity
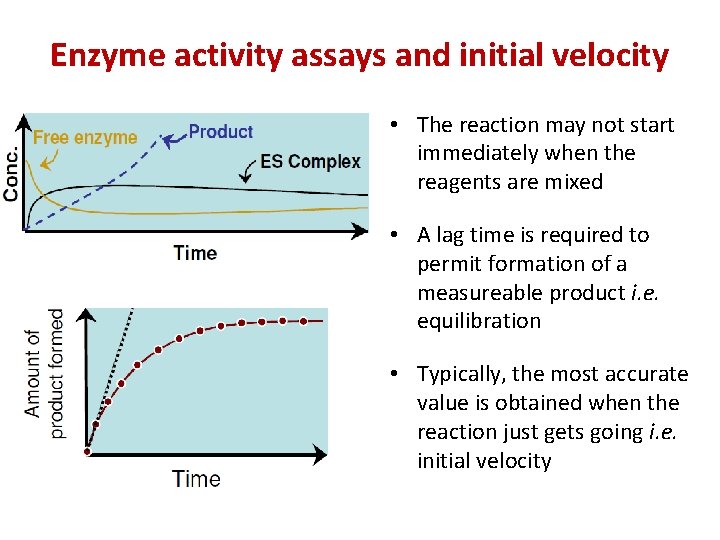
Enzyme activity assays and initial velocity • The reaction may not start immediately when the reagents are mixed • A lag time is required to permit formation of a measureable product i. e. equilibration • Typically, the most accurate value is obtained when the reaction just gets going i. e. initial velocity

Kinetic enzyme assays • We are measuring the change of absorbance over time or the rate at which the absorbance changes • It is critical to measure the activity of the enzyme when the reaction rate depends only on the amount of enzyme present (first order kinetics) • This is the linear phase of the progress curve
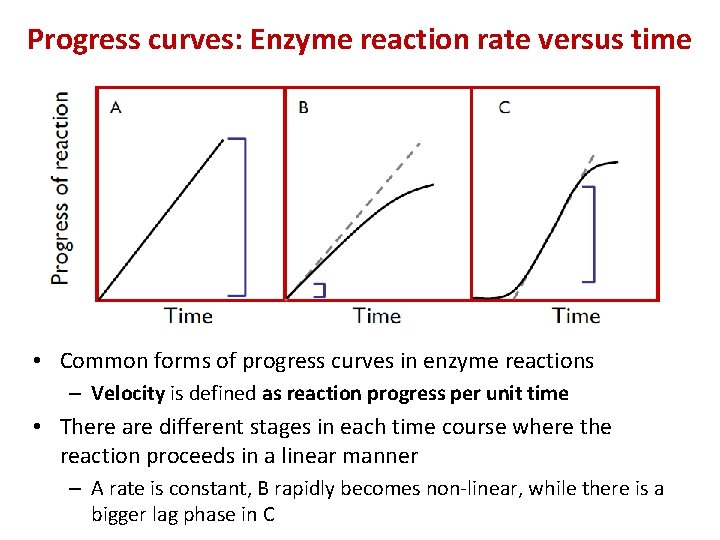
Progress curves: Enzyme reaction rate versus time • Common forms of progress curves in enzyme reactions – Velocity is defined as reaction progress per unit time • There are different stages in each time course where the reaction proceeds in a linear manner – A rate is constant, B rapidly becomes non-linear, while there is a bigger lag phase in C

