BIOGEOCHEMICAL CYCLES Definition Biogeochemical cycle is defined as

BIOGEOCHEMICAL CYCLES

Definition : Biogeochemical cycle is defined as a cyclic pathway which brings about the circulation of chemical elements from the environment to the organisms & back to the environment. BIO + GEO + CHEMICAL This pathway involves living organisms (bio) A series of chemical reactions (chemical) in abiotic environment (geo). • Cycling elements: Macronutrients : Required in large amount. C, H, O, N, P, S, K, Ca, Fe, Mg. Micronutrients : Required in small amount but still necessary. B, Cu, Mo.
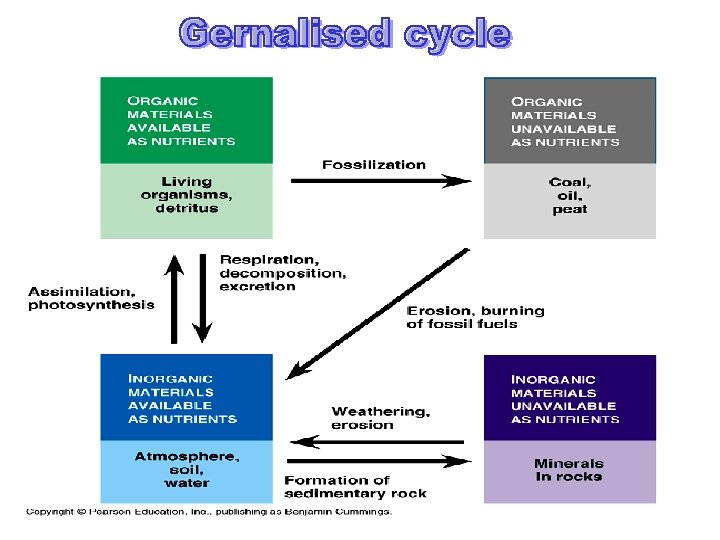

Pools & Fluxes • All bioelements reside in compartments or defined spaces in nature. • A compartment contains a certain quantity, or pool, of bioelements. • The reservoir portion is called the unavailable pool and cycling portion the available pool. • E. g. . The 3 natural pools of Carbon: CO 2 stored in atmosphere-: 0. 03% C stored in oceans-: soluble elements like HCO 3, CO 2, CO 32 -. C stored in continental biosphere-C in fossil fuels. • The rate of movement of different elements between pools through the biosphere is called the flux rate. It is measured as quantity of nutrients passing from one pools to another per unit of time. • Flux rate depends upon the physical & chemical properties of each element &the use to which the living organisms put them. Thus flux refers to the flow of materials. • The average length of time a bioelement remains in a compartment is called the mean residence time

Concept of mass-balance budgets: Scientist construct budgets to study how elements are gained and lost in ecosystems. • Some ecosystems are in steady state: this occurs when fluxes are balanced. Therefore: inputs = outputs • Inputs sometimes exceed outputs, and elements accumulate in compartments of ecosystems. Therefore: Inputs > outputs = storage (sink) (e. g. , community development following a disturbance; e. g. volcano) • Outputs sometimes exceed inputs of nutrients in communities or ecosystems Inputs < outputs = loss (source) (e. g. , fire, leaching by acid deposition)
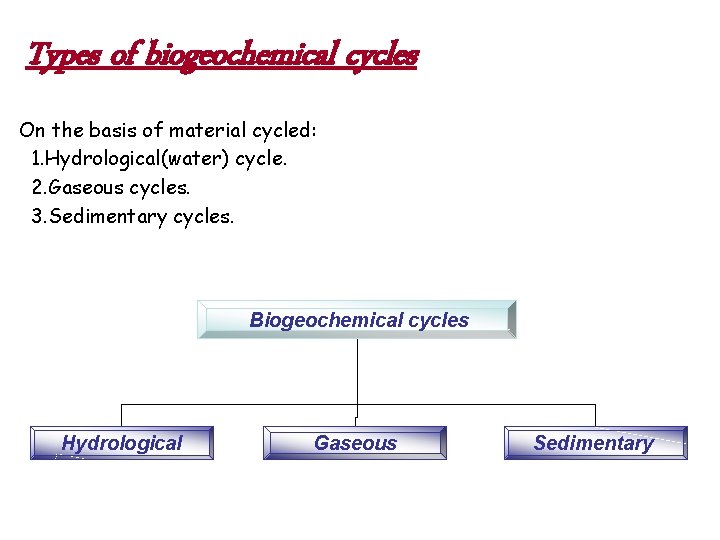
Types of biogeochemical cycles On the basis of material cycled: 1. Hydrological(water) cycle. 2. Gaseous cycles. 3. Sedimentary cycles. Biogeochemical cycles Hydrological Gaseous – Sedimentary

