Biogeochemical cycling Generalized Nutrient cycling consumers producers consumers

Biogeochemical cycling
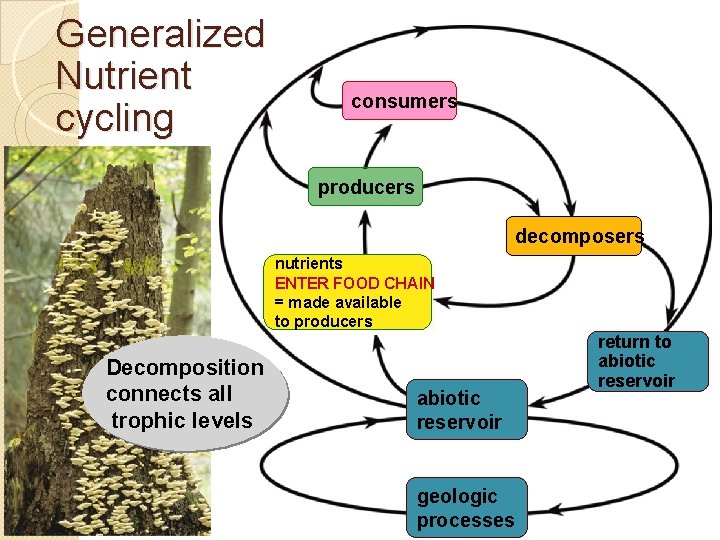
Generalized Nutrient cycling consumers producers consumers decomposers nutrients ENTER FOOD CHAIN made available = made available to producers Decomposition connects all trophic levels abiotic reservoir geologic processes return to abiotic reservoir

Carbon cycle CO 2 in atmosphere Diffusion Respiration abiotic reservoir: § CO 2 in atmosphere enter food chain: Combustion of fuels = § photosynthesis carbon fixation in Industry and home Calvin cycle Photosynthesis recycle: § return to abiotic: § respiration Plants § combustion Animals Dissolved CO 2 Bicarbonates Photosynthesis Animals Plants and algae Carbonates in sediment Deposition of dead material Fossil fuels (oil, gas, coal)

