Biogeochemical Cycles QQ Introducing the Biogeochemical Cycles Cycle

Biogeochemical Cycles QQ

Introducing the Biogeochemical Cycles Cycle of chemical through both biologic and geologic things. (it is necessary to understand & use these if you want to design or understand any ecosystem and it’s varying resources…) Water cycle Carbon cycle Nitrogen cycle. QQ#1:

Water is stored in earth and cycles throughout different stages. 97% of Earth’s water is stored in the ocean

2% of the Earth’s water is stored as ice

Most of the rest is stored as groundwater

Also, in Rivers & lakes As well as water vapor in the atmosphere

Also, it is stored inside Plant tissue QQ#2: How does this relate to our previous learning goal

The water cycles through Clouds, rain, snow… QQ#3: What do you know about the water cycle?

• water never leaves the Earth • constantly cycles through atmosphere, ocean, and • • • land this process is known as the water cycle driven by energy from the sun - radiation crucial to the existence of life
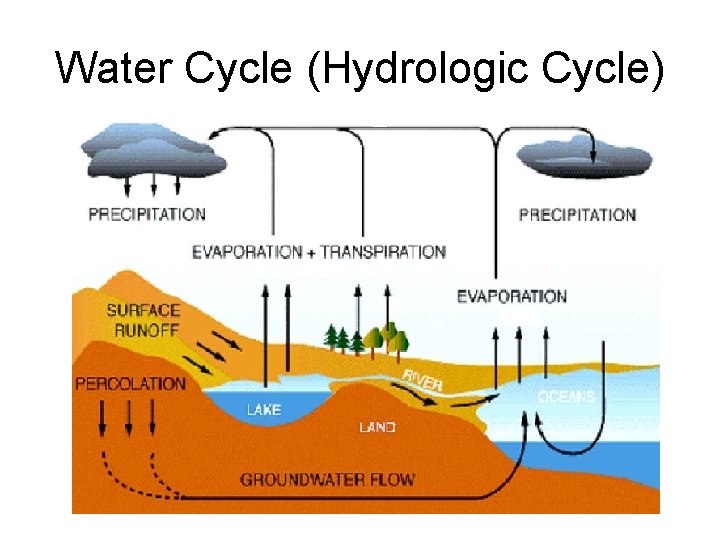
Water Cycle (Hydrologic Cycle)

Step 1: Evaporation/Transpiration • Through the process of radiation, Solar energy warms cool, heavier water and it becomes less dense and rises. • The process of evaporation from plants is called transpiration.

Step 2: Condensation • Condensation: Water vapor rises begins to cool and become clouds until the water becomes liquid again • This process is driven in part by convection currents QQ#4: where have you heard of convection currents before?

Step 3: Precipitation • Precipitation: Water becomes more dense as it collects in the clouds and it will fall
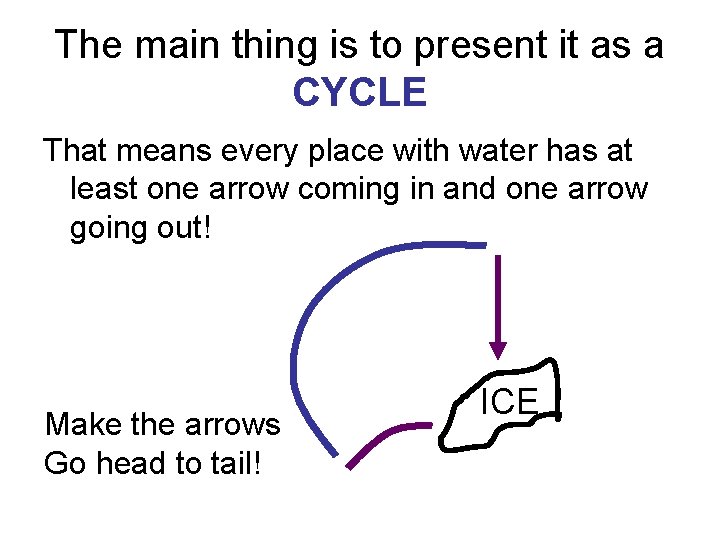
The main thing is to present it as a CYCLE That means every place with water has at least one arrow coming in and one arrow going out! Make the arrows Go head to tail! ICE

The Carbon Cycle: Jus t like H 2 O can cycle around the earth/atmosphere so can the carbon atom QQ#5: Where do you think Carbon is stored on earth? Carbon can be found in all living or once living things, as well as many abiotic factors.

Most of the carbon on earth is locked up in the crust in limestone rocks: Ca. CO 3

Some of the rest is in fossil fuels: coal, crude oil and natural gas. Coal = carbon Methane = CH 4

Some is in the atmosphere as CO 2:

A lot is dissolved in seawater as carbonate (CO 32 -), bicarbonate (HCO 3 -), carbonic acid (H 2 CO 3) and carbon dioxide (CO 2):

And of course some is in living & dead biological tissue (Biomass):

Two processes help recycle carbon around earth: PHOTOSYNTHESIS (plants and Algae): 6 CO 2 + 6 H 2 O →(sunlight) → C 6 H 12 O 6 + 6 O 2 RESPIRATION (all life) C 6 H 12 O 6 + 6 O 2 → 6 CO 2 + 6 H 2 O (+ energy!) C 6 H 12 O 6 = sugar O 2 = oxygen CO 2 = carbon dioxide H 2 O = water
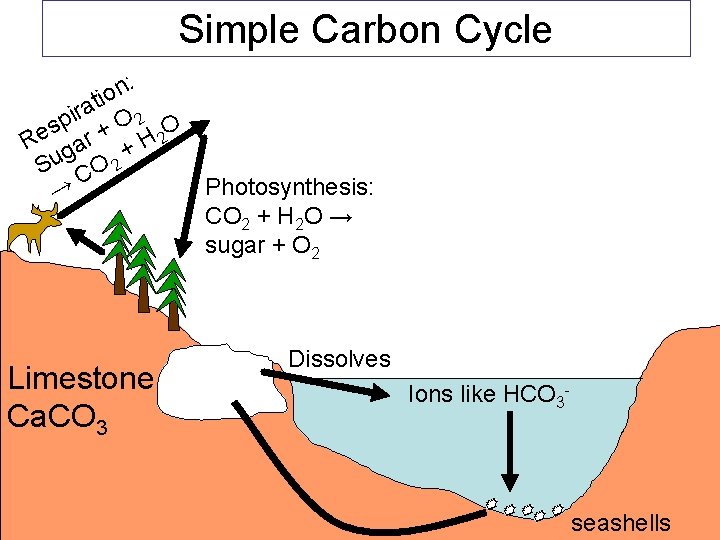
Simple Carbon Cycle : n o ti a r pi + O 2 O s Re gar + H 2 Su CO 2 Photosynthesis: → CO 2 + H 2 O → sugar + O 2 Limestone Ca. CO 3 Dissolves Ions like HCO 3 - seashells
- Slides: 22