BIOGEOCHEMICAL CYCLES AP Environmental Science Biogeochemical Cycles Flow

BIOGEOCHEMICAL CYCLES AP Environmental Science

Biogeochemical Cycles • Flow of atoms, ions and molecules as they continuously recycle between the nonliving and living parts of an ecosystem. • Called NUTRIENT CYCLES

Matter Cycling in Ecosystems • Biogeochemical (nutrient) cycles – Hydrologic cycle (H 2 O) – Atmospheric cycles (C, N) – Sedimentary cycles (S, P)

Sampling Methods • To determine how quickly nutrients recycle • Radioactive tracers C, H, O, N, P, S – FOLLOW THROUGH BIOTIC AND ABIOTIC • Shows how quickly the element goes through each phase of cycle and how much of element is present at each phase.
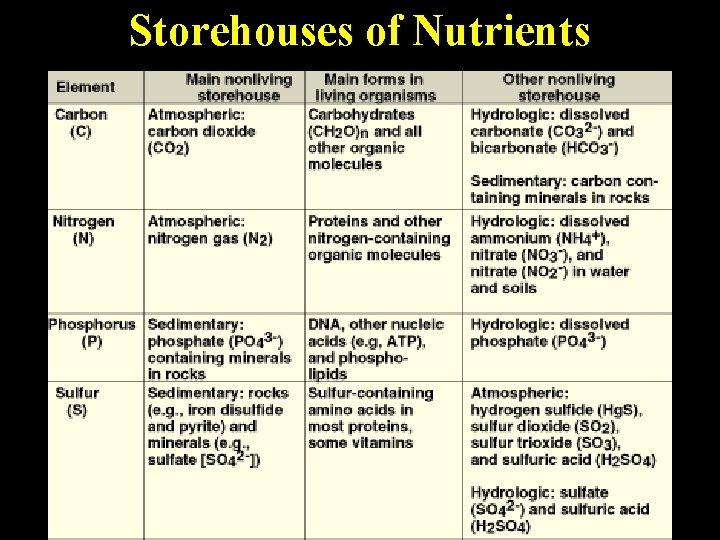
Storehouses of Nutrients
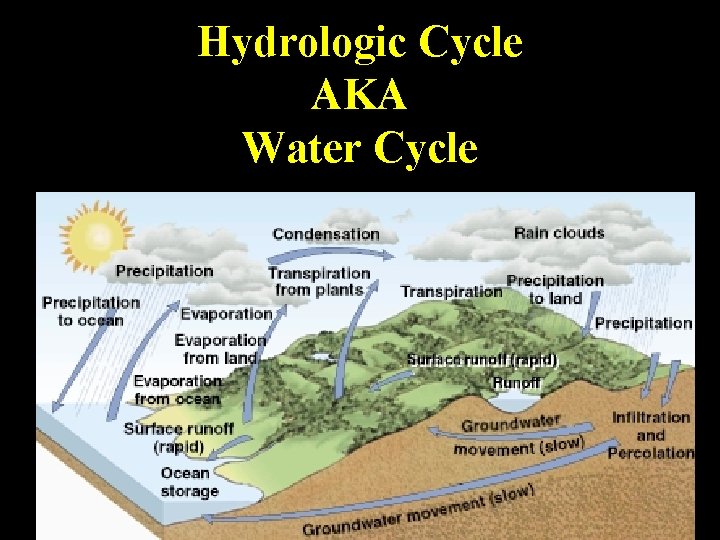
Hydrologic Cycle AKA Water Cycle

Hydrologic Cycle 1. Main factor that • Water Cycle Parts determines where – Condensation organisms can live – Precipitation 2. Powered by energy – Infiltration from sun and – Percolation to groundwater gravity – Absorption – Transpiration – Surface runoff – Evaporation

Humidity • Moisture in air – Depends on temperature – WARM AIR CAN HOLD MORE MOISTURE THAN COLD AIR • ABSOLUTE HUMIDITY – Amount on water found in a certain mass of air • RELATIVE HUMIDITY – % of water found in air at a certain temperature compared to total amount it can hold.