abiotic reservoir: § surface & atmospheric water enter food chain: § precipitation & plant uptake Solar energy recycle: § transpiration return to abiotic: Evaporation§ evaporation & runoff Water cycle Transpiration Water vapor Precipitation Oceans Runoff Lakes Percolation in soil Groundwater Aquifer
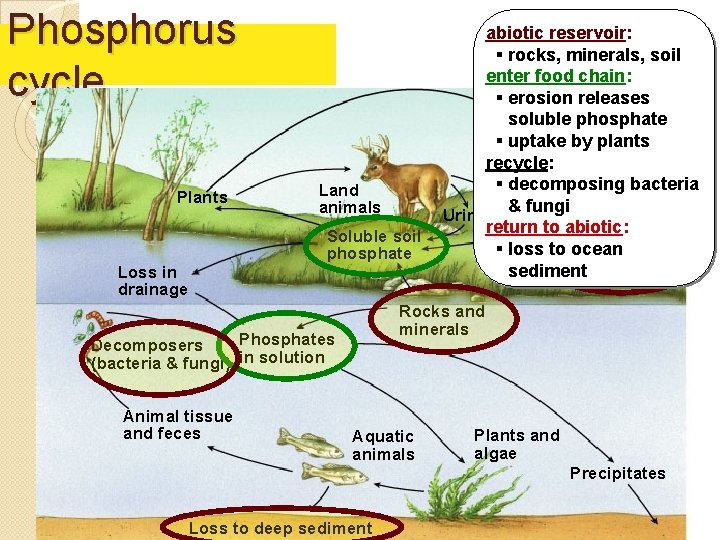
Phosphorus cycle Plants Land animals Soluble soil phosphate Loss in drainage abiotic reservoir: § rocks, minerals, soil enter food chain: § erosion releases soluble phosphate § uptake by plants recycle: § decomposing bacteria Animal tissue & fungi Urine and feces return to abiotic: § loss to. Decomposers ocean (bacteria and sediment fungi) Rocks and minerals Phosphates Decomposers (bacteria & fungi) in solution Animal tissue and feces Aquatic animals Plants and algae Precipitates Loss to deep sediment
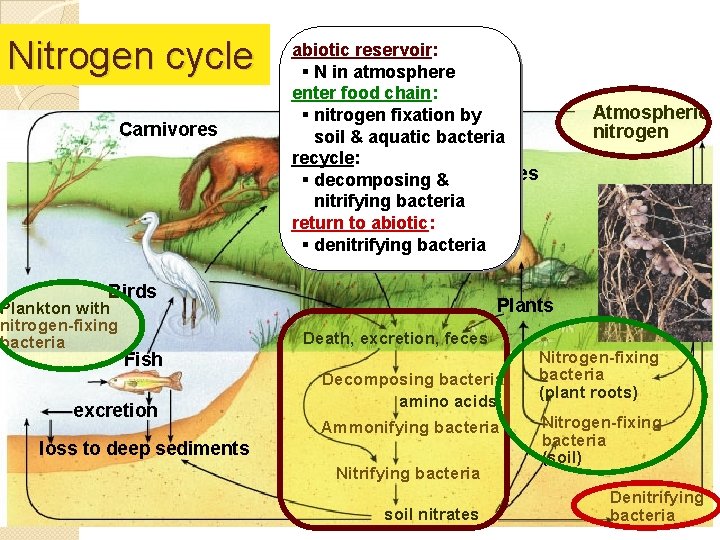
Nitrogen cycle Carnivores abiotic reservoir: § N in atmosphere enter food chain: § nitrogen fixation by soil & aquatic bacteria recycle: Herbivores § decomposing & nitrifying bacteria return to abiotic: § denitrifying bacteria Birds Plankton with nitrogen-fixing bacteria Atmospheric nitrogen Plants Death, excretion, feces Fish excretion Decomposing bacteria amino acids Ammonifying bacteria loss to deep sediments Nitrifying bacteria soil nitrates Nitrogen-fixing bacteria (plant roots) Nitrogen-fixing bacteria (soil) Denitrifying bacteria
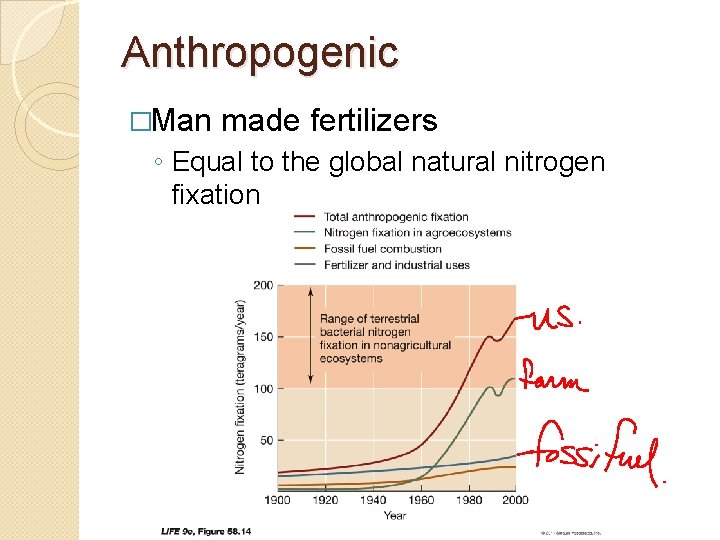
Anthropogenic �Man made fertilizers ◦ Equal to the global natural nitrogen fixation
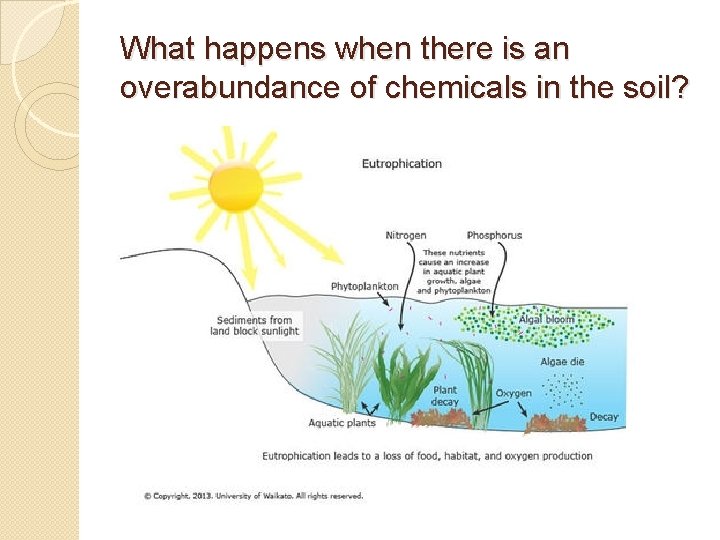
What happens when there is an overabundance of chemicals in the soil?

