BIOGEOCHEMISTRY What is Biogeochemistry The study of the
BIOGEOCHEMISTRY

What is Biogeochemistry? • The study of the biological, geological and chemical factors that influence the movement of chemical elements through living systems across space and time. • Processes can be studies at the microbial, ecosystem and global scales • Scales are becoming increasingly integrated. • Ex: The role of microorganisms in carbon cycling and global climate change • Elements “cycle” through reservoirs often termed as source or sink • Matter of perspective and what is being measured • Ex. Plant biomass is a sink of atmospheric carbon (CO 2) via photosynthesis Burning of fossil fuels (i. e. dead plants) is a source of carbon to the atmosphere

Four main elemental cycles • Carbon (C) • Nitrogen (N) • Phosphorous (P) • Sulfur (S) • Elements do not cycle independently. • Ex: Denitrification (N 03 N 2) requires C that is easily consumed by microbes

Carbon Cycle Notice: natural processes of photosynthesis and respiration are nearly equal if not slightly in favor of storage in biota, soils, and ocean surface water Short Term Storage Significant Microbial Component Long Term Storage UNITS! Gigatons C/Year
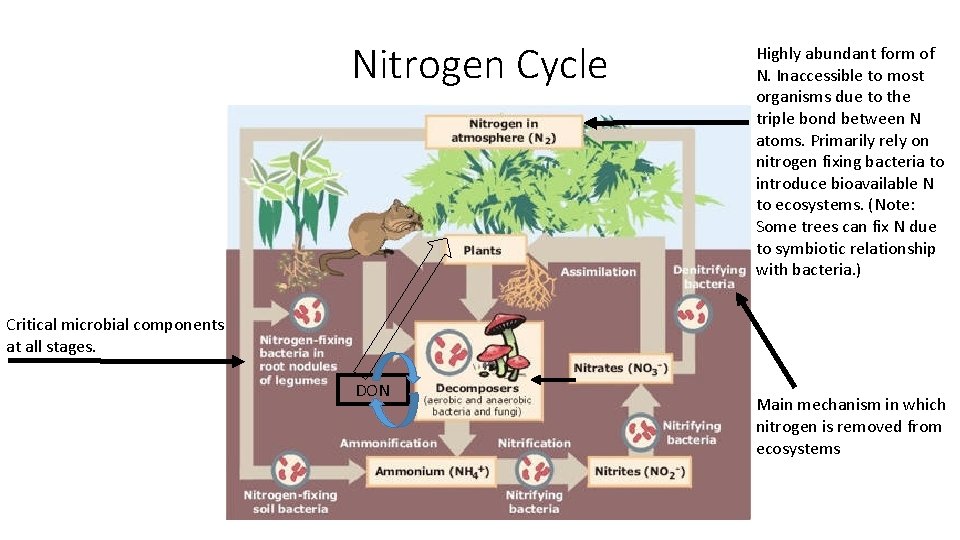
Nitrogen Cycle Highly abundant form of N. Inaccessible to most organisms due to the triple bond between N atoms. Primarily rely on nitrogen fixing bacteria to introduce bioavailable N to ecosystems. (Note: Some trees can fix N due to symbiotic relationship with bacteria. ) Critical microbial components at all stages. DON Main mechanism in which nitrogen is removed from ecosystems