Coupled enzyme assays • Coupled enzyme systems are common in enzymology • Reactions can be linked together such that a second reaction generates a compound which can be measured spectrophotometrically • The indicator reaction needs to be directly proportional to the rate of reaction (product formation) in the primary reaction Measuring enzyme activity Primary enzyme catalysed reaction Secondary indicator reaction
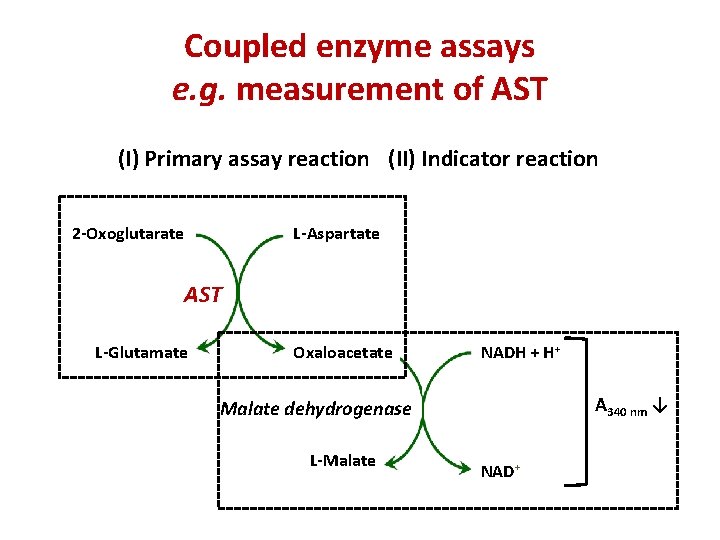
Coupled enzyme assays e. g. measurement of AST (I) Primary assay reaction (II) Indicator reaction L-Aspartate 2 -Oxoglutarate AST L-Glutamate Oxaloacetate NADH + H+ A 340 nm ↓ Malate dehydrogenase L-Malate NAD+

Diagnostic uses of Enzymes Enzyme used in for diagnostic assay Measurement of Urease Urea Uricase Uric Acid Glucose Oxidase Glucose Perioxidase Glucose / Cholesterol Hexokinase Glucose Cholesterol Oxidase Cholesterol Lipase Triglyceride Alkaline Phosphatase ELISA Reverse Transcriptase PCR

Measuring enzyme substrates • Can also exploit the specificity of an enzyme to measure the concentration of a substance – Typically the enzymes substrate • e. g. a common method to measure glucose based on glucose oxidase - peroxidase (GOD-PAP) Glucose oxidase Glucose + 2 H 2 O + O 2 Gluconic acid + 2 H 2 O 2 Peroxidase H 2 O 2 + chromogenic oxygen acceptor Coloured compound + H 2 O

Measuring enzyme substrates • This is an example of an end point method (or equilibrium method) using a coupled system to produce a colour – The glucose oxidase gives the specificity – The peroxidase generates the colour • Note that a trapping reaction is necessary to ensure that the enzyme will quantitatively convert substrate to product • This traps the product and makes the reaction go to completion

Indicator reactions in enzyme assays • In clinical chemistry there are several common chemistries which are used as indicator reactions in coupled assays: – The production of H 2 O 2 by an oxidase is followed by production of the red compound quinoneimine (e. g. used to measure cholesterol, triacylglycerides, glucose) – The consumption of NADH is followed via a decrease in absorbance at 340 nm (e. g. ALT, AST) – Release of the yellow coloured nitroanilide (e. g. GGT, ALP)

Advantages of enzyme assays • Specificity → contrast with chemical methods • Sensitivity → nmol and pmol amounts easy to do • Versatility → using coupled systems we can assay virtually anything • Reproducibility → as long as the same amount of enzyme is added to the tube the reaction rate will be the same • Ease of use → enzymes like mild conditions; most chemical methods need harsh ones