More Water Cycle Terms • Condensation nuclei - • Aquifer - large ash, soil, etc around underground areas of which water can porous rock that condense to form collect underground droplets water • Dew point • Water table - top of temperature at which the groundwater condensation occurs • Water cycle - purifies water

Human Intervention-Water Cycle 1. We withdraw too much water - both surface and ground – Irrigation – salt water intrusion 2. Clear vegetation – Increases runoff and reduces infiltration – RECHARGE - puts water back into ground 3. Modify water quality – Add nutrients and other pollutants

Carbon Cycle • Why do we need carbon? – Building blocks of carbohydrates, proteins, fats, nucleic acids such as DNA, RNA – Carbon dioxide gas one of main heattrapping gases (greenhouse gas) • A gaseous cycle – Also found dissolved in water – Carbon dioxide is thermostat for our atmosphere • Too little - too cold • Too much - too hot

Two Main Processes 1. Respiration -(aerobic ) organisms use free oxygen and break down glucose and other carbon based molecules and release CO 2 for reuse • Decomposition - a form of respiration 2. Photosynthesis producers remove CO 2 and convert it into complex carbohydrates such as glucose and release oxygen • Cycles fairly rapidly unless stored in trees as biomass or wetlands
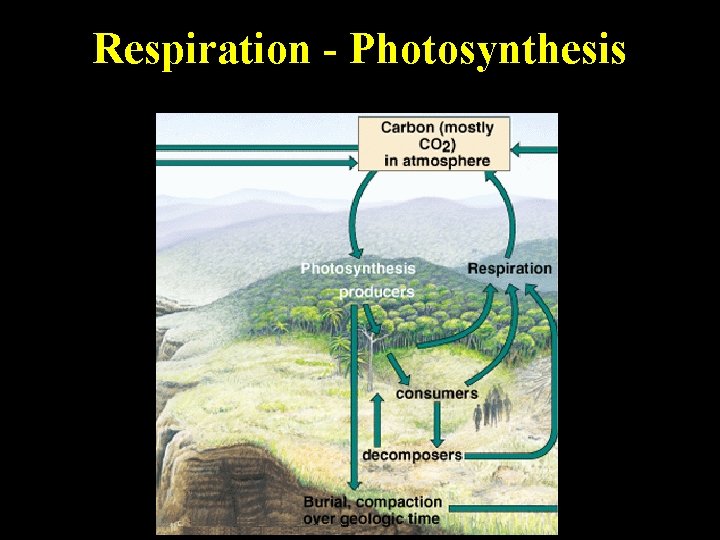
Respiration - Photosynthesis

Long-term Carbon Storage (Write Small!) • Organic matter • Limestone rock compressed and Ca. CO 3 on ocean formed into fossil floor sediments and fuels such as coal continents and oil – LARGEST STOREHOUSE – ONLY RELEASED INTO • Released when ATMOSPHERE sediments dissolve WHEN or acid rain EXTRACTED AND BURNED
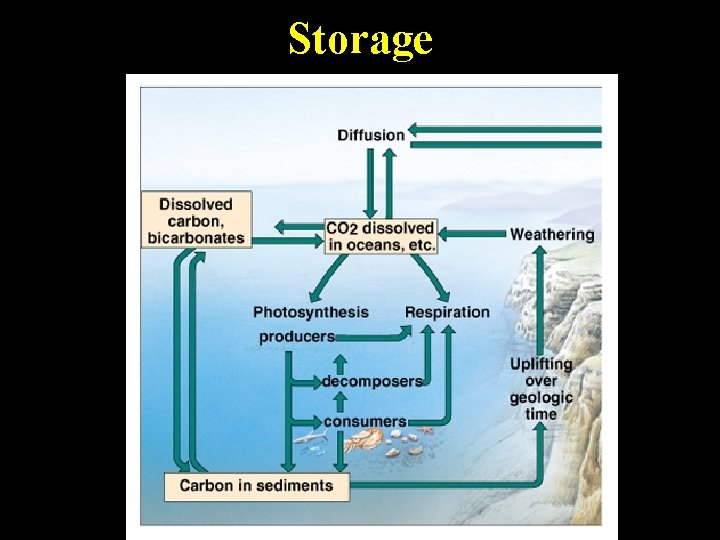
Storage

More Carbon Storage Oceans • SECOND LARGEST STOREHOUSE • CO 2 dissolved in ocean used for photosynthesis and also released into atmosphere • Marine organisms use this to make shells – Shells fall to ocean floor and are converted to limestone rock.

