BIOEQUIVALENCE AND GOOD MANUFACTURING PRACTICES GMP IN CHILE

BIOEQUIVALENCE AND GOOD MANUFACTURING PRACTICES (GMP) IN CHILE PRESENTER TO INSERT THEIR ORGANIZATION’S LOGO AND INFORMATION HERE

BIOEQUIVALENCE STUDIES Based on American normatives (FDA), European regulations, WHO recommendations and type IV Agencies: Establish the requirements for: • Submissions of study protocols or designs before performing bioequivalence studies • Submissions of BE study results performed under the jurisdictions of regulatory agencies accepted by the Agencia Nacional de Medicamentos (ANAMED) or • Submissions of prequalified drug products as bioequivalent under the WHO prequalification program (HIV, Tuberculosis) 2
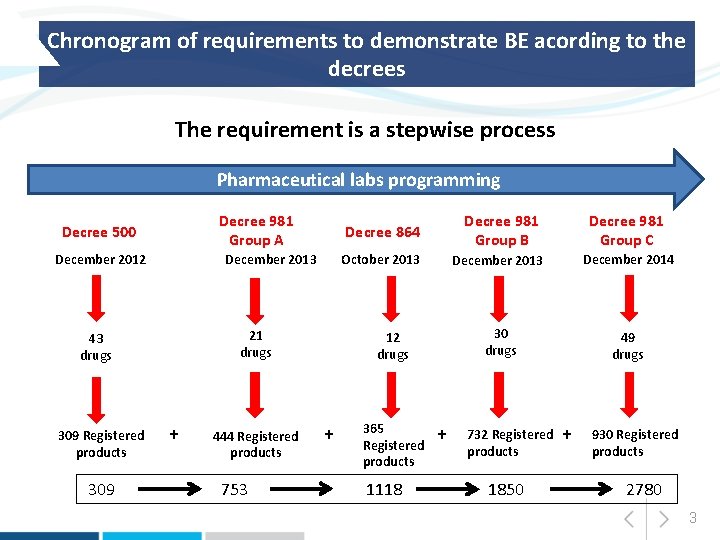
Chronogram of requirements to demonstrate BE acording to the decrees The requirement is a stepwise process Pharmaceutical labs programming Decree 981 Group A Decree 500 December 2012 December 2013 21 drugs 43 drugs 309 Registered products 309 + 444 Registered products 753 Decree 864 Decree 981 Group B Decree 981 Group C October 2013 December 2014 30 drugs 49 drugs 12 drugs + 365 Registered products 1118 + 732 Registered products 1850 + 930 Registered products 2780 3
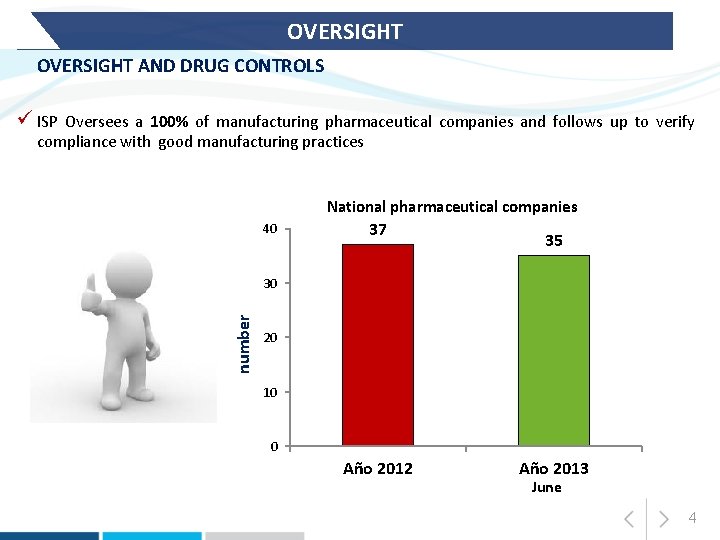
