BIOENERGETICS Copyright Cmassengale 1 What is Bioenergetics The

BIOENERGETICS Copyright Cmassengale 1

What is Bioenergetics? The study of energy in living systems (environments) and the organisms (plants and animals) that utilize them Copyright Cmassengale 2

Energy n n Required by all organisms May be Kinetic or Potential energy Copyright Cmassengale 3

Kinetic Energy n n Energy of Motion Heat and light energy are examples Copyright Cmassengale 4
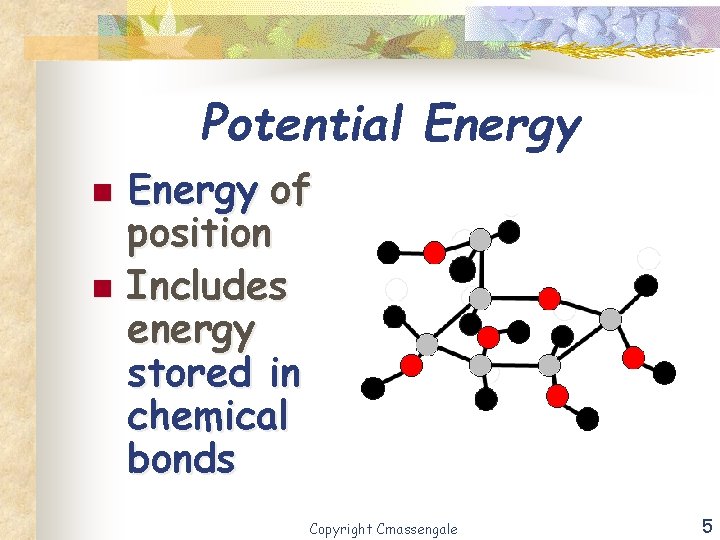
Potential Energy n n Energy of position Includes energy stored in chemical bonds Copyright Cmassengale 5

Two Types of Energy Reactions Copyright Cmassengale 6

Endergonic Reactions n n Chemical reaction that requires a net input of energy Photosynthesis SUN photons Light Energy 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 (glucose) Copyright Cmassengale 7

Exergonic Reactions n n Chemical reactions that releases energy Cellular Respiration Energy C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O+ ATP (glucose) Copyright Cmassengale 8