Eutrophication
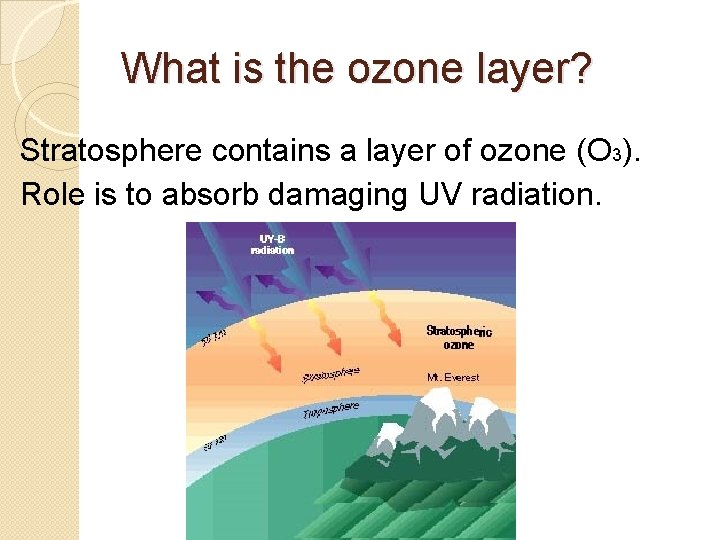
What is the ozone layer? Stratosphere contains a layer of ozone (O 3). Role is to absorb damaging UV radiation.

Cholorflourocarbon: CFC’s �Man-made chemical used in ◦ Refrigerants ◦ Car Coolants ◦ Aerosol cans
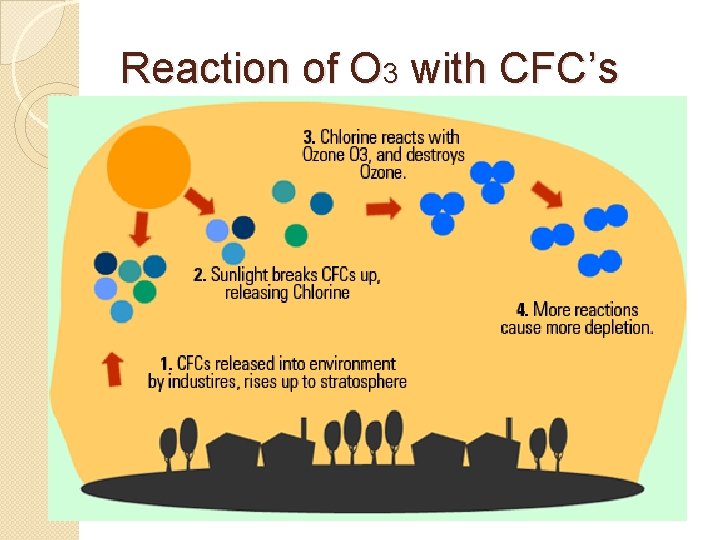
Reaction of O 3 with CFC’s
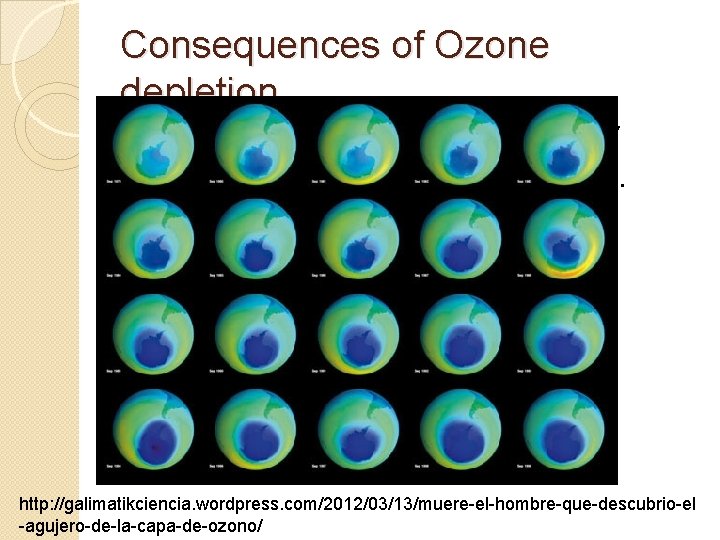
Consequences of Ozone depletion Thinning of this layer increases UV radiation to at the Earth’s surface. �Increase of skin cancer �Eye damage �Plant tissue damage http: //galimatikciencia. wordpress. com/2012/03/13/muere-el-hombre-que-descubrio-el -agujero-de-la-capa-de-ozono/

