BIOL 2207 Clinical Chemistry BSc Biomedical Science BSc

BIOL 2207 - Clinical Chemistry BSc Biomedical Science BSc Biomedical & Molecular Diagnostics BSc Human Nutrition & Dietetics BSc Public Health Nutrition
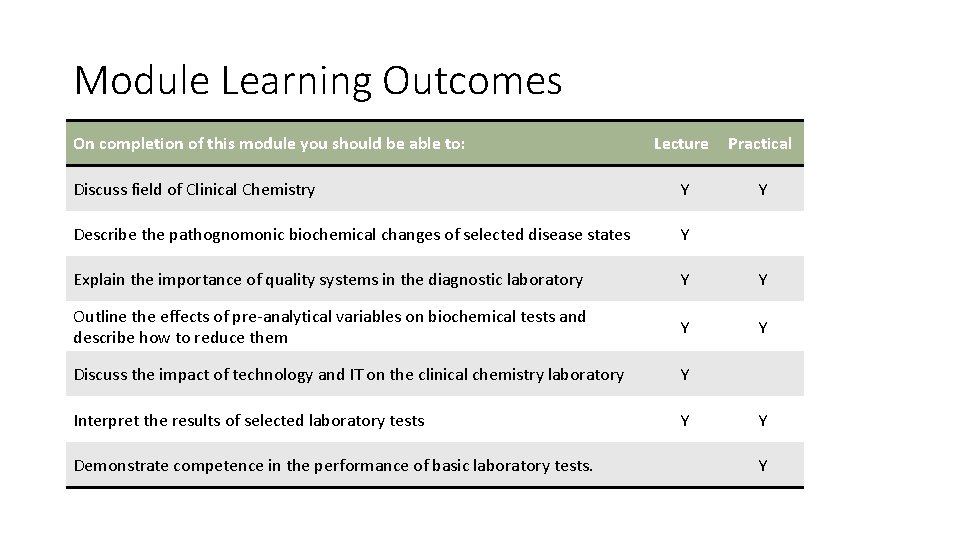
Module Learning Outcomes On completion of this module you should be able to: Lecture Practical Discuss field of Clinical Chemistry Y Y Describe the pathognomonic biochemical changes of selected disease states Y Explain the importance of quality systems in the diagnostic laboratory Y Y Outline the effects of pre-analytical variables on biochemical tests and describe how to reduce them Y Y Discuss the impact of technology and IT on the clinical chemistry laboratory Y Interpret the results of selected laboratory tests Y Demonstrate competence in the performance of basic laboratory tests. Y Y

Why do you carry out a test? Confirm a clinical suspicion (i. e. symptoms present) Exclude a diagnosis Assist in treatment (selection, optimisation, monitoring) Prognosis Screening (i. e. no symptoms)

Clinical chemistry in a snapshot Estimate concentrations or activities of substances, ions, molecules, complexes in body fluids like whole blood, plasma, serum, urine and cerebrospinal fluid and Comparing the results to the range of values expected for healthy people

Test results ≠ clinical outcomes Right test, right specimen, with right results and right interpretation at right time