Human Interference-Carbon • Deforestation • WE ARE ADDING removes biomass MORE CARBON which can store DIOXIDE TO THE carbon ATMOSPHERE THAN PLANTS • Burning fossil AND OCEANS fuels - releases CAN REMOVE!! carbon stored in coal and oil into the • Causing global warming atmosphere

Pesky Humans…

Nitrogen Cycle • Used to make amino acids, proteins, DNA & RNA • OFTEN A LIMITING FACTOR IN AN ECOSYSTEM!! • The most complex cycle • A gaseous cycle • Air is 78% nitrogen which cannot be used directly by plants and animals • Lightning and certain bacteria convert nitrogen gas into compounds that can be used by multicellular organisms.

“ Fixing” Nitrogen • Nitrogen must be combined with hydrogen or oxygen to provide compounds that plants can use.

Nitrogen Cycle Terms • Nitrogen fixation: N 2 NH 3 – Gas to ammonia, making a gas usable – Cyanobacteria, legumes (special nodules with specialized bacteria on their roots) • Nitrification: NH 3 NO 2 - and NO 3 - – Ammonia in the soil to nitrite and nitrate by aerobic bacteria – Nitrite = bad, Nitrate = good!




Nitrogen Cycle Terms • Assimilation: roots absorb ions – Make DNA, amino acids, proteins – Animals get nitrogen from plants/herbivores • Ammonification: decomposers – turn waste into NH 3 and NH 4+ (ammonia, ammonium ions) • Denitrification: anaerobic bacteria in bogs – Convert NH 3 and NH 4+ back to nitrite (N 02 -) and nitrate (NO 3 -) and then to N 2 and N 2 O • Begins the cycle again!
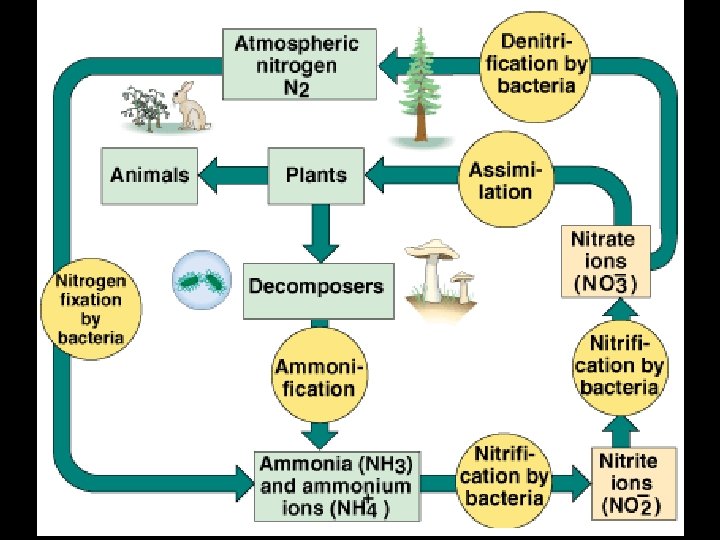
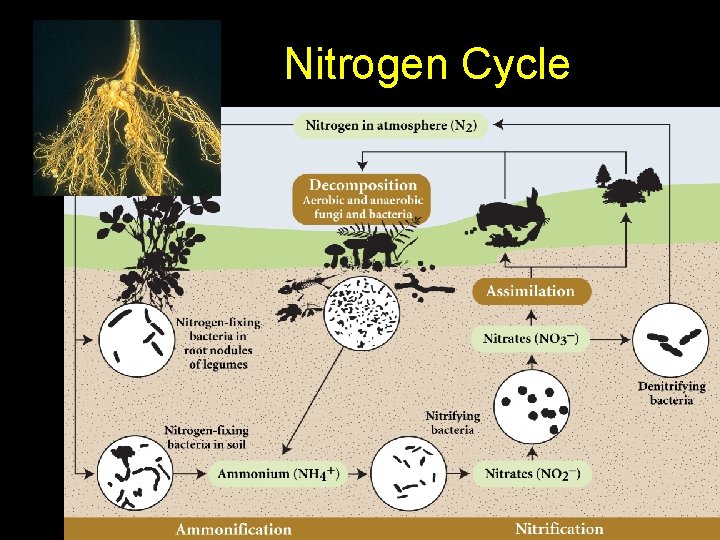
Nitrogen Cycle

Human Interference-Nitrogen • Humans have • Forms nitric acid deposition doubled amount of fixed nitrogen • N 2 O - nitrous oxide entering the – Anaerobic bacterial action on livestock nitrogen cycle waste and • 60% from inorganic commercial fertilizer – Contributes to • Burning fuels – N 2 + O 2 --> NO • Nitric oxide global warming

