BIOL 204 Lab For Week 9 Respiratory System

BIOL 204 Lab For Week 9 Respiratory System Physiology
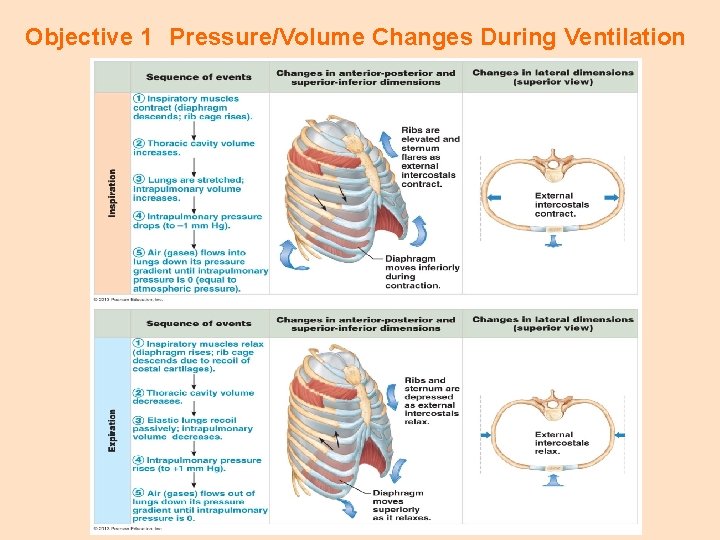
Objective 1 Pressure/Volume Changes During Ventilation
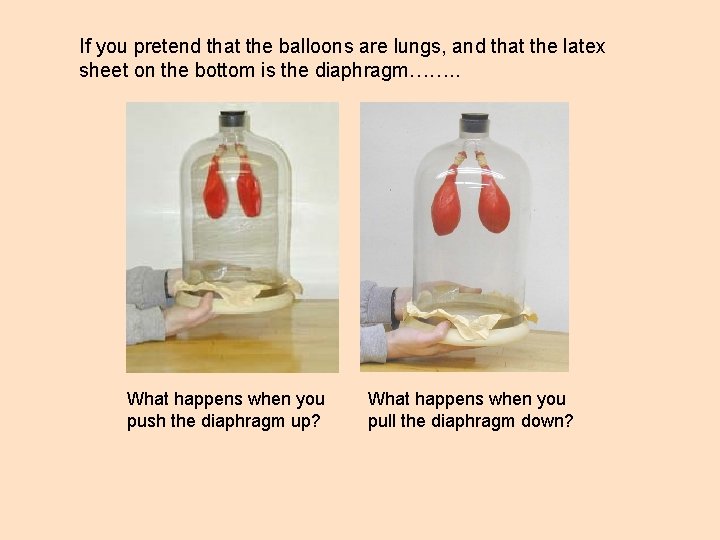
If you pretend that the balloons are lungs, and that the latex sheet on the bottom is the diaphragm……. . What happens when you push the diaphragm up? What happens when you pull the diaphragm down?
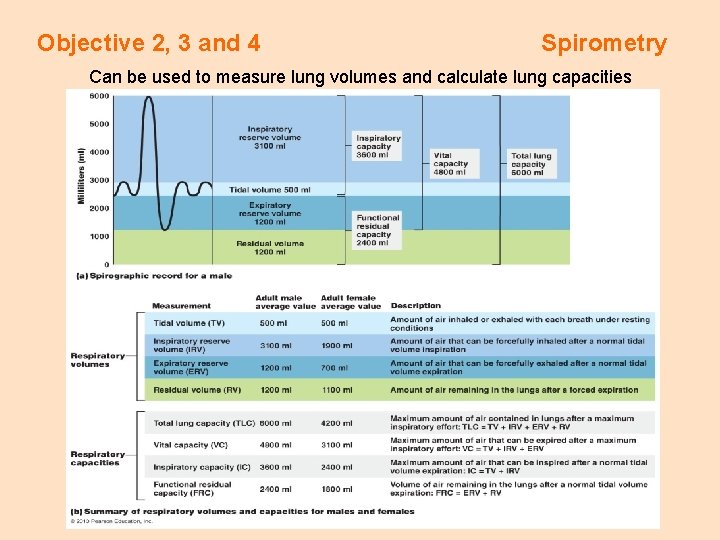
Objective 2, 3 and 4 Spirometry Can be used to measure lung volumes and calculate lung capacities