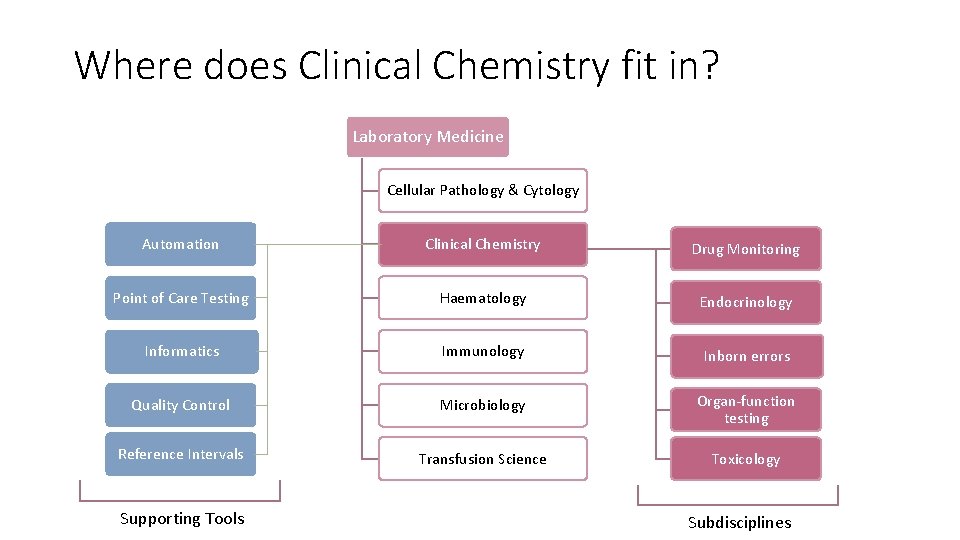
Where does Clinical Chemistry fit in? Laboratory Medicine Cellular Pathology & Cytology Clinical chemistry Automation Clinical Chemistry Point of Care Testing Haematology Endocrinology Informatics Immunology Inborn errors Quality Control Microbiology Organ-function testing Reference Intervals Transfusion Science Toxicology Supporting Tools Drug Monitoring Subdisciplines
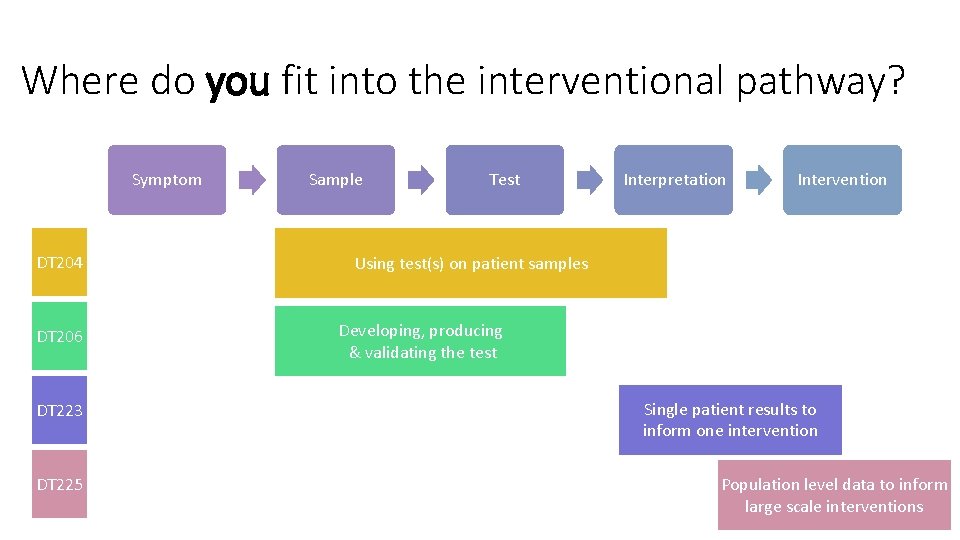
Where do you fit into the interventional pathway? Symptom DT 204 DT 206 DT 223 DT 225 Sample Test Interpretation Intervention Using test(s) on patient samples Developing, producing & validating the test Single patient results to inform one intervention Population level data to inform large scale interventions
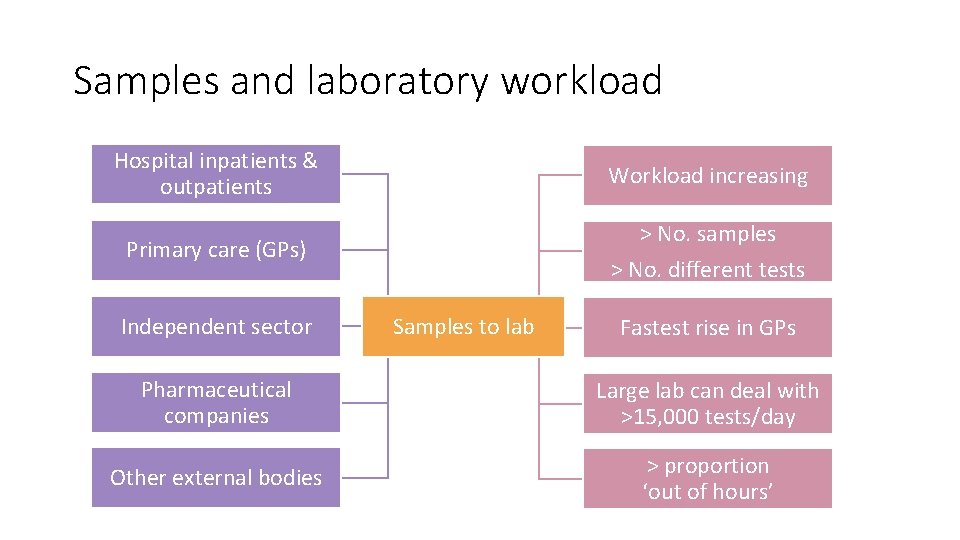
Samples and laboratory workload Hospital inpatients & outpatients Workload increasing Primary care (GPs) > No. samples > No. different tests Independent sector Samples to lab Fastest rise in GPs Pharmaceutical companies Large lab can deal with >15, 000 tests/day Other external bodies > proportion ‘out of hours’

Automation is now critical for the laboratory medicine – in particular the Clinical Chemistry Lab Integrated modular system Modules have different functions: capping/decapping, centrifugation, analysis, control etc.