Immobilised enzymes • Enzymes can also be immobilised onto surfaces • Allow for the measurement of the enzymes substrate (specific analytes)
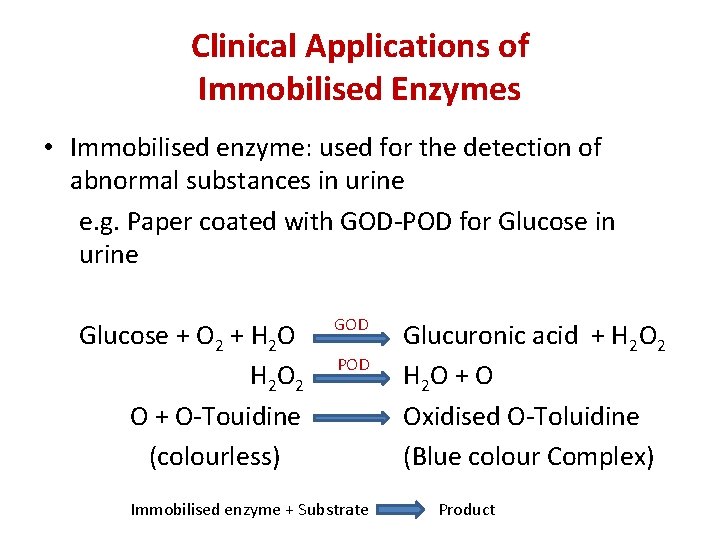
Clinical Applications of Immobilised Enzymes • Immobilised enzyme: used for the detection of abnormal substances in urine e. g. Paper coated with GOD-POD for Glucose in urine Glucose + O 2 + H 2 O H 2 O + O-Touidine (colourless) GOD POD Immobilised enzyme + Substrate Glucuronic acid + H 2 O 2 H 2 O + O Oxidised O-Toluidine (Blue colour Complex) Product

Factors which effect enzyme reactions 1. Enzyme concentration 2. Substrate concentration 3. p. H 4. Temperature 5. Inhibitors and activators 6. Coenzymes and prosthetic groups 7. Timing • To measure enzyme activity (or substrate concentration) reliably all factors that affect the reaction rate have to be optimised and controlled

Enzyme assay standardisation • One analyte may have a large number of different methods or different assay conditions used to measure it • Makes it difficult to compare results from different labs • Makes it more difficult for clinicians to interpret results • Standardisation of assay conditions is critical: – Comparable results – Common units – Common reference ranges – Indication of trueness (accuracy)

Enzyme assay standardisation: How is it done? • Enzyme preparation is generated with known catalytic activity • Used as a standard (reduces variability) • Leaves a path of ‘traceability’; patient results can be traced back to the original standard • Requires co-operation between international bodies, manufacturers, labs

Enzyme assay standardisation: How is it done? • In the interim, manufacturers standardise assay conditions by optimising: – p. H – Temperature – Substrate conditions – Method parameters (kinetic versus endpoint) – Units • In the lab we can ensure that: – Assay protocols are followed – Control material is assayed – Instrumentation is maintained and in proper working order

Enzyme units • Why do we need enzyme units? – Enzymes are not always available in pure form; they are often purified from tissues – e. g. trypsin from the intestine; the greater the activity per unit of protein the more pure the enzyme preparation – As the preparation may not be 100% pure you need to know what the activity of your preparation is i. e. how active the enzyme is

Enzyme units: the katal • The International System of Units (SI units) for enzyme activity is based on two other SI units: – The mole (amount) – The second (unit of time) • The SI unit for catalytic activity is the katal • The katal is the amount of enzyme which will catalyse the transformation of one mole of substrate to product per second or moles/sec • This tends to give very small numbers – nanokatal or microkatal used more often

International units • A more practical unit (which has been kept alive by generations) is the international unit (IU) • This is defined as the amount of enzyme which will catalyse the transformations of 1 micromole of the substrate to product per minute per litre, under standard conditions; mmol/L/min • Converting between the two units: 1 IU = 16. 67 nanokatal

Enzymes appearance in serum Functional serum enzyme responsible for reaction taking place in blood e. g; clotting of blood Non functional serum enzymes do not have their function in blood but they are found in low concentrations in serum due to wear and tear of tissues and normal cell turnover Obstruction to secretory pathway Reflux back into blood because of blockage

