BIOINORGANIC CHEMISTRY Presentation By Sitanshu Kumar Introduction Bioinorganic

BIOINORGANIC CHEMISTRY Presentation By: Sitanshu Kumar

Introduction Bioinorganic chemistry is concerned with the roles of inorganic elements in biological processes. Metal ions can have structural roles, catalytic roles, or both. Metals that have catalytic roles will be present at the active site of the biomolecule which will likely be a metalloprotein (a metalloenzyme). The reactivity of a metalloprotein is defined by the nature of the metal, particularly its electronic structure and oxidation state. This, in turn, is determined by its coordination environment (ligand donor atoms) and molecular geometry, which is provided by the architecture of the protein surrounding the metal.

Biologically Important Elements • 99% of human body is comprised of 11 elements: • Bulk biological elements: H, C, N, O, P, S, Cl (as PO 43 -, SO 42 -, Cl-) • Bulk metal ion nutrients: Na, Mg, K, Ca • Essential elements for a wide range of bacteria/plants/animals • Transition metals: V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Mo • Non-Metals: (B), F, (Si), Se I, F.

Some Other Facts • Mammals are believed to use only 25 of the known elements. • Eleven non-transition elements that make up 99. 9% of the human body (O, C, H, N, Ca, P, S, K, Cl, Na, Mg), • Three transition metals, Fe, Zn and Cu are needed in significant amounts. • “Trace quantities” of many other transition elements are required to maintain proper physical functioning. • Other elements in the human body (e. g. Rb, Zr, Sr, Al, Pb, Ba) are not essential but incorporated inadvertently because they share chemical and physical properties with essential elements. • Other elements are added to the list of elements thought to be essential as our knowledge of the chemistry of living systems increases.

Symptoms of Elemental Deficiency in Humans Ca Mg Fe Zn Cu Mn Mo Co Ni Cr Si F I Se As Retarded skeletal growth Muscle cramps Anemia, immune disorders Stunted growth, skin damage, retarded maturation Liver disorders, secondary anemia Infertility, impaired skeletal growth Retarded cellular growth Pernicious anemia Depressed growth, dermatitis Diabetes symptoms Skeletal growth disorders Dental disorders Thyroid disorders Cardiac muscular weakness Impaired growth (in animals)

Biological Roles of Metallic Elements Structural Skeletal roles via biomineralization Ca 2+, Mg 2+, P, O, C, Si, S, F as anions, e. g. PO 43 , CO 32. Charge neutralization. Mg 2+, Ca 2+ to offset charge on DNA - phosphate anions Charge carriers: Na+, K+, Ca 2+ Transmembrane concentration gradients ("ion-pumps and channels") Trigger mechanisms in muscle contraction (Ca). Electrical impulses in nerves (Na, K) Heart rhythm (K). Hydrolytic Catalysts: Zn 2+ , Mg 2+ Lewis acid/Lewis base Catalytic roles. Small labile metals. Redox Catalysts: Fe(II)/Fe(IV), Cu(I)/Cu(II), Mn(II)/Mn(III)/(Mn(IV), Mo(IV)/Mo(VI), Co(I)/Co(III) Transition metals with multiple oxidation states facilitate electron transfer - energy transfer. Biological ligands can stabilize metals in unusual oxidation states and fine tune redox potentials. Activators of small molecules. Transport and storage of O 2 (Fe, Cu) Fixation of nitrogen (Mo, Fe, V) Reduction of CO 2 (Ni, Fe) Organometallic Transformations. Cobalamins, B 12 coenzymes (Co), Aconitase (Fe-S)

Transition Metals in Biomolecules Iron. Most abundant metal in biology, used by all plants and animals including bacteria. Some roles duplicated by other metals, while others are unique to Fe. Iron use has survived the evolution of the O 2 atmosphere on earth and the instability of Fe(II) with respect to oxidation to Fe(III). Zinc. Relatively abundant metal. Major concentration in metallothionein (which also serves as a reservoir for other metals, e. g. Cd, Cu, Hg). Many well characterized Zn proteins, including redox proteins, hydrolases and nucleic acid binding proteins. Copper Often participatse together with Fe in proteins or has equivalent redox roles in same biological reactions. Reversible O 2 binding, O 2 activation, electron transfer, O 2 - dismutation (SOD). Cobalt. Unique biological role in cobalamin (B 12 -coenzymes) isomerization reactions. Manganese Critical role in photosynthetic reaction centers, and SOD enzymes. Molybdenum Central role in nitrogenase enzymes catalyzing N 2 NH 3, NO 3 NH 3 Chromium, Vanadium and Nickel Small quantities, uncertain biological roles. Sugar metabolism (Cr); Ni only in plants and bacteria (role in CH 4 production) and SOD enzymes.