Global Warming �Greenhouse gases- a natural layer of gases that are transparent to sunlight but trap heat radiating from the Earth’s surface. �Gases: Methane, CO 2, water vapor, N 2 O and O 3 when it is in the troposhere.

Evidence of global warming

Increase of Pathogens � Colorado pine bark beetles and fungus they carry damage pine trees �Mosquitoes carry deneque fever and malaria
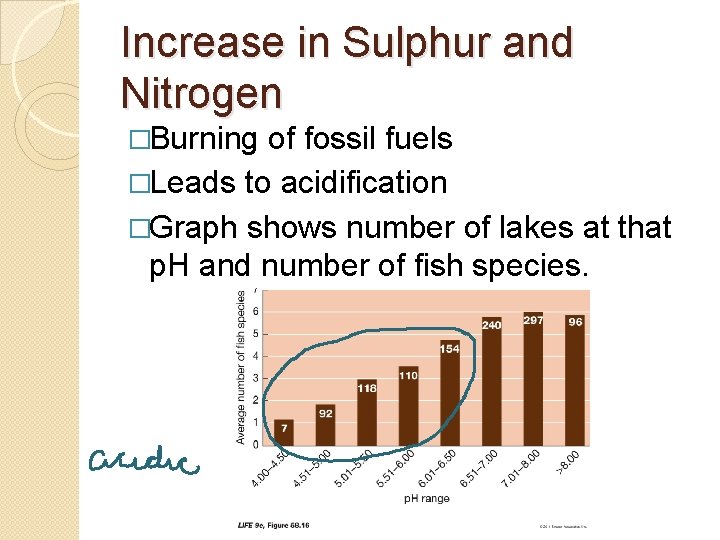
Increase in Sulphur and Nitrogen �Burning of fossil fuels �Leads to acidification �Graph shows number of lakes at that p. H and number of fish species.
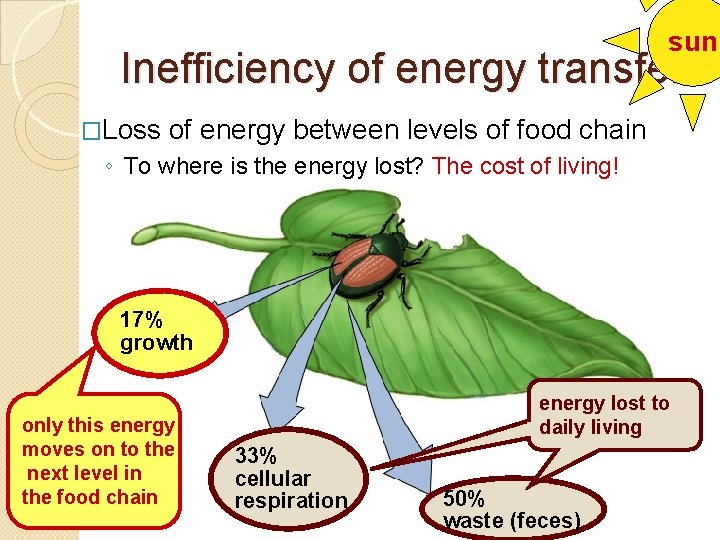
sun Inefficiency of energy transfer �Loss of energy between levels of food chain ◦ To where is the energy lost? The cost of living! 17% growth only this energy moves on to the next level in the food chain energy lost to daily living 33% cellular respiration 50% waste (feces)
- Slides: 18