Biogeochemical Cycles Carbon Nitrogen and Phosphorus Carbon n

Biogeochemical Cycles Carbon, Nitrogen, and Phosphorus

Carbon n Essential part of fats, proteins, and carbohydrates
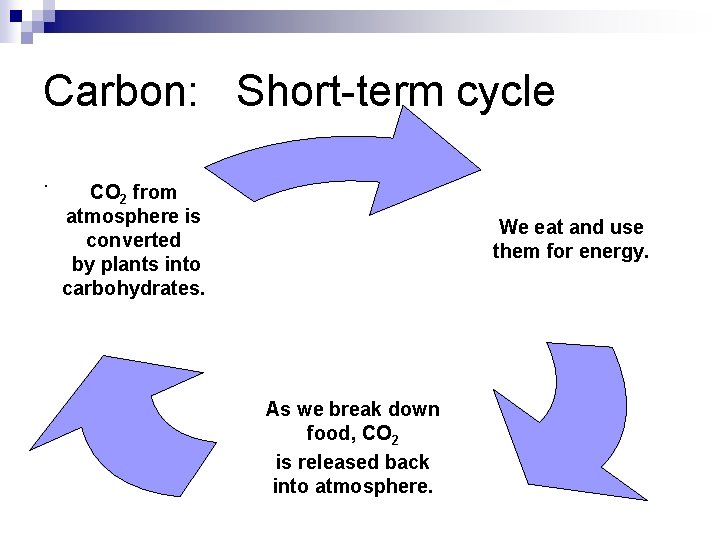
Carbon: Short-term cycle. CO 2 from atmosphere is converted by plants into carbohydrates. We eat and use them for energy. As we break down food, CO 2 is released back into atmosphere.

Carbon: Long-Term Cycle Carbonates Carbon Sinks Fossil Fuels Make up bones and shells Deposits of Carbonate form limestone—the largest reservoir of carbon. Carbohydrates stored in living material are released after it dies—fossil fuels CO 2 in air dissolves in ocean Coal, Oil, Natural Gas

Carbon: Human Affects Burning fossil fuels releases the stored carbon = CO 2 n Factories, vehicles, and power plants burn fossil fuels. n Burning any organic (living or once living) material releases its carbon n Contribute to greenhouse gases n

Nitrogen n Needed to build proteins (DNA, new cells, tissue, chlorophyll, organs, etc) n Makes (N 2) n Most up 78% of atmospheric gases organisms CAN NOT use the nitrogen in the air

Nitrogen-Fixing Bacteria n Are the only creatures to change N 2 into other compounds: ¨ NH 4 (ammonium) which is changed by other bacteria into n ¨ All NO 2, NO 3 (nitrates) these compounds are absorbed by plant roots or flow with water

Nitrogen-Fixing Bacteria n Most live on root nodules of legumes (peanuts, peas, beans, etc)

Lightning “fixes” Nitrogen too n Lightning is plasma, an ionized material. n We see electrons being pulled from gases in the atmosphere—causing a light. n This electrifying experience separates N 2, allowing it to bond with other atoms.

Nitrogen being decomposed n Nitrates are used and stored in living things—organic n Organic waste (urine, feces, carcass, leaves, etc) is decomposed by bacteria, releasing some N 2 back to the atmosphere and other nitrates to the soil and water.

Uses of Nitrogen n Fertilizer – stimulates chlorophyll (green) n Preserve freshness, incandescent lights, liquid explosives, production of electronic parts, filling tires, manufacturing stainless steel

Phosphorus Cycle n n n Essential in forming bones, teeth, DNA, and other molecules Are stored in rock as salts that erode away or are mined Not common in the atmosphere Plants absorb through roots; animals consume them Excreted in wastes and upon death

Uses of Phosphorus n Fertilizer—stimulates rapid growth n Toothpaste, detergent, explosives, nerve gas, and fireworks

Human affects on N and P n Overusing these elements and releasing them into waterways and atmosphere n N and P in fertilizers and detergents runoff into waterways and are eaten by algae. Too many algae use all of the oxygen in water, killing or displacing other living things

Gulf of Mexico Dead Zone

Human affects on N and P n Burning organic materials releases Nitrogen into the air ¨ This creates too much atmospheric nitrogen ¨ Precipitation is very acidic – acid rain, snow, etc. n Too much ammonia (product of N) is toxic to animals and plants

Build Compost Bin 2, 2 liter plastic bottles Organic material Air vents

Types of Materials: Biodegradable n n n Breaks down naturally Organic material (living or once living) Leaves, branches, grass Animal waste and remains Materials made from organic materials Non biodegradable n n n Does not break down naturally Inorganic materials (never living) Plastics Metals Ceramics; glass Silicon-based items (technology)

To degrade you need. . . Microbes, bacteria, or fungus to decompose (eat) the materials n The 3 things life needs to survive: n Liquid water 2. Right temperature 3. Energy (food source) 4. NOT OXYGEN 1.

Old Atmosphere n 4, 000 million years ago ¨ Carbon Dioxide, Ammonia, Water Vapor, and Methane n 3, 000 million years ago ¨ Nitrogen and Carbon Dioxide ¨ Water vapor condensed to liquid water ¨ Anaerobic life == does NOT need oxygen ¨ It produces oxygen as waste

Old Atmosphere n 2, 000 million years ago ¨ Oxygen-rich atmosphere formed ¨ Poisoned some anaerobic life forms ¨ Organisms with aerobic respiration appeared

Oxygen Isotopes Isotope had different number of neutrons than normal.

016, O 17, O 18 3 forms of Oxygen normally has 8 protons, 8 neutrons, and 8 electrons. n The Mass number (16) = protons + neutrons n Sometimes there are more or fewer neutrons called isotopes n

016, O 17, O 18 O 16 has 8 neutrons (8+8=16) normal n O 17 has 9 neutrons (8+9=17) normal n O 18 has 10 neutrons (8+10=18) normal n Which is heavier? O 18 n So it evaporates last of the three n
- Slides: 24