A Hand Held Spirometer is used to measure 1. tidal volume (TV) 2. expiratory reserve volume (ERV) 3. vital capacity (VC)
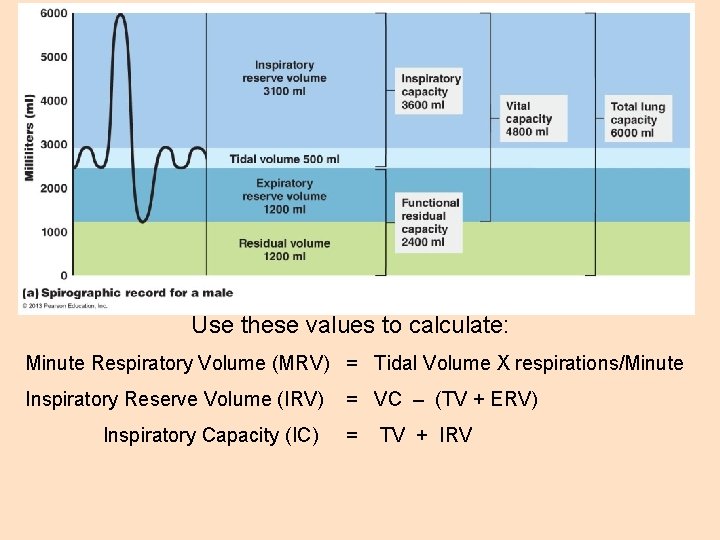
Use these values to calculate: Minute Respiratory Volume (MRV) = Tidal Volume X respirations/Minute Inspiratory Reserve Volume (IRV) Inspiratory Capacity (IC) = VC – (TV + ERV) = TV + IRV

Predicted Vital Capacity Males = 0. 052 (height) - 0. 022 (age) - 3. 60 Females = 0. 041 (height) - 0. 018 (age) - 2. 69 VC = vital capacity in liters H = height in centimeters A = age in years Conversions: 2. 54 cm per 1 inch 1000 ml per 1 liter average measured VC % of predicted vital capacity = X 100 predicted value (from tables)