Biochemical Classification of Metallobiomolecules Transport and storage proteins : O 2 binding/transport: Transferrin (Fe) , Ferritin (Fe), Metallothionein (Zn) Myoglobin (Fe), Hemerythrin (Fe) Hemocyanin (Cu) Enzymes (catalysts) Hydrolases: Carbonic anhydrase (Zn), Carboxypeptidase (Zn) Oxido-Reductases: Alcohol dehydrogenase (Zn), Superoxide dismutase (Cu, Zn, Mn, Ni), Catalase, Peroxidase (Fe), Nitrogenase (Fe, Mo), Cytochrome oxidase (Fe, Cu), Hydrogenase (Fe, Ni) Isomerases: B 12 coenzymes (Co), Aconitase (Fe-S) Oxygenases: Cytochrome P 450 (Fe), Nitric Oxide Synthases (Fe) Electron carriers: Cytochromes (Fe) Electron transferases Iron-sulfur (Fe) Blue copper proteins (Cu) Non Proteins Transport Agents: Siderophores (Fe)

Porphyrins and Related Complexes in Bioinorganic Molecules • A porphyrin ring has a square planar geometry with a “pocket” in the center. • A metalloporphyrin complex can result by incorporating a metal atom into the pocket Axial sites are available for other ligands. • Structure, specificity, and reactivity are changed by differing the side chains, metal ions, and surrounding species.

Haemoglobin and Myoglobin • Oxygen transfer and storage agents in the blood and muscle tissue. • Hemoglobin transports oxygen (O 2) from the lungs/gills to tissues and muscles. • Myoglobin stores oxygen (O 2) in the muscles and tissues. Oxygen commonly transfers from the hemoglobin to the myoglobin for later use.

Haemoglobin • Made up of four globin protein subunits ( and ). • Each protein partially encloses a heme group. • Each heme group is in a porphyrin pocket. • One axial position of the iron is bound to an imidazole nitrogen from the protein. • One axial position is available/vacant or has H 2 O bound to it. • Dissolved O 2 can bind reversibly to this axial position.
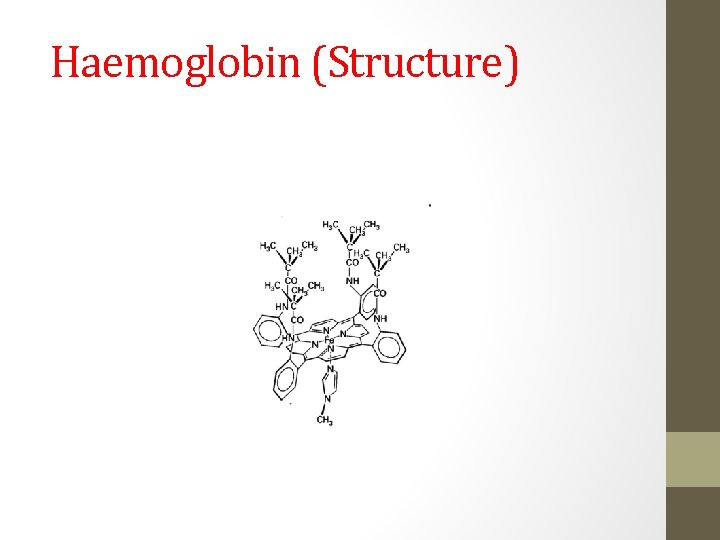
Haemoglobin (Structure)

