Bioethics and commercial RD in the life sciences

Bioethics and commercial R&D in the life sciences WIPO LIFE SCIENCES SYMPOSIUM ON INTELLECTUAL PROPERTY (IP) AND BIOETHICS SEPTEMBER 4, 2007

Our mission at Novartis We want to discover, develop and successfully market innovative products to prevent and cure diseases, to ease suffering, and to enhance quality of life. We also want to provide a shareholder return that reflects outstanding performance and to adequately reward those who invest ideas and work in our company. 2 | Corporate Presentation | Corporate Communications | May 2007 Daniel Vasella Chairman & CEO

Novartis at a glance § Unique portfolio to meet changing healthcare needs: • Leading innovative pharmaceuticals • High-quality, low-cost generics • Preventive vaccines • Consumer health products § World’s third largest pharmaceutical company by sales § One of 20 largest companies by market capitalization § Ranked among most respected companies worldwide 3 | Corporate Presentation | Corporate Communications | May 2007
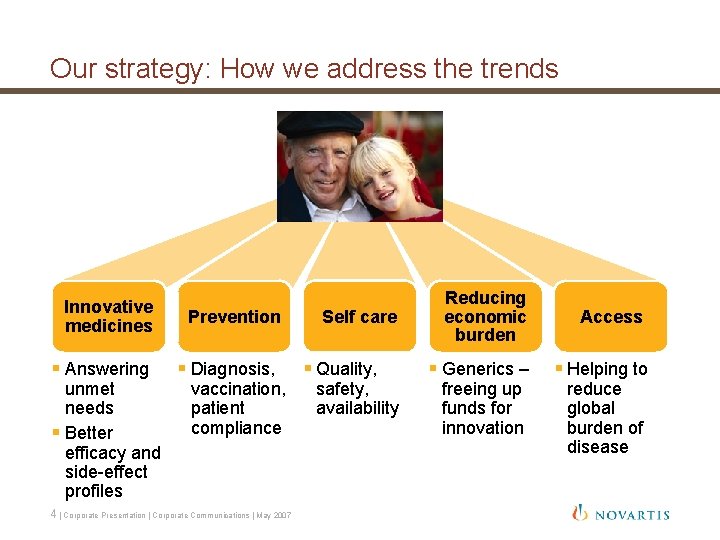
Our strategy: How we address the trends Innovative medicines § Answering unmet needs § Better efficacy and side-effect profiles Prevention § Diagnosis, vaccination, patient compliance 4 | Corporate Presentation | Corporate Communications | May 2007 Self care § Quality, safety, availability Reducing economic burden § Generics – freeing up funds for innovation Access § Helping to reduce global burden of disease

Bioethics, IP and Commercial R&D (Bio)Ethics IP R&D 5 | Corporate Presentation | Corporate Communications | May 2007

Bioethics & Commercial R&D § Bioethical issues are inherent in almost all life-science R&D § Many of them relate to very fundamental ethical questions (e. g. use of animals in biomedical R&D, healthy volunteers) § Large, multinational corporations represent a “melting-pot” of diverse intra- and international values and norms § Proper understanding of key ethical issues is essential to gain and sustain trust and credibility internally and externally § For many of these issues an external ethical review is mandatory (e. g. ethics committees for clinical trials) § However, often neither societies at large nor legal systems provide clear guidance (e. g. somatic nuclear transfer technologies) 6 | Corporate Presentation | Corporate Communications | May 2007
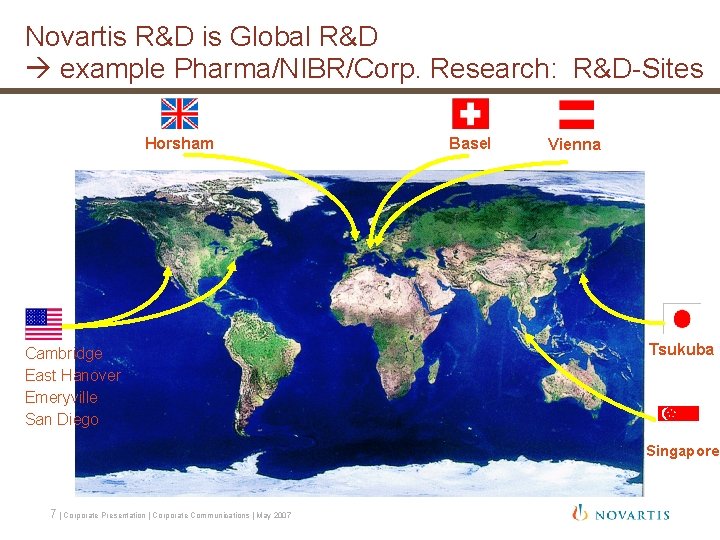
Novartis R&D is Global R&D example Pharma/NIBR/Corp. Research: R&D-Sites Horsham Cambridge East Hanover Emeryville San Diego Basel Vienna Tsukuba Singapore 7 | Corporate Presentation | Corporate Communications | May 2007
Global R&D requires common grounds – an immense challenge for applied ethics Huge conceptual differences Highly diverse norms 8 | Corporate Presentation | Corporate Communications | May 2007