Clinical Analysis of Enzymes • The measurement of serum levels of numerous enzymes have been shown to be of clinical significance • Enzymes are normally intracellular and injury or death of tissues can cause the release of tissuespecific enzymes into the bloodstream. • Serum enzyme level may be increased by disease that cause increase in its rate of release or decrease in their rate of excretion.
![Commonly assayed enzymes • • • Transaminases - ALT and AST Alkaline Phosphate [ALP] Commonly assayed enzymes • • • Transaminases - ALT and AST Alkaline Phosphate [ALP]](http://slidetodoc.com/presentation_image/5d03b1f189cadea199170c5ba8347ec3/image-27.jpg)
Commonly assayed enzymes • • • Transaminases - ALT and AST Alkaline Phosphate [ALP] Acid Phosphatases [ACP] Gamma-Glutamyl Transferase [GGT] Amylase [AMY] Lipase [LPS] Creatinine Kinase [CK] Lactic Dehydrogenase [LDH] Prostate specific antigen [PSA]

Isoenzymes • Are enzymes that occur in different molecular forms which differ in their physiochemical properties but catalyze the same chemical reaction • They vary with respect to their kinetic parameters, electrophoretic mobility, and localization • They all have independent action • E. g. Creatinine Kinase (CK) has 3 isoenzymes (CK-MM, CK-MB, CK-BB) • They can be used to identify the specific affected tissues • They can be differentiated from each other and can be clinically quantified in the lab
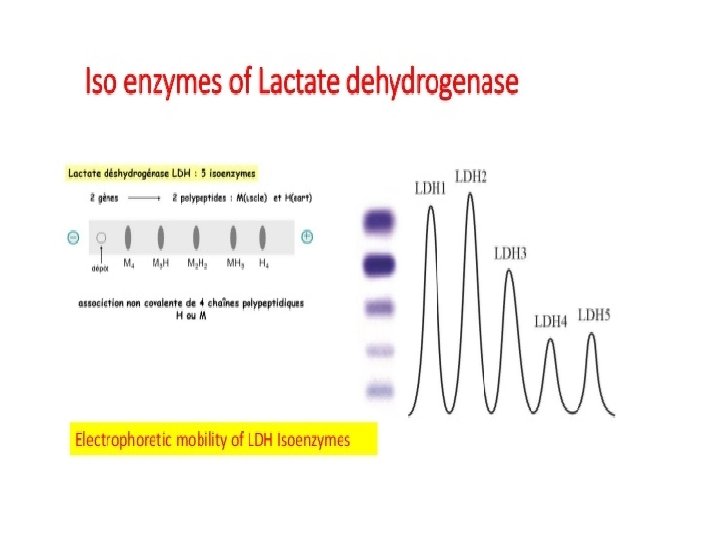

Some problems with measuring enzyme activity • Enzymes can be common to more than one tissue – An increase in the activity of the enzyme could reflect damage to one of several different tissues – Two ways of overcoming this problem 1. Measuring test profiles (more than one analyte associated with the disease) 2. Measuring specific isoforms or isoenzymes • Timing of sample collection is critical – Enzymes will rise and then fall as they are cleared • Macroenzymes (rarely pathological) can cause confusion – Enzymes can form complexes causing reduced clearance

Specificity of Enzymes Greater specificity is achieved in three ways: 1. Interpreting investigations in the light of clinical features: – Jaundice patient with hepatomegaly – ALT in Hepatitis & therefore not Muscle injury 2. Combining measurement with other analytes: - ALP in Cholestasis & bone diseases : - Confirmed by GGT in Cholestasis. 3. Isoenzyme determination: – CK in Muscle injury: – CK -MB in Cardiac Muscle injury:

Genetic Mutations of Enzymes and Diseases Disease Albinism Alkaptonuria Phenylketonuria Homocystinuria Glucose 6 phosphate dehydrogenase Defective Enzyme Tyrokinase Homogenitisate Dioxygenase Phenylalanine mono-oxygenase Cysthathione beta synthase G 6 PD deficiency

Enzymes in Therapy • Substitution of missing production of digestive enzymes (e. g. digestive enzymes) – pepsin, trypsin… • Removal of deposits of death tissue or fibrin (e. g. in lungs, eyes), treatment of skin defects – proteinases, nucleases, collagenase • Clot lysis in myocardial infarction – streptokinase, urokinase
- Slides: 33