Sedimentary cycles The mineral or the sedimentary cycle is the one in which the reservoir is in the lithosphere i. e. . earth’s crust. It consists of 2 phases: (i) the salt solution phase (ii) rock phase. In sedimentary cycle the element concerned is continually lost from the biological system thorough erosion & ultimately deposited in the oceans. Hence these are not perfect cycles.

Phosphorous cycle • P is a component of nucleic acid, ADP, ATP, NADP, phospholipids. • Occurs in soil as rock phosphate, apatite, Ca phosphate, fluorapatite [Ca 10 Fe 2(PO 4)6]etc. • P occurs in soil in 5 forms: P 1(stable organic) P 2(labile organic) P 3(labile inorganic) P 4(soluble) P 5(mineral form) P 3 & P 4 are in equilibrium. Entry of P in green plants is through labile inorganic pool.

Phosphorus cycle continued …. CYCLE: • P is a earth bound element, lacks significant atmospheric component. • Global resource: geological deposits of 2000 billion tons (but rate of weathering is slow). • Phosphates originate through mechanical or chemical weathering of rocks & from human excreta (2 mg of phosphate/person/day). • Sea is the richest source of available phosphate. Phosphate rich rock tend to be of marine origin. • Main rout back to land: through sea birds feeding on phosphate rich fish & defecating on land. Their dung forms guano (used as manure). • Natural transfer of P from ocean to land is very small less than 0. 03 mmt/year for sea spray & 0. 01 mmt/year for guano.

Phosphorus cycle continued…. . • Plants and animals concentrate P in their bodies through biological process as formation of teeth & bones. • On death it can be recycled into living system OR become immobilized in the soil. In soil it remain for 1000’s of years, eventually going into sea through erosion. • Insolubility is the fate of most free P hence it is main limiting nutrient in many ecosystems. • Number of plants have symbiotic association with fungi to secure additional P. This is VAM.


Sulphur cycle links air, water & land. S is essential constituent of certain amino acids. It occurs -in soil & rocks as sulphides (Fe. S, Zn. S etc. ) & crystalline sulphates. - in atmosphere in form of SO 2 & H 2 S gas. CYCLE: S in form of SO 2 is formed during combustion of fossil fuel or decomposition. H 2 S gas is released from H 2 O logged soil, continental shelf lakes & springs. Organic & inorganic S & SO 2 are formed through oxidation of H 2 S in atmosphere. Few organisms require S in organic form as amino acid & cystine.

Sulphur cycle continued… Inorganic sulphate major source of biologically significant sulphur. Biologically incorporated S is produced in the soil from aerobic breakdown of proteins by bacteria & fungi 2 H 2 S + O 2 Baggiatoa spp 2 S + 2 H 2 O + 3 O 2 Thiobacillus(Thio-oxidation) 2 H 2 SO 4 Green & purple bacteria use H of H 2 S as the O 2 acceptor reducing CO 2. Green bacteria oxidize sulphide to elemental sulphur. Purple bacteria can carry oxidation to sulphate stage. Under anaerobic condition sulphate is reduced to elemental sulphur or to sulphides (H 2 S) by heterotrophic bacteria such as Desulfavibrio. Sulphate (SO 42 -) is soluble in H 2 O , hence it is a source of elemental sulphur.