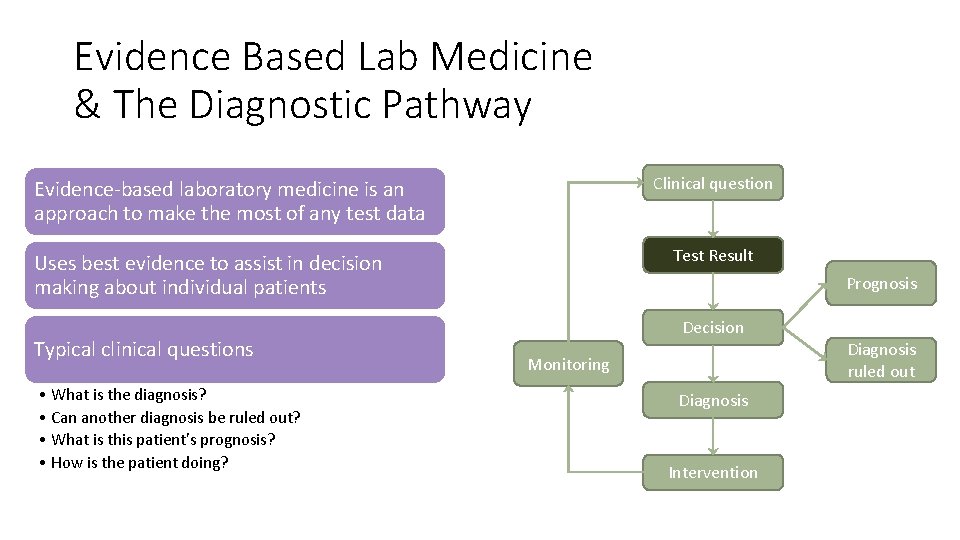
Evidence Based Lab Medicine & The Diagnostic Pathway Clinical question Evidence-based laboratory medicine is an approach to make the most of any test data Test Result Uses best evidence to assist in decision making about individual patients Typical clinical questions • What is the diagnosis? • Can another diagnosis be ruled out? • What is this patient's prognosis? • How is the patient doing? Prognosis Decision Monitoring Diagnosis Intervention Diagnosis ruled out

What does the test result look like? • Initial demographic details - As per original request form • Results - From laboratory analysis • Reference ranges - Derived from scientific knowledge • Comments & advice - From experience & algorithms

How do you know that it’s a believable result? Stringent controls are in place in the laboratory to support test quality Diagnostic labs accredited by external body (INAB) to ensure quality Must have systems to ensure ‘good quality’ results • Standard Operating Procedures (SOPs) • Quality Control Process • External Quality Assurance • Knowledge of potential sources of error Aim is to achieve a ‘six sigma’ approach – 99. 99966% of perfection • 3. 4 errors per million tests • As good as it gets

Performance measures for tests/methods Accuracy • How close is the test to the true value? Precision • How close are repeated measurements to one another? Linearity • Is the response proportional to the analyte? Interference • Is the test effected by haemolysis, icterus, lipaemia? Analytical sensitivity • What is the smallest amount that you can measure? Analytical specificity • Does the method measure the correct analyte?
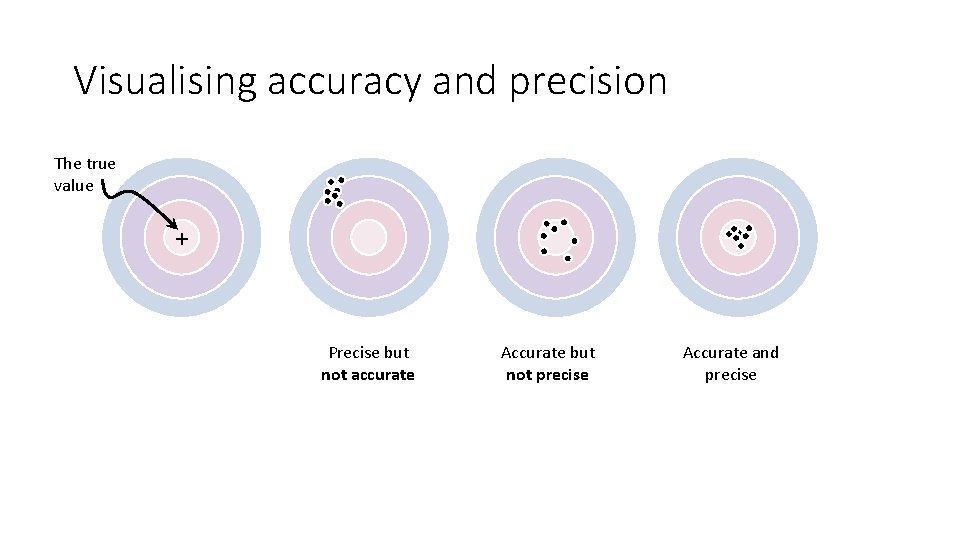
Visualising accuracy and precision The true value + Precise but not accurate Accurate but not precise Accurate and precise
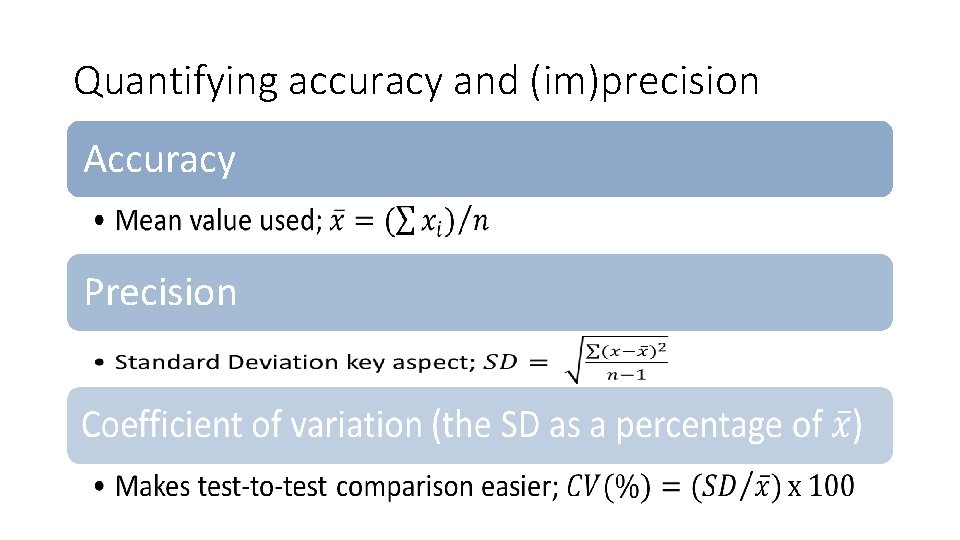
Quantifying accuracy and (im)precision Accuracy • Precision • •

How would you test accuracy and precision?
