BIOENERGY Potential Photosynthesis Fuels From biomass Biomass processes
BIO-ENERGY Potential Photosynthesis Fuels From bio-mass Bio-mass processes Applications of biochemical Conversions Chei From. Bioes:

INTRODUCTION Bio-energy may be simply defined as that energy produced from bio-masses. A bio-mass is an organic material resulting from the photochemical solar energy conversion and which can be used for energy generation. Bio mass can also be defined as all types of animals and plant materials which can be converted into energy. It includes trees and shrubs, grasses, algae, aquatic plants, agricultural and forest residues, energy crops and all forms of wastes. There is a significant worldwide potential for the utilization of bio-energy. In many of the developing countries, bio-mass already accounts for up to 90% of their energy use. The renewed emphasis on biological conversion systems arises from the fact that solar energy can be converted directly into a storable fuel and other methods of utilizing solar energy require a separate energy storage system. The carbohydrates can be reduced to very desirable fuels such as alcohol, hydrogen or methane, a process which can also be applied to organic waste materials which result from food or wood production.

Bio-energy may be simply defined as that energy produced from bio-masses. A bio-mass is an organic material resulting from the photochemical solar energy conversion and which can be used for energy generation. Bio mass can also be defined as all types of animals and plant materials which can be converted into energy. It includes trees and shrubs, grasses, algae, aquatic plants, agricultural and forest residues, energy crops and all forms of wastes.

solar energy can be converted directly into a storable fuel and other methods of utilizing solar energy require a separate energy storage system. The carbohydrates can be reduced to very desirable fuels such as alcohol, hydrogen or methane, a process which can also be applied to organic waste materials which result from food or wood production.

Biological Conversion of Solar Energy (Photosynthesis): Photosynthesis is the utilization of solar energy to synthesize organic material, e. g. carbohydrates, from carbon dioxide and water in the presence of chloroplast. Oxygen is also liberated from the reaction which could be represented as shown below. Photosynthesis produces complex forms of organic materials which vary considerably in composition. Their average composition however, is close to that of a carbohydrate. The elementary molecule, CH 2 O, may be considered as typical of the groups of molecules from which carbohydrates are formed. The carbohydrates may be sugar (C 12 H 22 O 11) or the more complex starches or cellulose which account for a large portion of the living material of the earth's biosphere and the highly polymerized forms of a related molecule, C 5 H 10 O 5.








Glossary of chapter 7 (Bio- energy): Bio- energy: Is that energy produced from bio- masses. A bio- mass: Is an organic material resulting from the photochemical solar energy conversion and which can be used for energy generation. Photosynthesis: Is the utilization of solar energy to synthesize organic material.

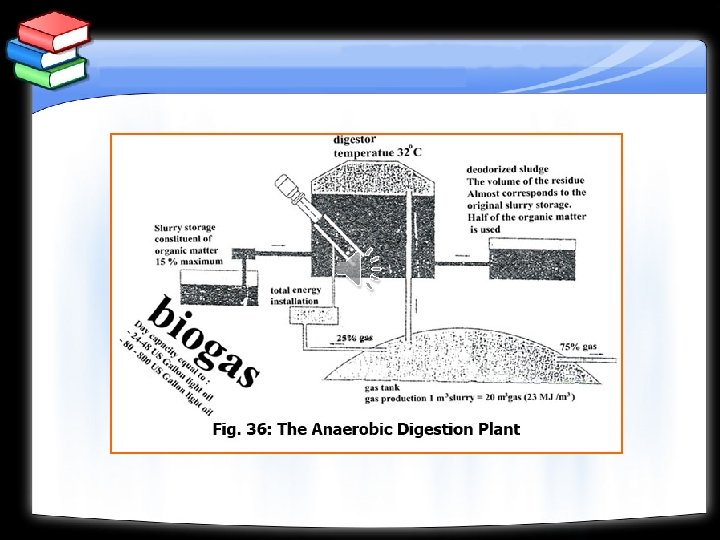
Anaerobic fermentation: Is a biological process in which anaerobic bacteria converts the organic matter into bio- gas, sludge and a final effluent and produces methanol. Aerobic fermentation: Is a process where the materials containing starches and simple sugars can be fermented with the aid of enzymes to produce ethyl alcohol. Pyrolysis: Is a process of destructive distillation of organic materials. It is conducted for organic materials to produce gaseous and liquid fuels.

Equations of chapter 7 Bio- energy: The formation of CH 2 O requires an energy of 112 X 106 calorie for each kilogram- mole and the reaction is represented as follows: Chloroplast → CH 2 O + O 2 (112 X 106 cal/Kg mole) On combustion, CH 2 O breaks resulting in an energy release of 112 X 106 cal/ mole, as follows: CH 2 O + O 2 → CO 2 + H 2 O + 112, 000 cal The hydrolysis of cellulose, to be ready for fermentation, is represented by the equation: C 6 H 10 O 5 + n. H 2 O →( C 6 H 10 O 6)n + 3 n. CO 2 + 3 n. CH 4

In the anaerobic fermentation process, yeast is used where it converts hexose sugars to ethanol and carbon dioxide according to the equaton: C 6 H 12 O 6 →yeast 2 C 2 H 5 OH + 2 CO 2 When pyrolysis is used for producing hydrogen, organic materials are partially converted to oils when heated to temperatures of 300 - 400 OC at pressures of 2000 - 4000 psi. The equations are as follows: n. CO + n. H 2 O → n. CO 2 + n. H 2 Cn (H 2 O)n → n H 2 + H (CH 2)n H



- Slides: 20