Sulphur cycle continued S returns back to the environment through the decay of dead organic remains. Sedimentary aspects of S cycling involves precipitation of S in presence of iron under anaerobic condition sulphides of Fe, Cu, Zn, Cd, Co are insoluble in water. Hence S is bound to limit the amount of these elements. Thus, cycle afford excellent e. g. of interaction & biochemical regulation between different mineral cycles.


Nitrogen cycle N most important for plant growth. It is required for the synthesis of amino acids, proteins, enzymes, chlorophyll, nucleic acid etc. Atmospheric N (79% ) is not directly available to the organisms with exception of some prokaryotes like BGA, N fixing bacteria. CYCLE: N fixation- Conversion of free N of atmosphere to the biologically acceptable form or nitrogenous compounds. It is of 2 types: a) Physicochemical or non biological. b) Biological N fixation.

• Physicochemical N fixation- Atmospheric N combines with oxygen during lightening or electrical discharge in the cloud & produce different nitrogen oxides: N 2 + O 2 2 NO 2 Nitrogen oxides dissolve in rain water & on earth they react with mineral compounds forming nitrates &nitrogenous compounds. 4 NO 2 + 2 H 2 O + O 2 4 HNO 3 2 HNO 3 + Ca. O Ca(NO 3) 2 + H 2 O • Biological N fixation-carried by certain prokaryotes. - BGA fix N in oceans, lakes, soil. - symbiotic BGA: species of Nostoc, Anabaena, found in thalli of Anthoceros, Salvenia, Azolla, coralloid roots of Cycas. ( Mutualistic relation) - Free living N fixing bac. : Azotobactor, Clostridium, Rhodospirillium. - Fungus (actinomycetous): Frankia found in root of Alnus, Casuarina.
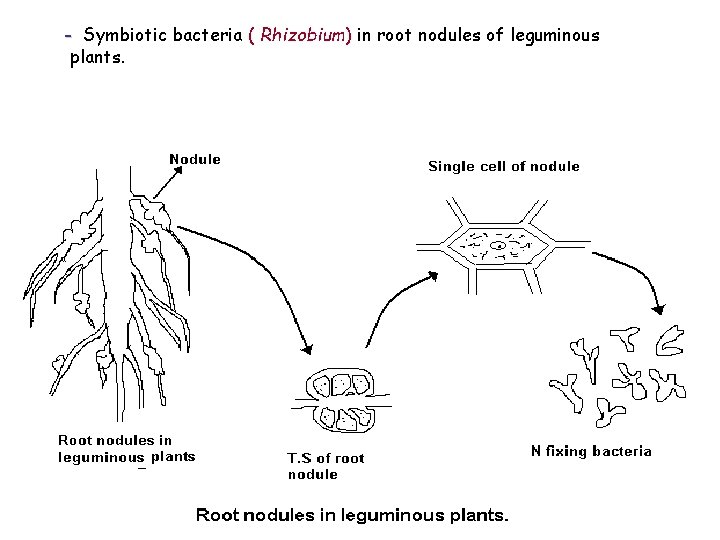
- Symbiotic bacteria ( Rhizobium) in root nodules of leguminous plants.

Nitrogen cycle continued. . • N assimilation- conversion of inorganic nitrogen (nitrates, nitrites, ammonia) to nitrogenous organic compound by green plants. NITRATES AMMONIA +organic acids AMINOACIDS used in PLANT PROTEIN SYNTHESIS ( ENZYMES CHLOROPHYLL NUCLEIC ACID) TRANSFERRED TO ANIMALS • Ammonification - Release of ammonia. Dead organic remains are acted upon by microorganisms actinomycetes & bacilli. (Bacillus vulgaris, B. ramosus )