Metabolic Reactions of Cells Copyright Cmassengale 9
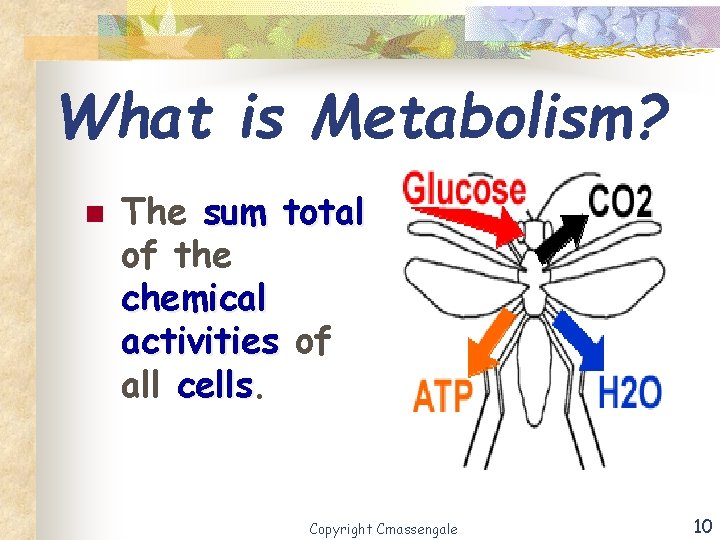
What is Metabolism? n The sum total of the chemical activities of all cells Copyright Cmassengale 10
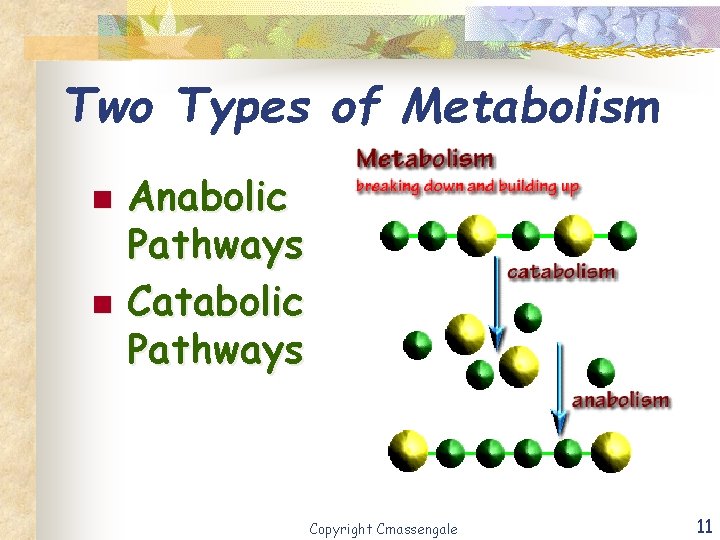
Two Types of Metabolism n n Anabolic Pathways Catabolic Pathways Copyright Cmassengale 11
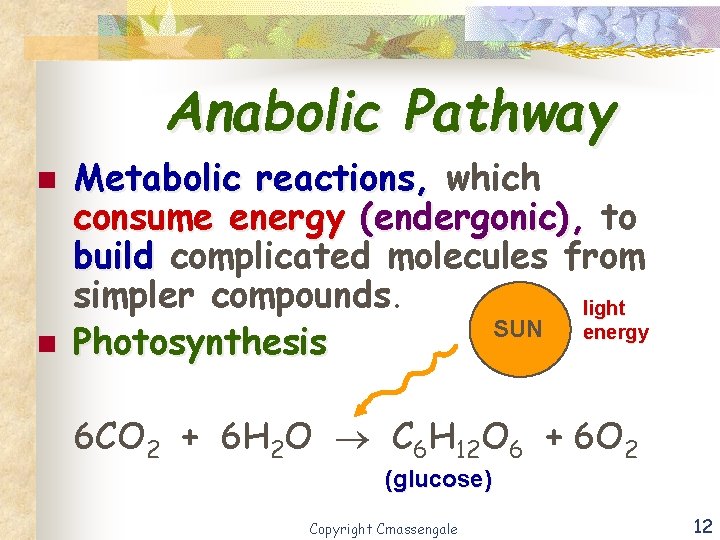
Anabolic Pathway n n Metabolic reactions, which consume energy (endergonic), to build complicated molecules from simpler compounds. light SUN energy Photosynthesis 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 (glucose) Copyright Cmassengale 12