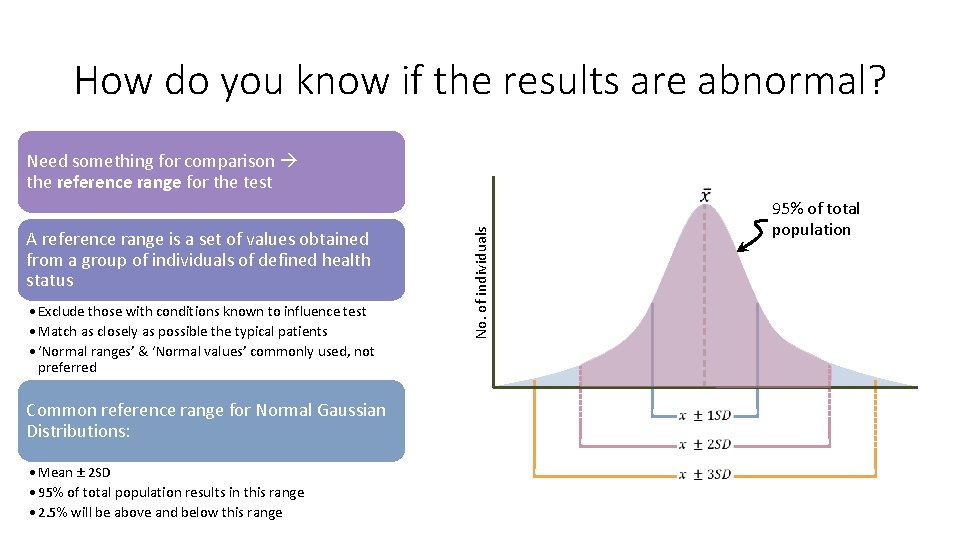
How do you know if the results are abnormal? Need something for comparison the reference range for the test • Exclude those with conditions known to influence test • Match as closely as possible the typical patients • ‘Normal ranges’ & ‘Normal values’ commonly used, not preferred No. of individuals A reference range is a set of values obtained from a group of individuals of defined health status Common reference range for Normal Gaussian Distributions: • Mean ± 2 SD • 95% of total population results in this range • 2. 5% will be above and below this range 95% of total population
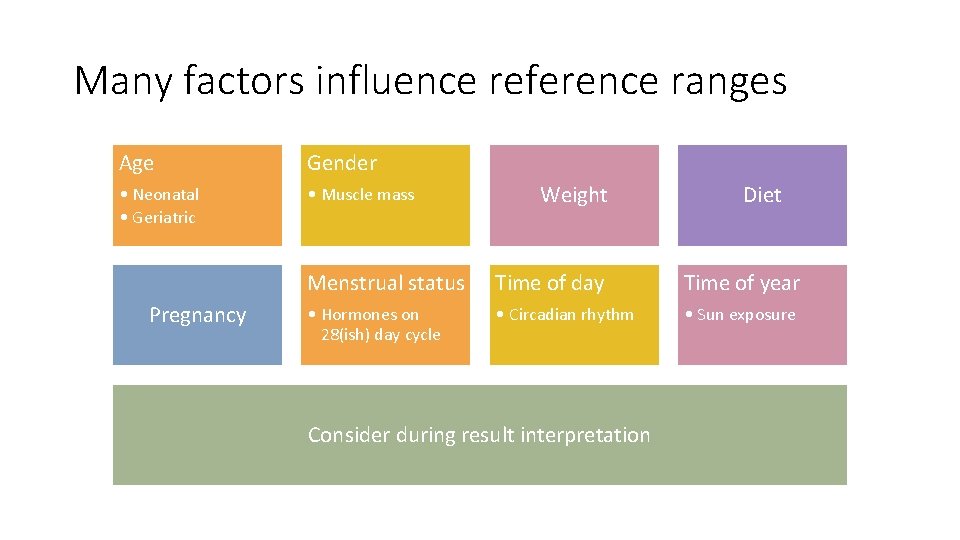
Many factors influence reference ranges Age Gender • Neonatal • Geriatric • Muscle mass Pregnancy Weight Diet Menstrual status Time of day Time of year • Hormones on 28(ish) day cycle • Circadian rhythm • Sun exposure Consider during result interpretation
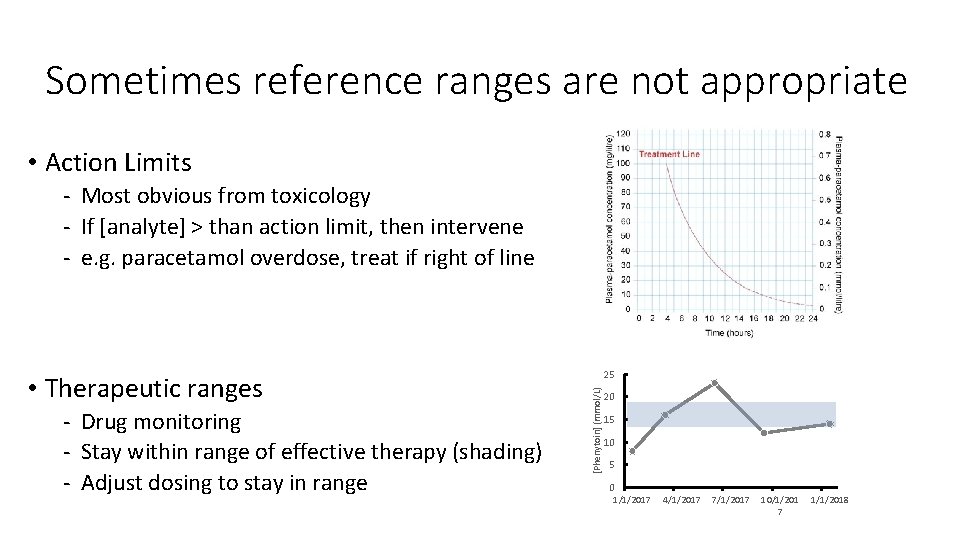
Sometimes reference ranges are not appropriate • Action Limits - Most obvious from toxicology - If [analyte] > than action limit, then intervene - e. g. paracetamol overdose, treat if right of line - Drug monitoring - Stay within range of effective therapy (shading) - Adjust dosing to stay in range [Phenytoin] (mmol/L) • Therapeutic ranges 25 20 15 10 5 0 1/1/2017 4/1/2017 7/1/2017 10/1/201 7 1/1/2018
