Biogeochemical Cycles Water Carbon Nitrogen Phosphorus Sulfur What

Biogeochemical Cycles (Water, Carbon, Nitrogen, Phosphorus, Sulfur)

What is a biogeochemical cycle? O A biogeochemical cycle is the continual recycling of nutrients through the air, water, rock, soil, and living organisms. O All of these nutrients are essential to life. O No new molecules are created – what we have is what was here at the beginning.

The Water Cycle
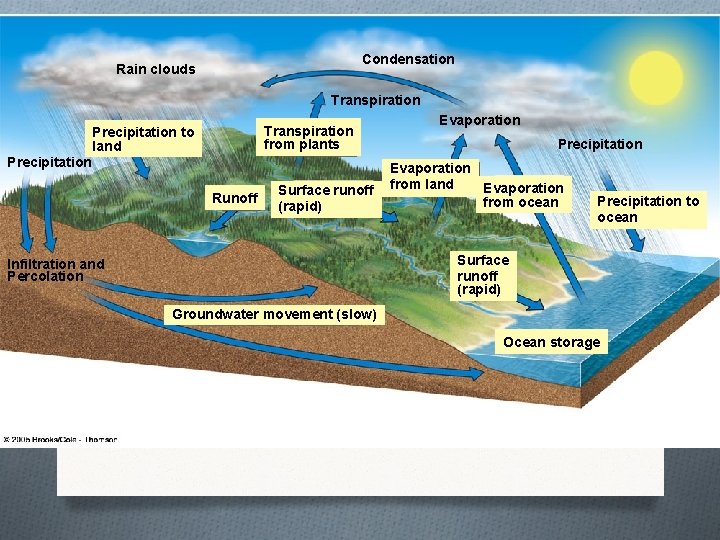
Rain clouds Figure 4 -28 Page 76 Condensation Transpiration from plants Precipitation to land Precipitation Runoff Surface runoff (rapid) Evaporation Precipitation Evaporation from land Evaporation from ocean Precipitation to ocean Surface runoff (rapid) Infiltration and Percolation Groundwater movement (slow) Ocean storage

The Water Cycle O Groundwater is water that sinks into the soil and is stored in aquifers. O An aquifer is a groundwater storage area.

Humans alter the water cycle by… O Withdrawing large quantities of freshwater for agriculture leading to groundwater depletion. O Clearing vegetation for agriculture, roads, building, and mining. O Covering land with buildings and concrete which increases runoff, and prevents water from becoming ground water. O Altering the quality of the water by the addition of nutrients from chemicals and pollutants.

The Carbon Cycle
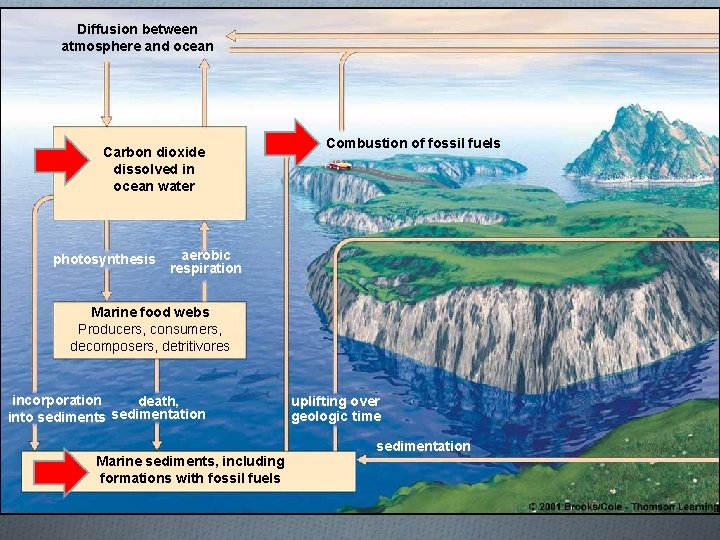
Diffusion between atmosphere and ocean Carbon dioxide dissolved in ocean water photosynthesis Combustion of fossil fuels aerobic respiration Marine food webs Producers, consumers, decomposers, detritivores incorporation death, sedimentation into sediments Marine sediments, including formations with fossil fuels uplifting over geologic time sedimentation
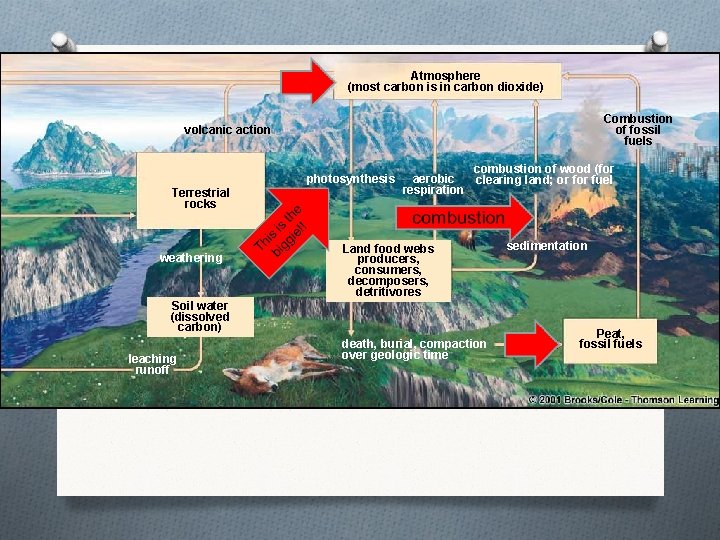
Atmosphere (most carbon is in carbon dioxide) Combustion of fossil fuels volcanic action photosynthesis Terrestrial rocks weathering combustion of wood (for aerobic clearing land; or fuel respiration Land food webs producers, consumers, decomposers, detritivores Soil water (dissolved carbon) leaching runoff death, burial, compaction over geologic time sedimentation Peat, fossil fuels