OVERSIGHT AND DRUG CONTROLS ü ISP Oversees a 100% of manufacturing pharmaceutical companies and follows up to verify compliance with good manufacturing practices National pharmaceutical companies 40 37 35 number 30 20 10 0 Año 2012 Año 2013 June 4
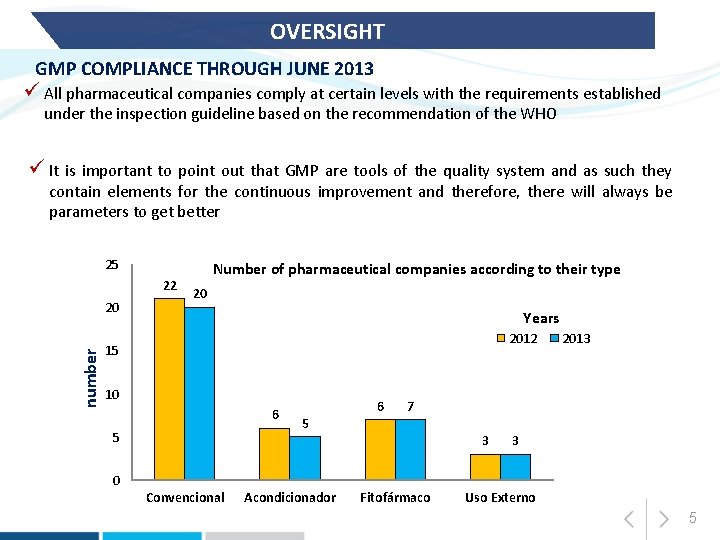
OVERSIGHT GMP COMPLIANCE THROUGH JUNE 2013 ü All pharmaceutical companies comply at certain levels with the requirements established under the inspection guideline based on the recommendation of the WHO ü It is important to point out that GMP are tools of the quality system and as such they contain elements for the continuous improvement and therefore, there will always be parameters to get better 25 22 number 20 Number of pharmaceutical companies according to their type 20 Years 2012 15 10 6 5 6 2013 7 5 3 3 0 Convencional Acondicionador Fitofármaco Uso Externo 5
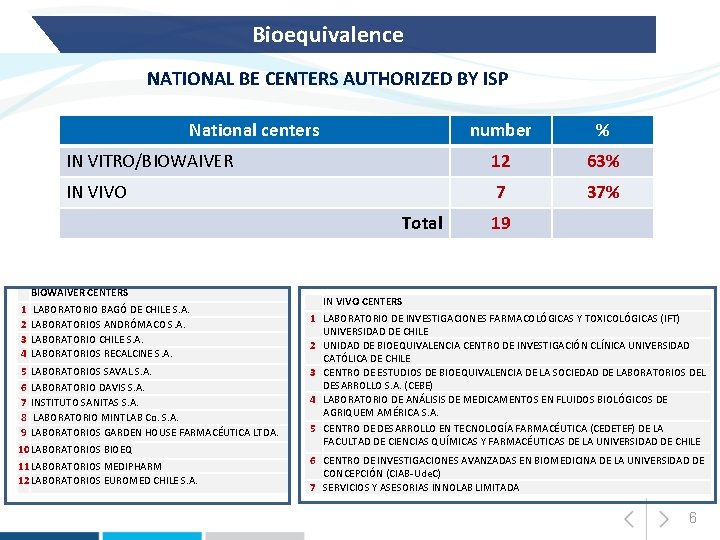
Bioequivalence NATIONAL BE CENTERS AUTHORIZED BY ISP National centers number % IN VITRO/BIOWAIVER 12 63% IN VIVO 7 37% Total BIOWAIVER CENTERS 1 2 3 4 LABORATORIO BAGÓ DE CHILE S. A. LABORATORIOS ANDRÓMACO S. A. LABORATORIO CHILE S. A. LABORATORIOS RECALCINE S. A. 5 LABORATORIOS SAVAL S. A. 6 LABORATORIO DAVIS S. A. 7 INSTITUTO SANITAS S. A. 8 LABORATORIO MINTLAB Co. S. A. 9 LABORATORIOS GARDEN HOUSE FARMACÉUTICA LTDA. 10 LABORATORIOS BIOEQ 11 LABORATORIOS