Objective 5 Iworx- Spirometry Familiarize yourself with the Iworx hardware and the operation of the Labscribe software. You can find the relevant materials on pages 9. 7 to 9. 15 of the BIOL 204 Lab Manual. The pictures on the next couple of slides show a normal spirometry recording would look like and the various lung volumes and capacities.
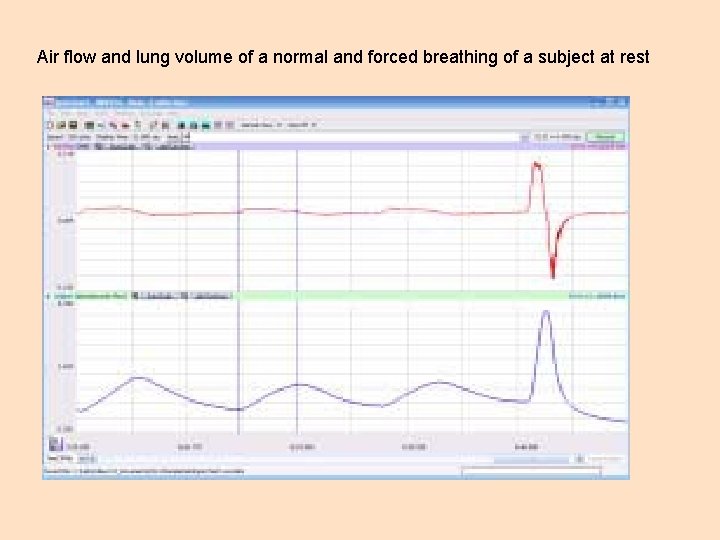
Air flow and lung volume of a normal and forced breathing of a subject at rest
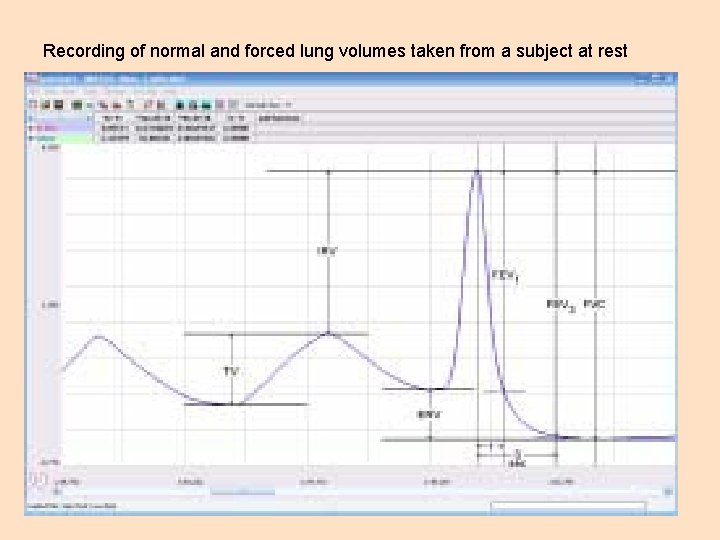
Recording of normal and forced lung volumes taken from a subject at rest
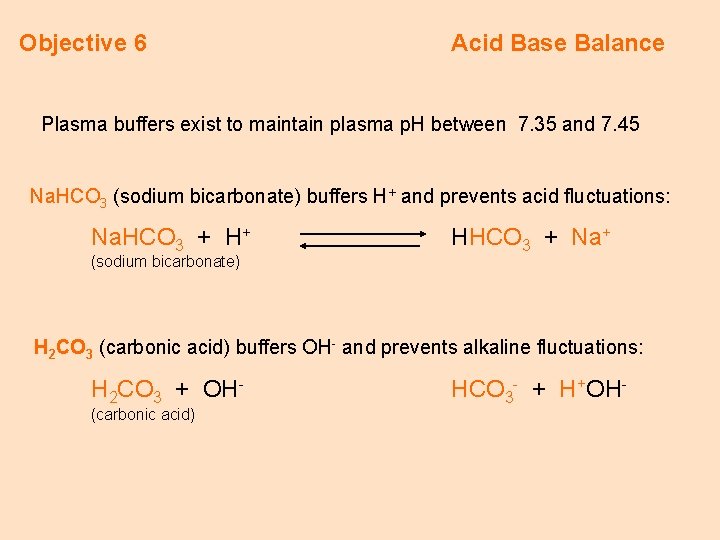
Objective 6 Acid Base Balance Plasma buffers exist to maintain plasma p. H between 7. 35 and 7. 45 Na. HCO 3 (sodium bicarbonate) buffers H+ and prevents acid fluctuations: Na. HCO 3 + H+ (sodium bicarbonate) HHCO 3 + Na+ H 2 CO 3 (carbonic acid) buffers OH- and prevents alkaline fluctuations: H 2 CO 3 + OH(carbonic acid) HCO 3 - + H+OH-

The respiratory system controls plasma levels of CO 2 and thus controls plasma levels of H 2 CO 3: Hypoventilation leads to increased H 2 CO 3 and decreased p. H and Hyperventilation leads to decreased H 2 CO 3 and increased p. H
- Slides: 12