Oxygen Addition to Haemoglobin

Cooperativity • Cooperativity: The function of hemoglobin is to bind O 2 at high oxygen pressure and carry it through the blood to needed areas (and myoglobin for storage). Hb + 4 O 2 Hb(O 2)4 + 4 Mb(O 2) + Hb • As one iron binds an oxygen molecule in Hb, the molecular shape changes to make binding of additional oxygen molecules easier. In a similar fashion, initial removal of oxygen triggers the release of the remaining oxygens.
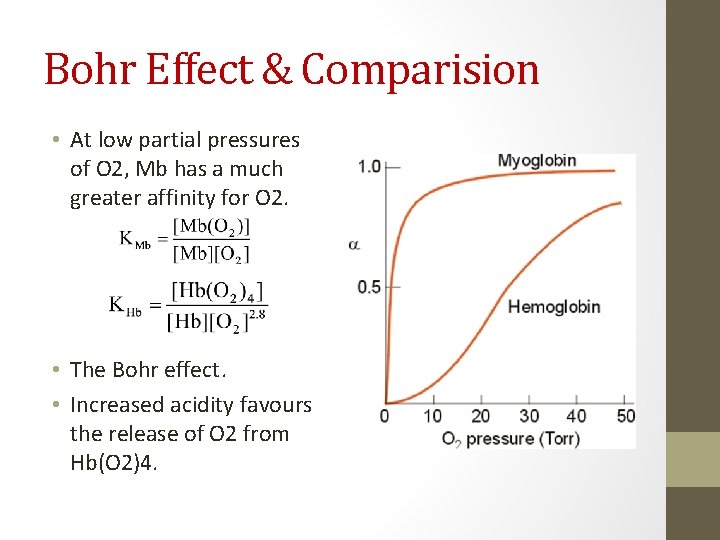
Bohr Effect & Comparision • At low partial pressures of O 2, Mb has a much greater affinity for O 2. • The Bohr effect. • Increased acidity favours the release of O 2 from Hb(O 2)4.