More Human Interference • Removing nitrogen from earth’s crust by mining nitrogen containing mineral deposits • Removing nitrogen from topsoil by harvesting nitrogen rich crops, irrigation, burning or clearing grasslands • Adding nitrogen to aquatic ecosystems from agricultural runoff and wastewater treatment plants causes “algal blooms”

Outcomes of Reactive N Increases • The Good: – Huge increases in world food production – Food independence for Asian countries – Widespread famines are (for now) a thing of the past • The Bad: – African countries have largely been left behind—still little N fertilizer use in equatorial and most of southern Africa. • The Ugly: – Nitrate contamination of ground and surface water (human health, eutrophication) – Ozone depletion by N 2 O – Acid rain as a result of NOx, resulting ecosystem disturbance

Improving N Use Efficiency • Limit N applications to crop needs plus a reasonable expectation of loss. • Place N fertilizers so as to limit loss. • Use organic N sources appropriately • Use efficient irrigation methods • Use methods to limit N losses—nitrification inhibitors, slow-release N fertilizers • Evaluate crop N needs and soil N-supplying capacity with soil/plant testing

Conclusions • Nitrogen confronts us with a dilemma: – Feeding the world’s growing population will demand increases in use of N fertilizers are responsible for 30 -50% of crop production. – However, soil-crop systems are “leaky” with respect to N. The farther up the yield curve we go, the leakier the system is. This leakage releases reactive N to the environment, with many potentially adverse environmental consequences. • Technologies exist to improve N use efficiency. However, distribution and adoption of these technologies remains problematic, mostly for economic reasons.

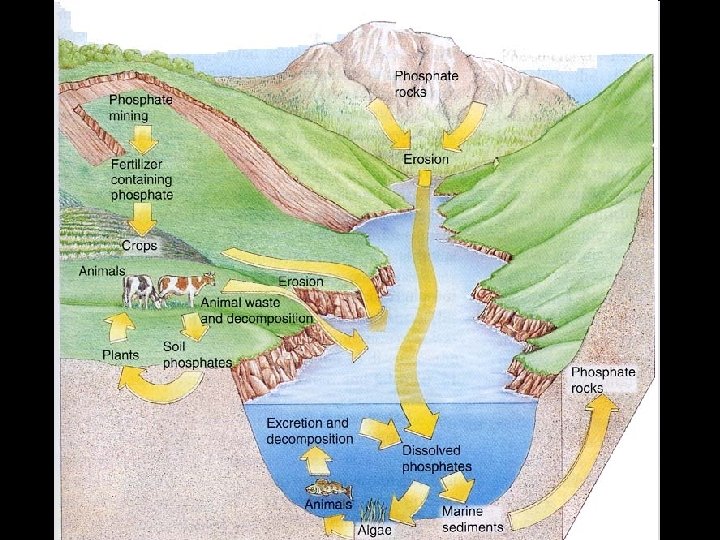
Phosphorus cycle • Phosphorus needed • Phosphorus moves in DNA, nucleic from phosphate acids, certain fats in deposits on land in cell membranes and shallow ocean bones and teeth sediments to living organisms and back to land ocean. • A sedimentary • Very slow cycle - no gaseous phase • One way flow from land to ocean

More. . . • Mainly found as • Along with nitrogen, phosphate ions (PO 4 ) can be a limiting factor in terrestrial rock and in soils. ocean bottom • Animals get sediments - released phosphorus by eating by weathering and excrete it by waste dissolved in water or decomposition and taken in by plants • Returns to soil - can get in streams - go to ocean.

More. . . • Returned only when geologic processes push up an expose sea floor. • Bird feces (guano) is rich in phosphates from sea birds but this is a small return