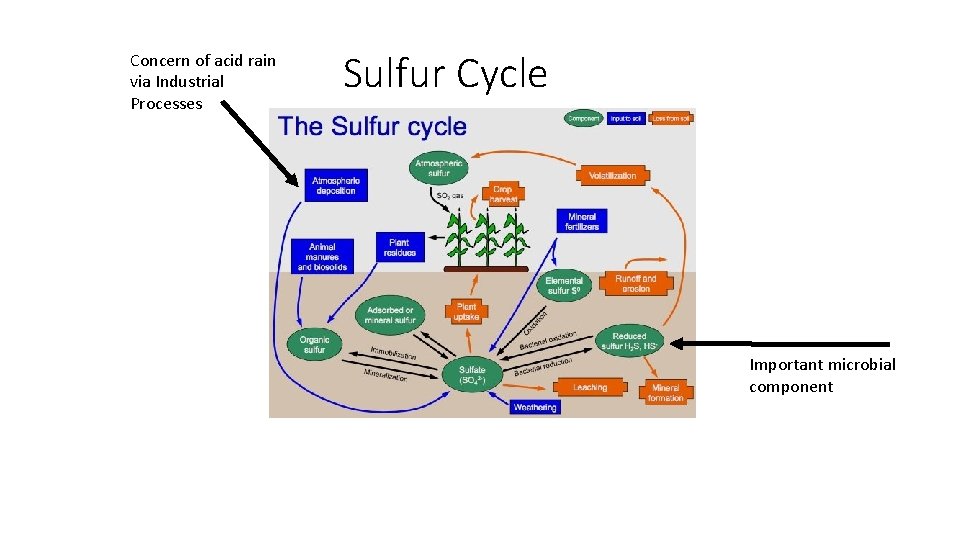
Concern of acid rain via Industrial Processes Sulfur Cycle Important microbial component
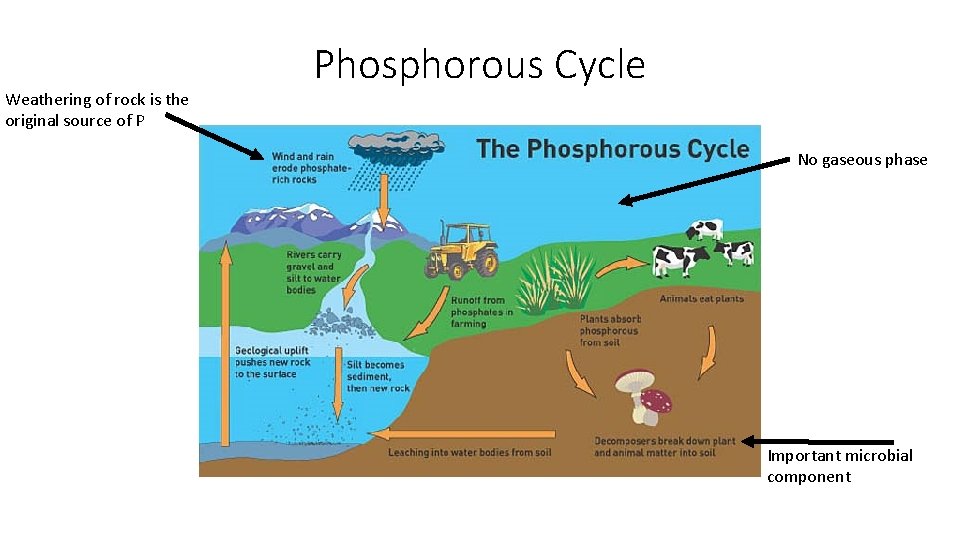
Weathering of rock is the original source of P Phosphorous Cycle No gaseous phase Important microbial component

Isotopes: a powerful technique in Biogeochemistry • Isotopes: atoms of the same element that vary in atomic mass due to different number of neutrons • Two most common in Biogeochemistry research • Carbon: 12 C, 13 C Nitrogen: 14 N, 15 N • Different reservoirs have isotopic signatures • Plants: higher 13 C: 12 C than the atmosphere • Nitrogen in the ocean: higher 15 N: 14 N signature than terrestrial bound nitrogen } • Organisms higher on the food chain have higher 15 N values • Vegetarian/vegans have lower 15 N values Research implications?

Two main isotopic techniques • Relative Abundance: measure isotopic ratios to determine if something is changing • Ex: The ratio of atmospheric 13 C: 12 C is increasing indicating more plant-based carbon is entering the atmosphere • What is the source of this new plant-based carbon? • Stable Isotopes: Raise the amount of the rarer isotope way above background levels and follow it through a system • Ex: 15 N can be added to a stream and measured downstream to determine the demand for N by microorganisms.

- Slides: 11