Nitrogen cycle continued…. Nitrification - Formation of nitrates Nitrosomonas, Nitrococcus, Nitrospira, Nitrosogloea bacteria in oceans &soil convert ammonia into nitrites. 2 NH 4+ + 2 O 2 2 NO 2 - + 2 H 2 O + Energy Nitrites are converted into nitrates by several microbes like Penicillium sps. Nitrobacter, Nitrocystis (marine autotroph). 2 NO 2 - + O 2 NO 3 - + Energy Some nitrates are formed by weathering of nitrate containing rocks. Denitrification Conversion of ammonia & free nitrites into free nitrogen. Includes those dissimilatory reductive reactions which result in the production of any or all of the following gases: NO, N 2. 2 NO 3 - 2 NO 2 - 2 NO N 2 Denitrifying bac. : Pseudomonas, Thiobacillus denitrificans. The N 2 O (nitrous oxide) released, diffuses from troposphere to stratosphere where it changes to NO

NO reacts with O 3 to form NO 2 - + O 2. NO 2 - formed changes to NO. The nitrogen oxides are slowly converted to HNO 3 which returns to earth. Thus , increased use of combined nitrogen inputs from biological or industrial N fixation will increase the rate of denitrification thus may cause O 3 depletion. CYCLE Source of N for plants & animals- Biologically fixed N -Inorganic N fixed by lightening -N released from dead organic matter. Plants utilize &convert nitrates & ammonia into amino acid. Consumers convert them to different type of proteins. Animals release N by excretion. Breakdown of dead plants & animals by bacteria &fungi release ammonia. Ammonia utilized by plants or find its way to atmosphere. Nitrates & nitrites carried by river to lakes, seas. The denitrifying bacteria use them as nutrient &convert them in molecular N.


Carbon cycle “Without CO 2 earth would be as cold as moon” moon Carbon one of the primary element forming human tissues. Necessary to plants , basis of human food. Forms of carbon. CO 2 –free state in atmosphere=0. 03 % CH 4 –in atmosphere 0. 0002% Organic carbon compounds Dissolved state in oceans as bicarbonates etc. CO 2 enters living system through photosynthesis by green plants & phytoplankton in presence of sunlight & chlorophyll. Carbon is taken from the atmosphere at the surface of the oceans near the poles, where the water becomes cooler and is able to dissolve more carbon dioxide.

Carbon cycle continued… Net gain of C in ecosystem by- forest plantation, accumulation of humus and litter in forest, in grasslands & swamps in boreal zones, peat accumulation in peat lands. C is released to atmosphere - as CO 2 in respiration by plants, animals, - by bacteria & fungi attack on dead remains. - burning of forest (jhum cultivation), fossil fuels. - volcanic eruptions. Source of C - fossil fuel, deforestation, oxidation of humus. organic C incorporate in earth’s crust as coal, gas, petroleum, limestone, coral reef. Forests have vast reservoir of fixed but readily oxidisable C in form of wood, humus. Most C involved in cycle is in ocean.

Carbon cycle continued… Ocean acts to buffer or keep constant CO 2 concentration. Excess of CO 2 in atmosphere can dissolve in ocean as bicarbonates or carbonate ions. The oceans can also release CO 2 to the atmosphere. Overview of fluxes of CO 2 per year (in billion tons of CO 2) Source: - (i) Emission by use of fossil fuel- 20 (ii) Emission by deforestation & changes in land use- 5. 5 Sink: - (i) Uptake in oceans -5. 5 (ii) Uptake by CO 2 fertilization -7. 3 Disturbances in carbon cycle: Rate of release of CO 2 in atmosphere is increased up to about 50% of the expected magnitude. It has been estimated that its effect will bring about a 30 C rise in surface temperature.



Difference between gaseous & sedimentary cycle Gaseous cycles 1. Reservoir –hydrosphere or atmosphere 2. Perfect & complete as amount of nutrient in any one phase tend to remain fairly constant. Sedimentary cycle 1. Reservoir –lithosphere 2. Imperfect as some of the element may get stuck in a certain phase of the cycle.

References • • • Odum, E. P. – Fundamental of Ecology Kormody, E. J. – Concepts of Ecology Sharma, P. D. – Ecology and Environment Sharma, B. K. – Environmental Chemistry www. google. com www. ecologicalsocieties. com

- Slides: 30