Humans alter the carbon cycle by… O Clearing trees therefore, the trees cannot remove the carbon dioxide from the air O Burn fossil fuels which release a large amount of carbon into the air

The Phosphorous Cycle
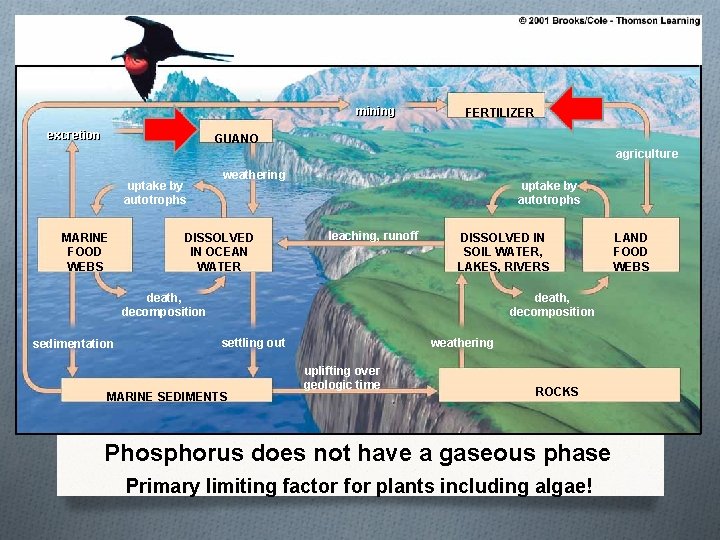
mining excretion FERTILIZER GUANO agriculture uptake by autotrophs MARINE FOOD WEBS weathering DISSOLVED IN OCEAN WATER uptake by autotrophs leaching, runoff DISSOLVED IN SOIL WATER, LAKES, RIVERS death, decomposition sedimentation death, decomposition weathering settling out MARINE SEDIMENTS uplifting over geologic time ROCKS Phosphorus does not have a gaseous phase Primary limiting factor for plants including algae! LAND FOOD WEBS

Humans alter the phosphorus cycle by… O Mining for phosphate rock to make inorganic fertilizers O Cutting down tropical trees reduces the amount of phosphorus in the tropical soil O Runoff from animal wastes, fertilizers, and sewage add phosphorus to aquatic systems

The Sulfur Cycle
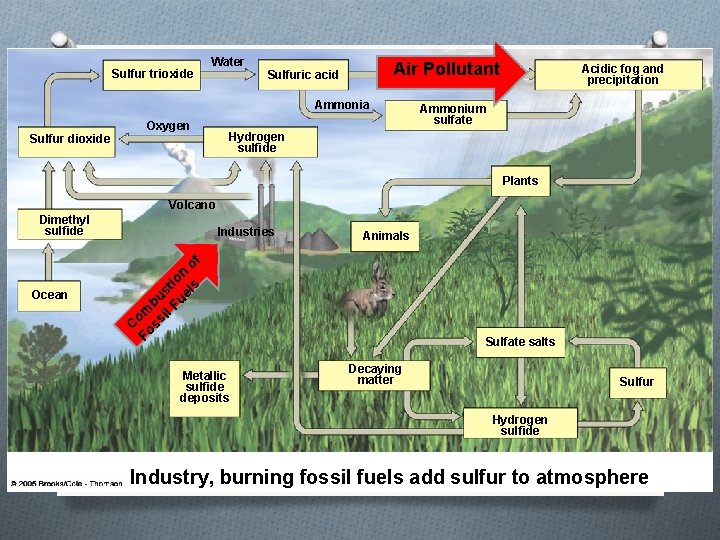
Sulfur trioxide Water Air Pollutant Sulfuric acid Ammonia Sulfur dioxide Oxygen Acidic fog and precipitation Ammonium sulfate Hydrogen sulfide Plants Volcano Dimethyl sulfide Animals C Fo om ss bu il st Fu io el n o s f Ocean Industries Metallic sulfide deposits Sulfate salts Decaying matter Sulfur Hydrogen sulfide Industry, burning fossil fuels add sulfur to atmosphere