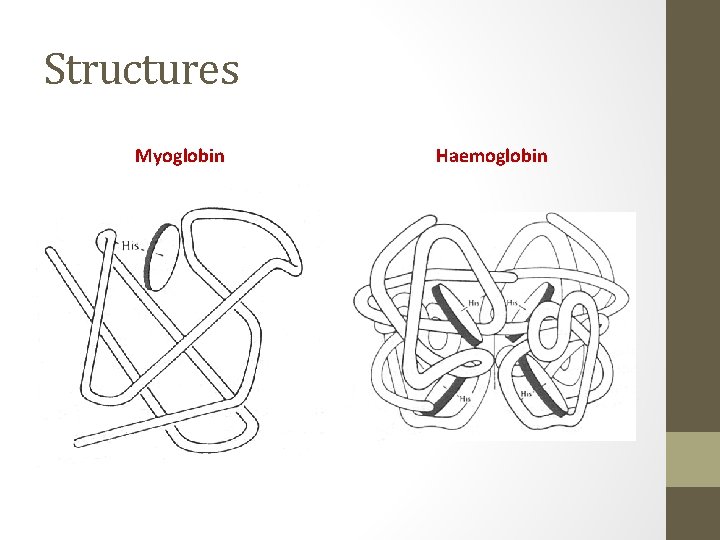
Structures Myoglobin Haemoglobin
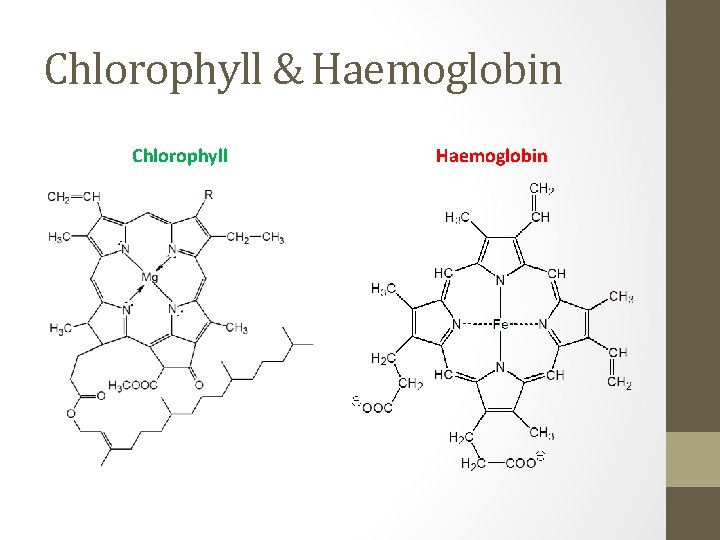
Chlorophyll & Haemoglobin Chlorophyll Haemoglobin
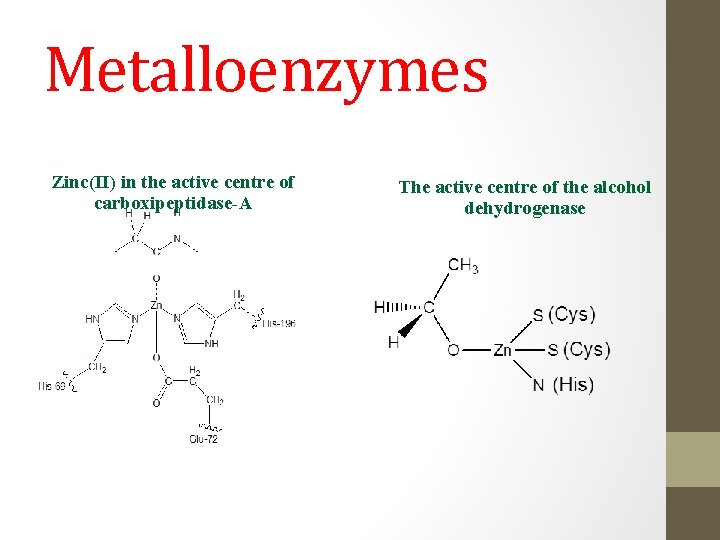
Metalloenzymes Zinc(II) in the active centre of carboxipeptidase-A The active centre of the alcohol dehydrogenase
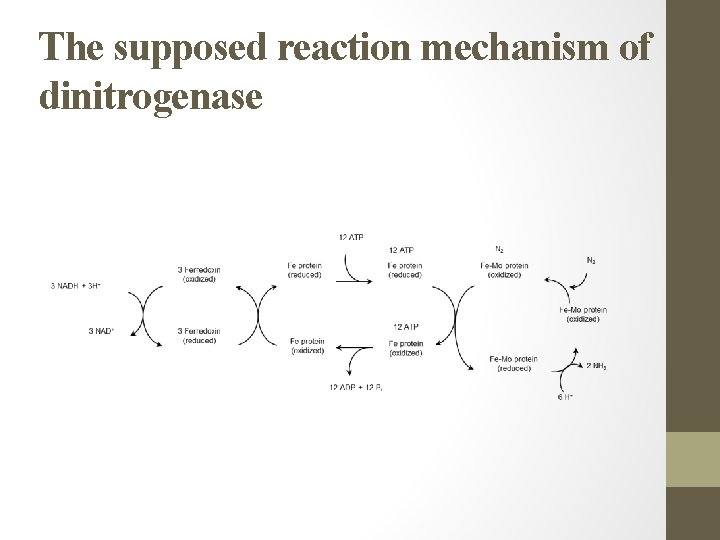
The supposed reaction mechanism of dinitrogenase
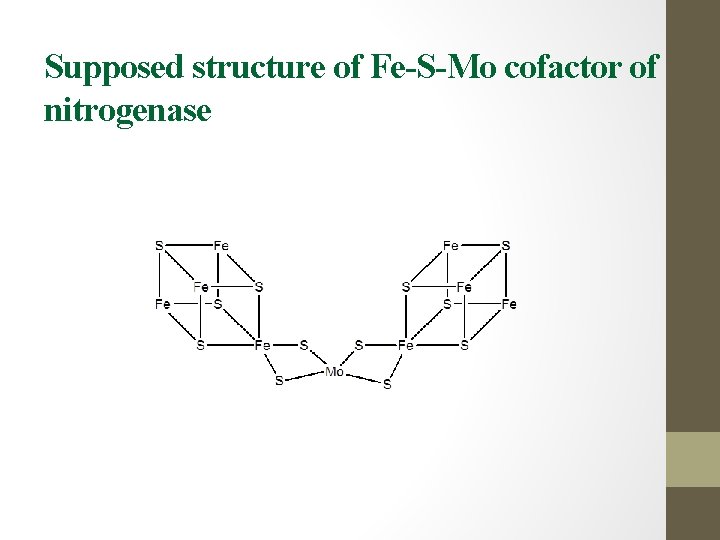
Supposed structure of Fe-S-Mo cofactor of nitrogenase
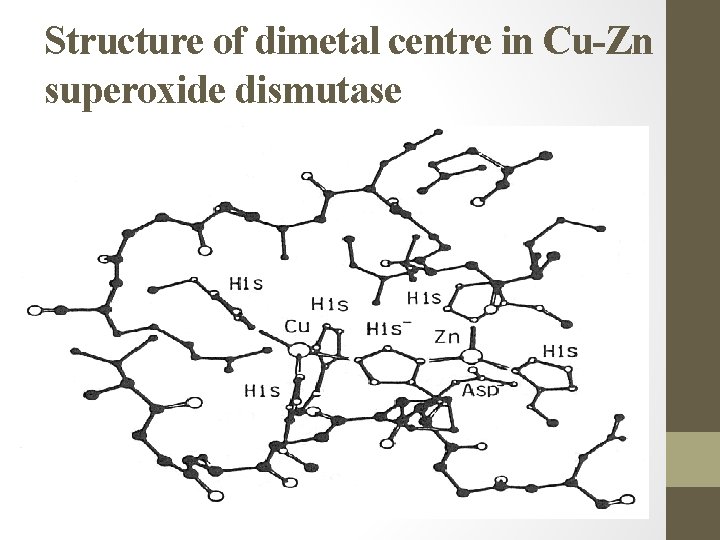
Structure of dimetal centre in Cu-Zn superoxide dismutase

! ! ! ! U O Y K N A TH
- Slides: 22