Human Interference • Mining for fertilizer and detergents • Deforestation - soil loses phosphorus • Animal waste runoff & discharge from wastewater treatment plants causes algal blooms in water
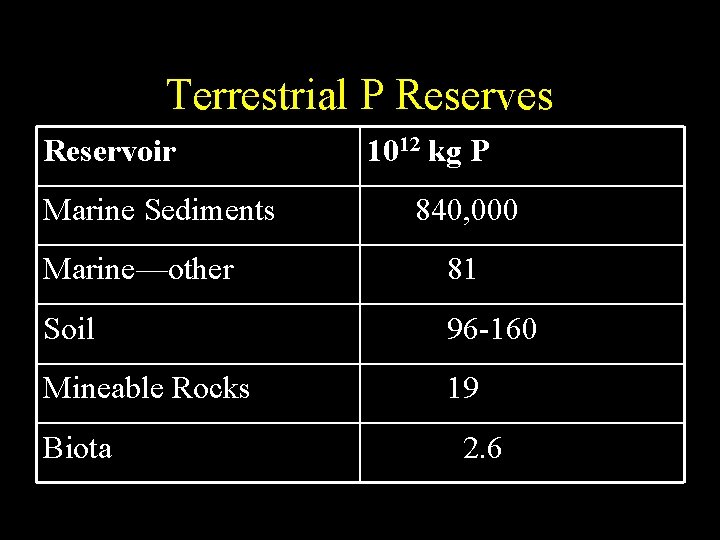
Terrestrial P Reserves Reservoir Marine Sediments 1012 kg P 840, 000 Marine—other 81 Soil 96 -160 Mineable Rocks 19 Biota 2. 6

The Problems with Phosphorus • There is no P counterpart to N fixation—i. e. virtually all soils must receive P fertilizers for optimum productivity of crops. • P fertilizer uptake by crops is low (29% of P applied in U. S. , <1% in Hawaii!) • Natural P sources (e. g. ‘phosphate rock’) are very poor sources of P for plants. • Small amounts of P added to surface waters can cause eutrophication.

Sulfur cycle • Needed for proteins and vitamins • A gaseous cycle • Much is stored in rocks as sulfates • Gets into atmosphere from: – – Volcanoes Swamps and bogs Salt marshes Anaerobic respiration • Marine algae produce dimethylsulfate - DMS - affects cloud cover and climate • Sulfur dioxide forms sulfuric acid • Organisms get it by eating and excrete or decay to release it.

Human Interference-Sulfur • Burning high sulfur coal for electric power • Refining petroleum • Smelting metals such as copper, lead, and zinc
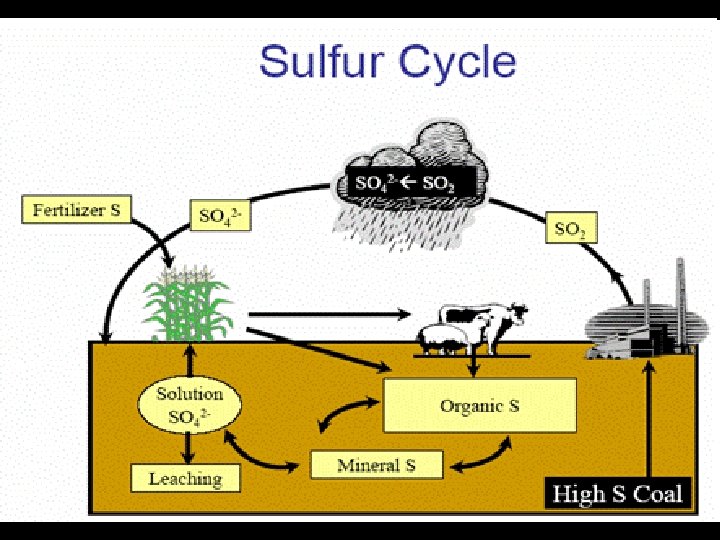

Interesting Things About S • The sulfur and nitrogen cycles have several similarities: – Organic forms important in soils – Microbial reactions important – Gas phase important – Subject to redox – Atmospheric deposition of S

Reservoir • • Reservoir: pool of stock of material source (emits), sink (takes in), can be both Flux: rate of transfer between reservoirs per unit time Average residence time: time taken for material to cycle through system • average residence time = total size of reservoir/rate of flow • short residence time: small reservoirs, high transfer rates, easy to impact • long residence time: large reservoirs, slow transfer, slower to change

____ is the absorption of nitrogen into plants through the soil. A. ) nitrogen fixation B. ) assimilation C. ) nitrification D. ) denitrification E. ) ammonification

Which of the following processes makes the nitrogen product usable by the plant? A. ) nitrogen fixation B. ) assimilation C. ) nitrification D. ) denitrification E. ) ammonification

Which trophic level has the highest amount of biomass? A. ) tertiary consumers B. ) zooplankton C. ) phytoplankton D. ) primary producers E. ) primary consumers
- Slides: 47