Humans alter the sulfur cycle by… O Burning coal and oil to create electricity which releases sulfur into the air O Refining petroleum to create gasoline O Creating metals from ores

The Nitrogen Cycle
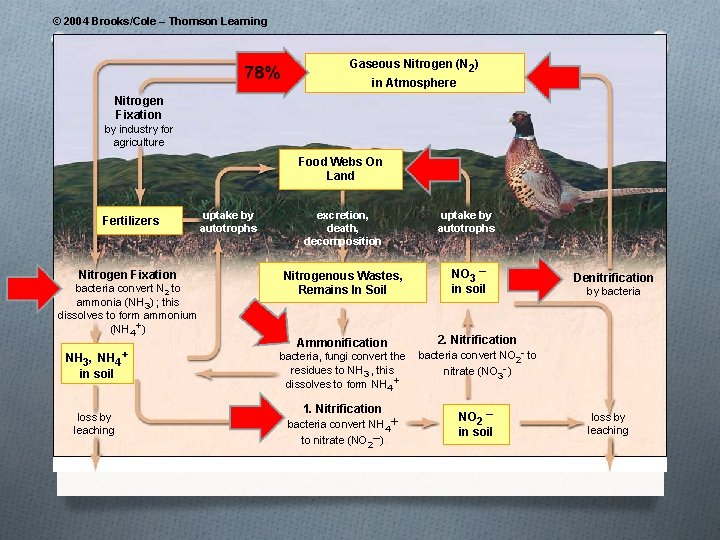
© 2004 Brooks/Cole – Thomson Learning Gaseous Nitrogen (N 2) in Atmosphere Nitrogen Fixation by industry for agriculture Food Webs On Land Fertilizers Nitrogen Fixation bacteria convert N 2 to ammonia (NH 3) ; this dissolves to form ammonium (NH 4+) NH 3, NH 4+ in soil loss by leaching uptake by autotrophs excretion, death, decomposition Nitrogenous Wastes, Remains In Soil Ammonification bacteria, fungi convert the residues to NH 3 , this dissolves to form NH 4+ 1. Nitrification bacteria convert NH 4+ to nitrate (NO 2–) uptake by autotrophs NO 3 – in soil Denitrification by bacteria 2. Nitrification bacteria convert NO 2 - to nitrate (NO 3 -) NO 2 – in soil loss by leaching

The Nitrogen Cycle O Nitrogen in the air is in the form of N 2 which is unusable to plants and animals. The N 2 needs to be converted to a useable form – this is done by 1) lightening and 2) bacteria in the water and soil. O Nitrogen fixation – bacteria in the soil convert N 2 to O O NH 3 so that plants can use it Nitrification – bacteria take the NH 3 and turn it into nitrite (NO 2 - toxic) and nitrate (NO 3) ions Assimilation – Plants absorb the ammonia and nitrate; animals get nitrogen by eating plants Ammonification – decomposer bacteria take animal and plant wastes and turn it into ammonia Denitrification – ammonia is converted into nitrites and nitrates and then to N 2 where it is released into the air

The Nitrogen Cycle O Plants need nitrogen in the form of nitrates to make nucleic acids (DNA) and amino acids.

Humans alter the nitrogen cycle by… O Burning fuel releases nitric oxide (NO) which O O converts to (HNO 3) in the atmosphere and falls back to the earth as acid rain Using inorganic fertilizers which releases N 2 O (nitrous oxide) into the atmosphere which depletes the ozone layer Destroy forests, grasslands, and wetlands which releases the N in the soil and the plants Agricultural runoff and sewage goes into waterways and the nitrates disrupt the aquatic ecosystems When working with crops (irrigating, harvest, burn to create room for) nitrogen is taken from the topsoil.
- Slides: 21