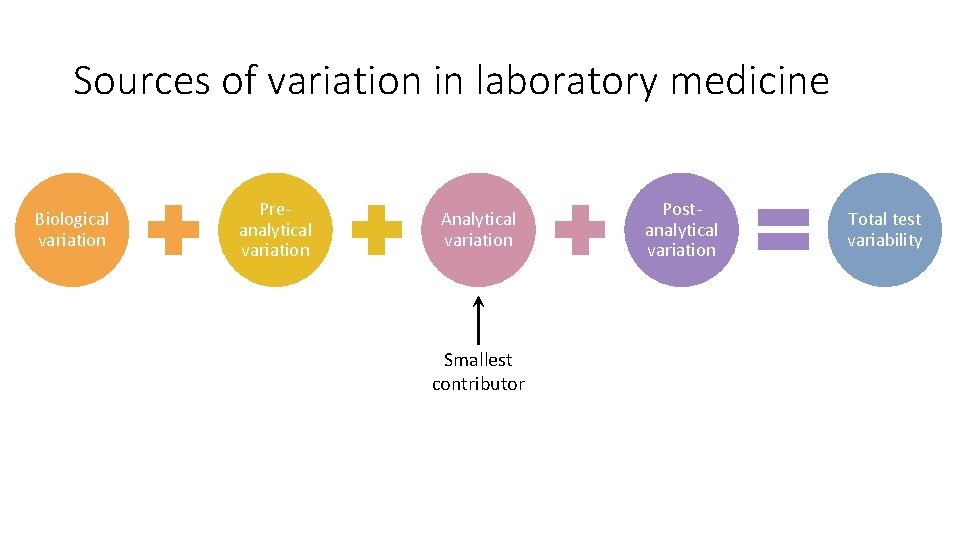
Sources of variation in laboratory medicine Biological variation Preanalytical variation Analytical variation Smallest contributor Postanalytical variation Total test variability
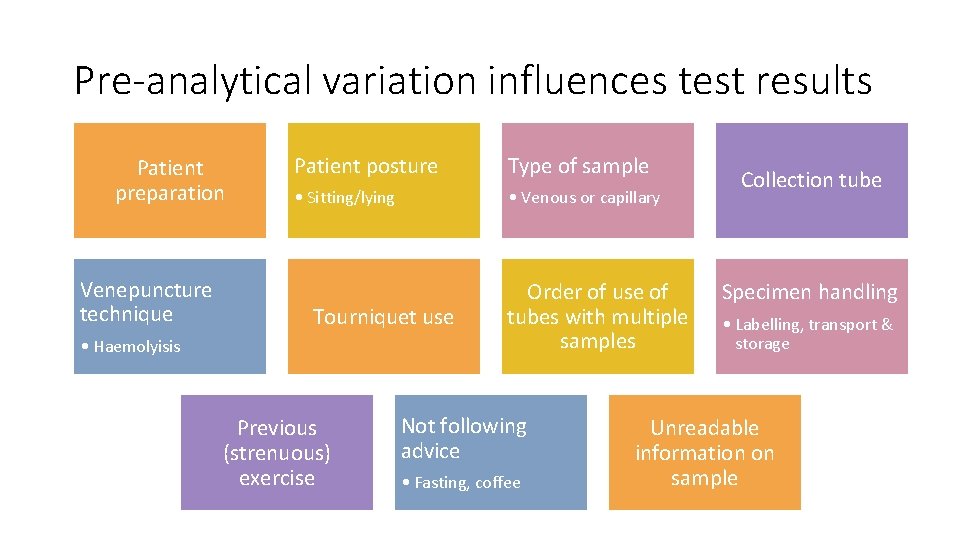
Pre-analytical variation influences test results Patient preparation Venepuncture technique Patient posture Type of sample • Sitting/lying • Venous or capillary Tourniquet use • Haemolyisis Previous (strenuous) exercise Order of use of tubes with multiple samples Not following advice • Fasting, coffee Collection tube Specimen handling • Labelling, transport & storage Unreadable information on sample

Many analytes vary significantly biologically • Not always recognised - Source of error in serial measurement (e. g. in patient) • Interindividual - A patient compared to a population mean • Intraindividual - A patient compared to themselves - Usually smaller than interindividual Examples of Intraindividual variation • Bilirubin drops during the day • Triglyceride, phosphate, urea, creatinine are rise during day (lowest am) • Morning Hb values usually highest • WBC count usually highest in afternoon