MEDIPHARM 12 LABORATORIOS EUROMED CHILE S. A. 19 IN VIVO CENTERS 1 LABORATORIO DE INVESTIGACIONES FARMACOLÓGICAS Y TOXICOLÓGICAS (IFT) UNIVERSIDAD DE CHILE 2 UNIDAD DE BIOEQUIVALENCIA CENTRO DE INVESTIGACIÓN CLÍNICA UNIVERSIDAD CATÓLICA DE CHILE 3 CENTRO DE ESTUDIOS DE BIOEQUIVALENCIA DE LA SOCIEDAD DE LABORATORIOS DEL DESARROLLO S. A. (CEBE) 4 LABORATORIO DE ANÁLISIS DE MEDICAMENTOS EN FLUIDOS BIOLÓGICOS DE AGRIQUEM AMÉRICA S. A. 5 CENTRO DE DESARROLLO EN TECNOLOGÍA FARMACÉUTICA (CEDETEF) DE LA FACULTAD DE CIENCIAS QUÍMICAS Y FARMACÉUTICAS DE LA UNIVERSIDAD DE CHILE 6 CENTRO DE INVESTIGACIONES AVANZADAS EN BIOMEDICINA DE LA UNIVERSIDAD DE CONCEPCIÓN (CIAB-Ude. C) 7 SERVICIOS Y ASESORIAS INNOLAB LIMITADA 6
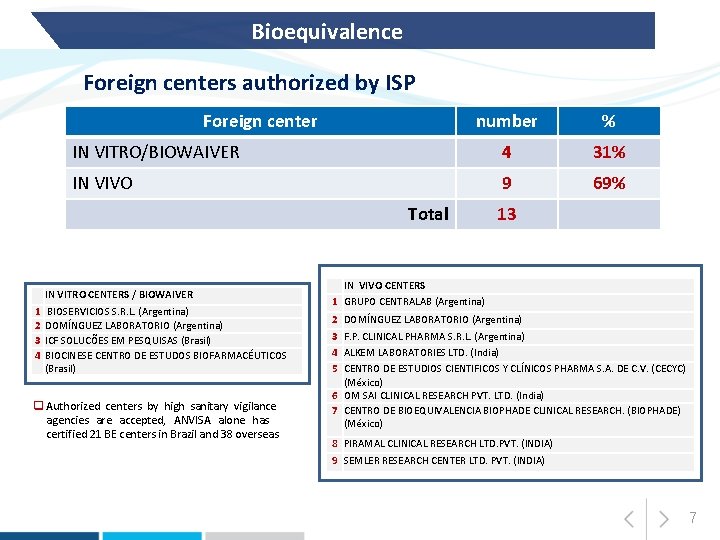
Bioequivalence Foreign centers authorized by ISP Foreign center number % IN VITRO/BIOWAIVER 4 31% IN VIVO 9 69% Total IN VITRO CENTERS / BIOWAIVER 1 2 3 4 BIOSERVICIOS S. R. L. (Argentina) DOMÍNGUEZ LABORATORIO (Argentina) ICF SOLUCÕES EM PESQUISAS (Brasil) BIOCINESE CENTRO DE ESTUDOS BIOFARMACÉUTICOS (Brasil) q Authorized centers by high sanitary vigilance agencies are accepted, ANVISA alone has certified 21 BE centers in Brazil and 38 overseas 13 IN VIVO CENTERS 1 GRUPO CENTRALAB (Argentina) 2 DOMÍNGUEZ LABORATORIO (Argentina) 3 F. P. CLINICAL