In a global environment…. Which “Ethics” is the right one? 9 | Corporate Presentation | Corporate Communications | May 2007

IP and Commercial R&D § IP is central to all Novartis R&D activities is f l e § Our unique position in the market (strong Rx, Gx and Vaccines s it m e st y business) enables us to better analyze and understand many s P I e h t controversial issues e f d i n n a k us ion t o a m v a o can be f n § However, very rarely intellectual property per se e n g i h n t i e e v e b k e i L a leading cause for (bio)ethical e hi r considered as problems c : a a n. o s o ti t n n i o o s. i i n l t t o a a ra se t c v i o f u rop o i h t e n P c sethical problems n incasesalthe u s j l y § In the majority of publicly debated l n l u f a a e ic ss or h m e t m c e e c arise either prior ofuthe e IP process (e. g. “production” th to hinitiation u r i s r e q h c t of. It transgenic whi hichorreby the way successful inventions are is ra s inorganisms) wappropriate benefit sharing or adequate y d a e commercialized (e. g. w liz a i the c r emedicines) access to m m co 10 | Corporate Presentation | Corporate Communications | May 2007
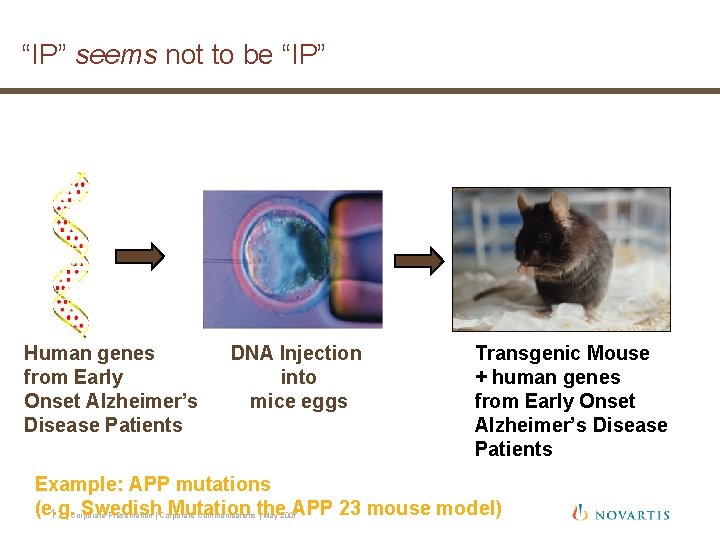
“IP” seems not to be “IP” Human genes from Early Onset Alzheimer’s Disease Patients DNA Injection into mice eggs Transgenic Mouse + human genes from Early Onset Alzheimer’s Disease Patients Example: APP mutations (e. g. 11 Swedish Mutation the APP 23 mouse model) | Corporate Presentation | Corporate Communications | May 2007

Bioethics, IP and Commercial R&D § Although different disciplines, cultural values, moral and ethics are essentially different throughout the globe – and often even within a given nation/society § Commercial entities have to operate in an ethically sound way – however, they are not (and shall not) act as normsetting entities § If critical assessment of existing norms and their applicability to real-life business/R&D activities reveals a gap of interculturally acceptable guidance ethical support/facilitation is most appropriate § Novartis is seeking external ethical advise on different levels example: Novartis Ethics Advisory Board 12 | Corporate Presentation | Corporate Communications | May 2007
- Slides: 12