Panel approaches used to overcome limitations of individual tests Combining multiple tests to address a clinical question makes overall result more robust BUT More tests more chance of finding a result outside of the reference range by chance when nothing is wrong This is known as a false positive Sequential tests also used First and second line tests
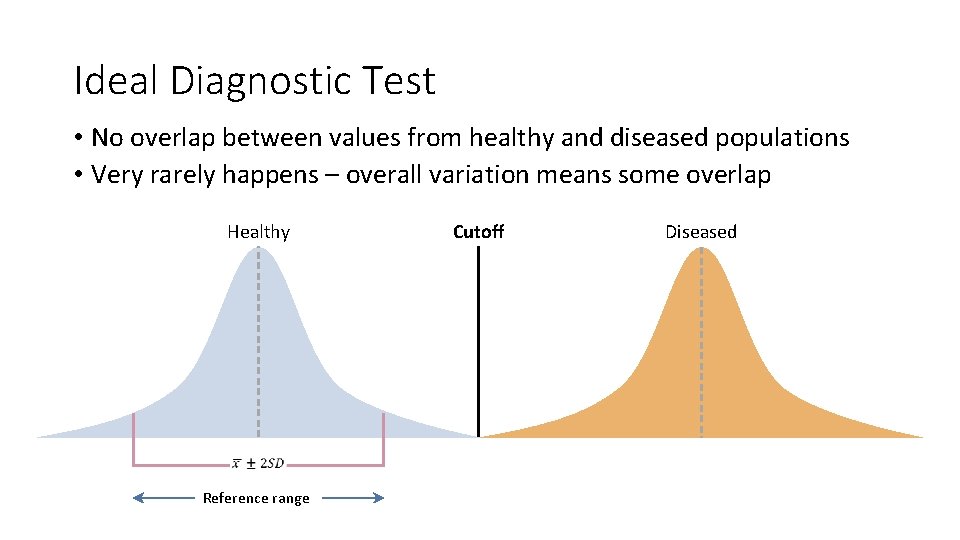
Ideal Diagnostic Test • No overlap between values from healthy and diseased populations • Very rarely happens – overall variation means some overlap Healthy Reference range Cutoff Diseased

Overlap is a more common situation • Generates false positives and false negatives • Changing cut off will alter relative proportions of each category Healthy Diseased Cutoff TN - True Negatives TP - True Positives FN FP False Negatives Negative result but diseased False Positives Positive result but healthy

Diagnostic sensitivity and specificity • Different from the previous analytical parameters - Diagnostic sensitivity – frequency of positive test result when disease present - Diagnostic