PHARMA S. R. L. (Argentina) 4 ALKEM LABORATORIES LTD. (India) 5 CENTRO DE ESTUDIOS CIENTIFICOS Y CLÍNICOS PHARMA S. A. DE C. V. (CECYC) (México) 6 OM SAI CLINICAL RESEARCH PVT. LTD. (India) 7 CENTRO DE BIOEQUIVALENCIA BIOPHADE CLINICAL RESEARCH. (BIOPHADE) (México) 8 PIRAMAL CLINICAL RESEARCH LTD. PVT. (INDIA) 9 SEMLER RESEARCH CENTER LTD. PVT. (INDIA) 7
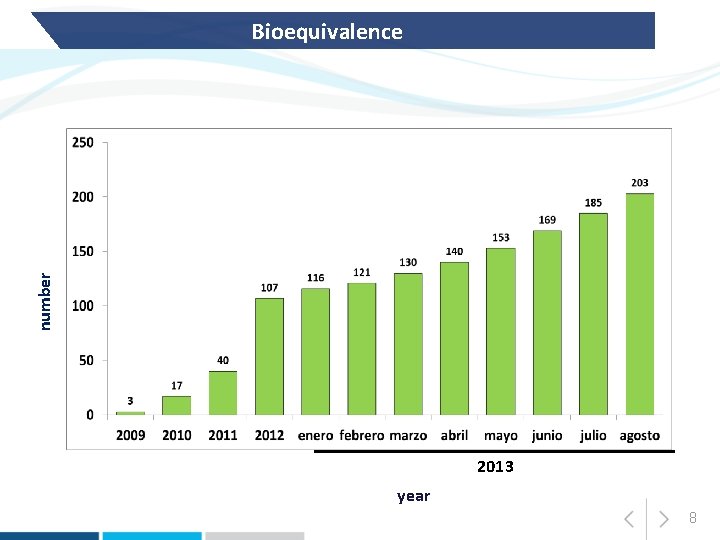
number Bioequivalence 2013 year 8
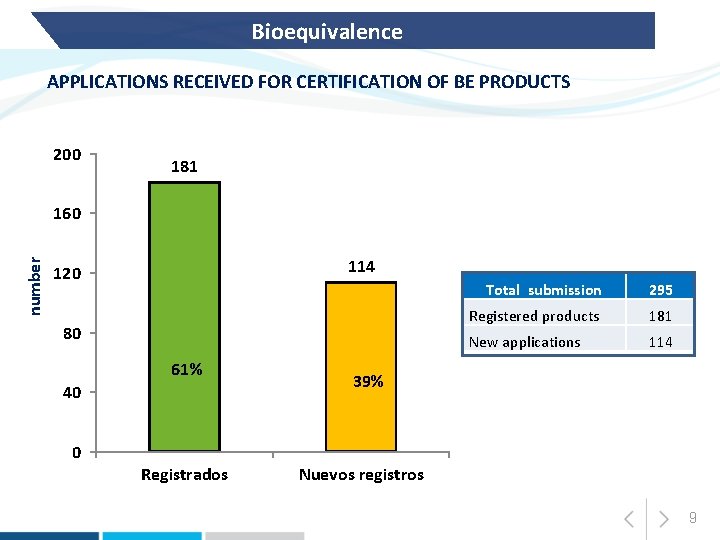
Bioequivalence APPLICATIONS RECEIVED FOR CERTIFICATION OF BE PRODUCTS 200 181 number 160 114 120 80 61% 40 0 Registrados Total submission 295 Registered products 181 New applications 114 39% Nuevos registros 9
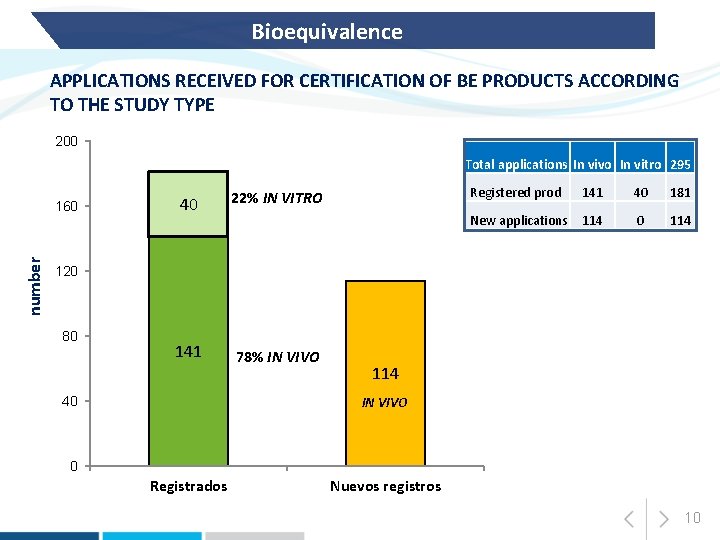
Bioequivalence APPLICATIONS RECEIVED FOR CERTIFICATION OF BE PRODUCTS ACCORDING TO THE STUDY TYPE 200 Total applications In vivo In vitro 295 number 160 40 22% IN VITRO 141 78% IN VIVO Registered prod 141 40 181 New applications 114 0 114 120 80 114 IN VIVO 40 0 Registrados Nuevos registros 10
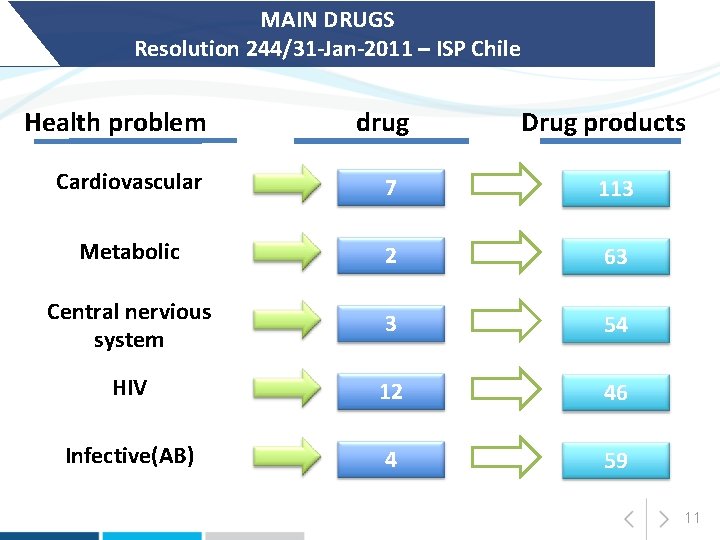
MAIN DRUGS Resolution 244/31 -Jan-2011 – ISP Chile Health problem drug Drug products Cardiovascular 7 113 Metabolic 2 63 Central nervious system 3 54 HIV 12 46 Infective(AB) 4 59 11

Instituto de Salud Pública de Chile THANKS!!! 12
- Slides: 12