specificity – frequency of negative test result when disease absent • Very important to clinicians - Impacts the predictive value of a test (and thus the decision making) - More about predictive values later Sens = TP/(TP + FN) i. e. test positive/all positive results (aka True positive rate) Spec = TN/(TN+FP) i. e. test negative/all negative results (aka True negative rate)
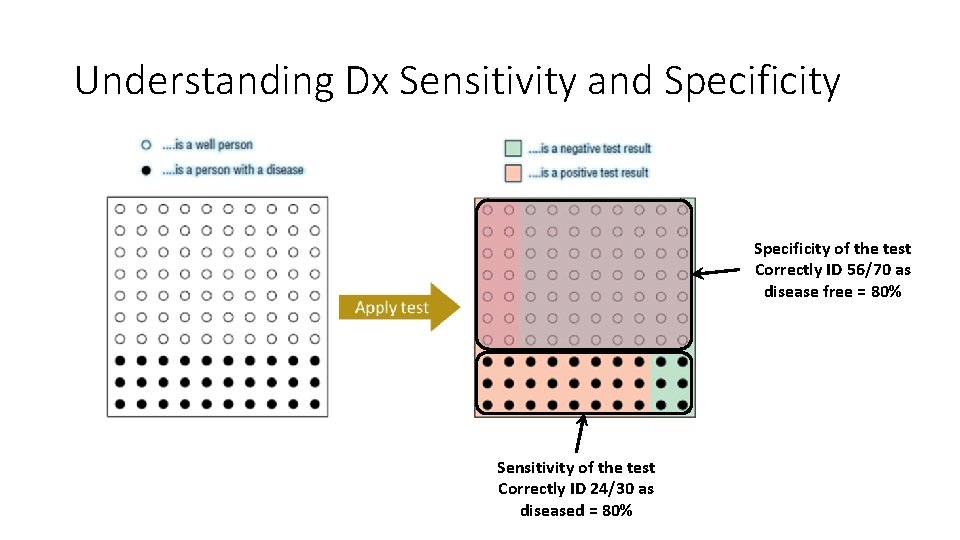
Understanding Dx Sensitivity and Specificity of the test Correctly ID 56/70 as disease free = 80% Sensitivity of the test Correctly ID 24/30 as diseased = 80%
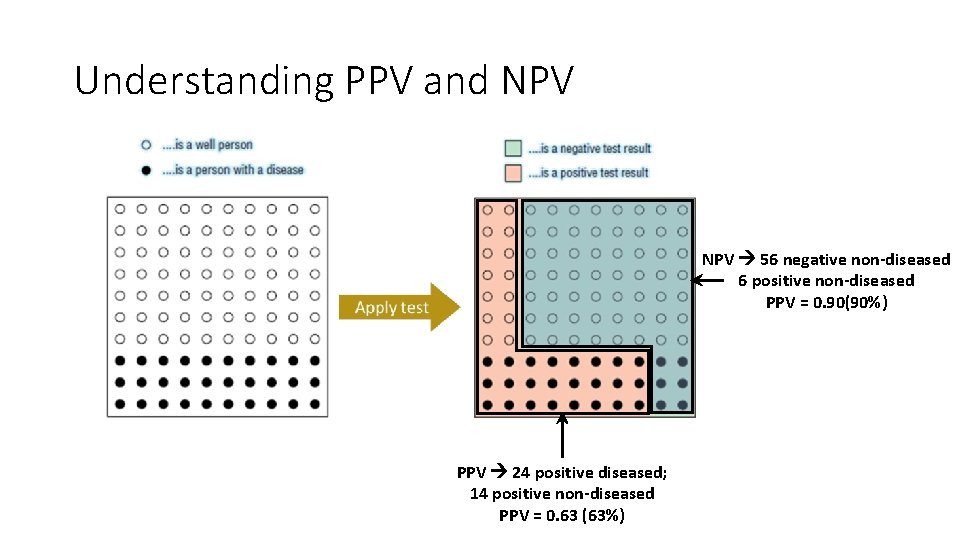
Understanding PPV and NPV 56 negative non-diseased 6 positive non-diseased PPV = 0. 90(90%) PPV 24 positive diseased; 14 positive non-diseased PPV = 0. 63 (63%)
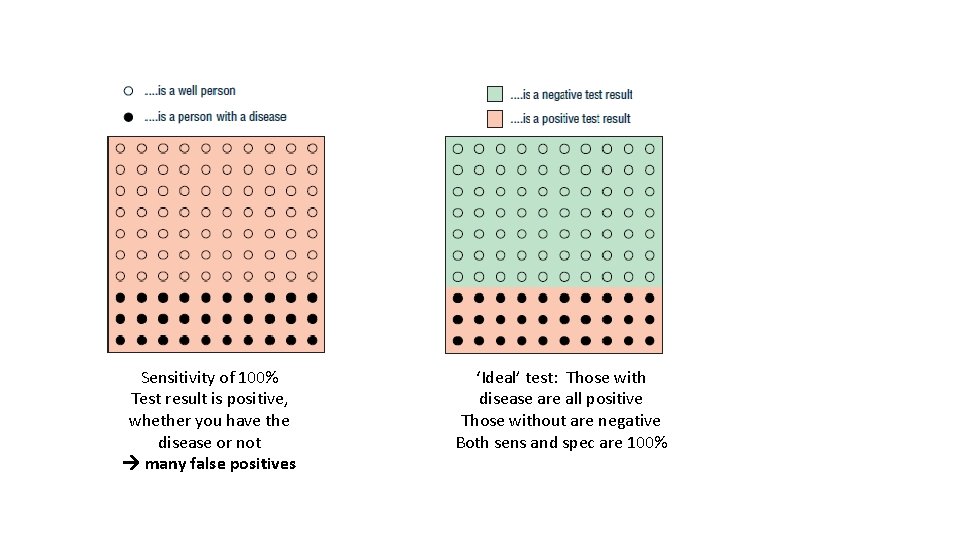
Sensitivity of 100% Test result is positive, whether you have the disease or not many false positives ‘Ideal’ test: Those with disease are all positive Those without are negative Both sens and spec are 100%

Limitations of Dx Sensitivity and Specificity • Sensitivity helps to rule out • Specificity helps to rule in • Both calculated on basis of a diagnosis (i. e. TP, TN) - Not as useful for diagnostics • Use predictive values instead - Positive predictive value (PPV) - Negative predictive value (NPV) • Answers the question: - What is the probability that a patient’s results indicate the presence of disease? PPV = TP/(TP + FP) x 100 probability a person has the disease if they have a positive test result (test pos. with disease) NPV = TN/(TN + FN) x 100 probability a person is disease free if they have a negative test result (test neg. without disease
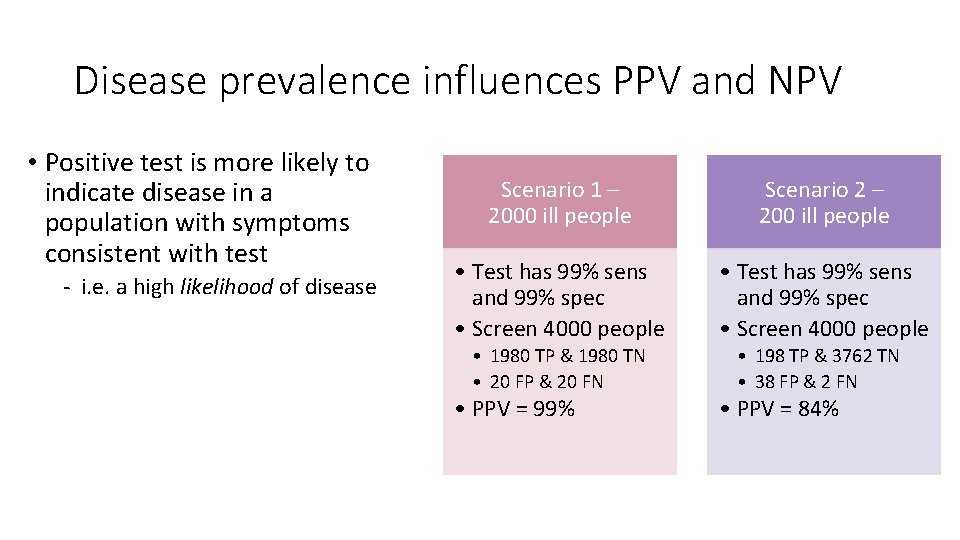
Disease prevalence influences PPV and NPV • Positive test is more likely to indicate disease in a population with symptoms consistent with test - i. e. a high likelihood of disease Scenario 1 – 2000 ill people Scenario 2 – 200 ill people • Test has 99% sens and 99% spec • Screen 4000 people • 1980 TP & 1980 TN • 20 FP & 20 FN • 198 TP & 3762 TN • 38 FP & 2 FN • PPV = 99% • PPV = 84%
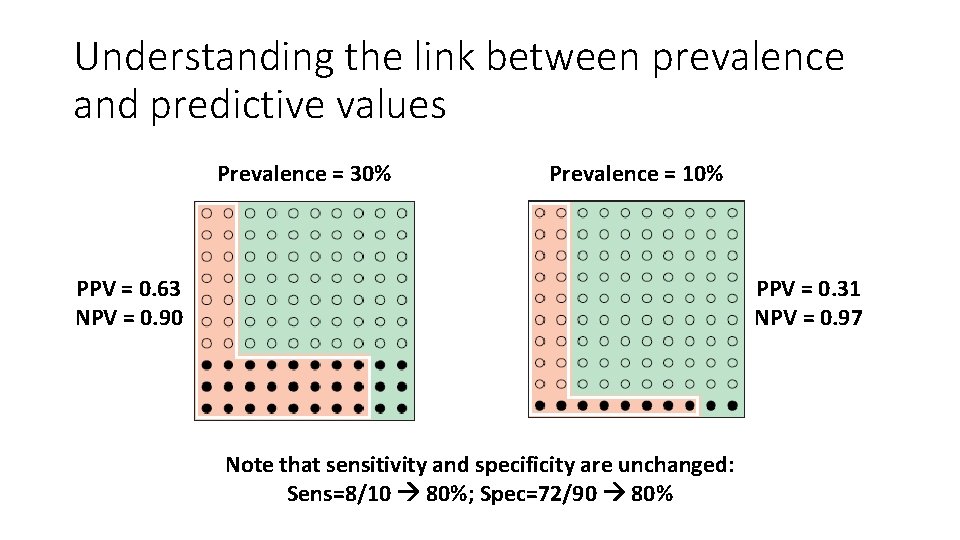
Understanding the link between prevalence and predictive values Prevalence = 30% Prevalence = 10% PPV = 0. 63 NPV = 0. 90 PPV = 0. 31 NPV = 0. 97 Note that sensitivity and specificity are unchanged: Sens=8/10 80%; Spec=72/90 80%
Take Home Message
- Slides: 33