Catabolic Pathway n Metabolic reactions which release energy (exergonic) by breaking down complex molecules in simpler compounds energy Cellular Respiration n C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + ATP (glucose) Copyright Cmassengale 13
Cellular Energy ATP Copyright Cmassengale 14
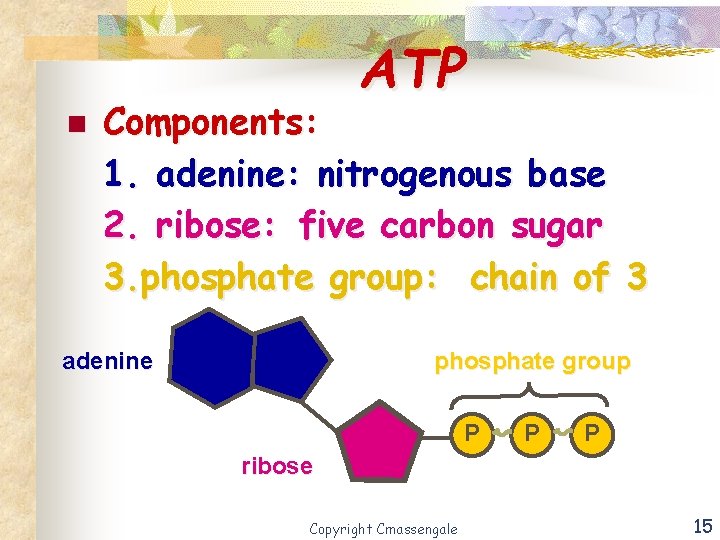
ATP n Components: 1. adenine: nitrogenous base 2. ribose: five carbon sugar 3. phosphate group: chain of 3 adenine phosphate group P P P ribose Copyright Cmassengale 15
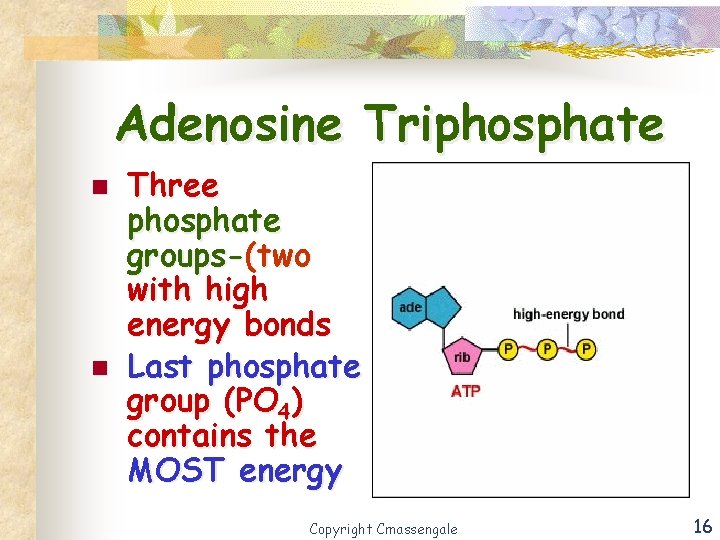
Adenosine Triphosphate n n Three phosphate groups-(two with high energy bonds Last phosphate group (PO 4) contains the MOST energy Copyright Cmassengale 16

Breaking the Bonds of ATP n n n Process is called phosphorylation Occurs continually in cells Enzyme ATP-ase can weaken & break last PO 4 bond releasing energy & free PO 4 Copyright Cmassengale 17

How does ATP work ? n n Organisms use enzymes to break down energy-rich glucose to release its potential energy This energy is trapped and stored in the form of adenosine triphosphate(ATP) Copyright Cmassengale 18

How Much ATP Do Cells Use? n It is estimated that each cell will generate and consume approximately 10, 000 molecules of ATP per second Copyright Cmassengale 19
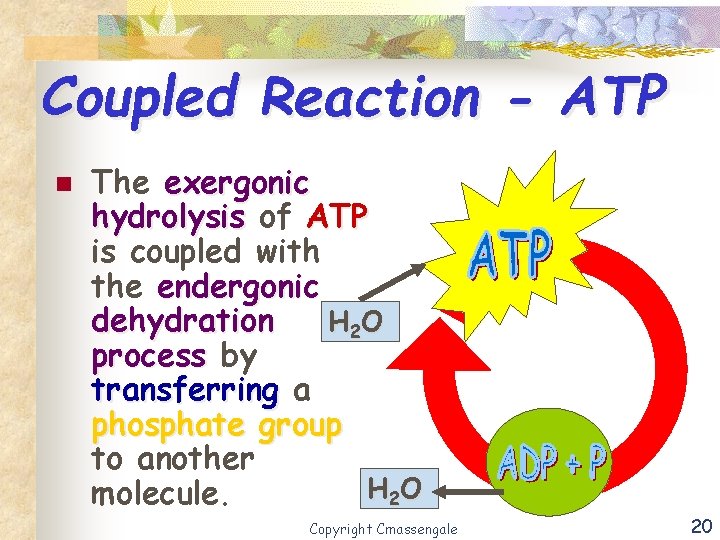
Coupled Reaction - ATP n The exergonic hydrolysis of ATP is coupled with the endergonic dehydration H 2 O process by transferring a phosphate group to another H 2 O molecule. Copyright Cmassengale 20
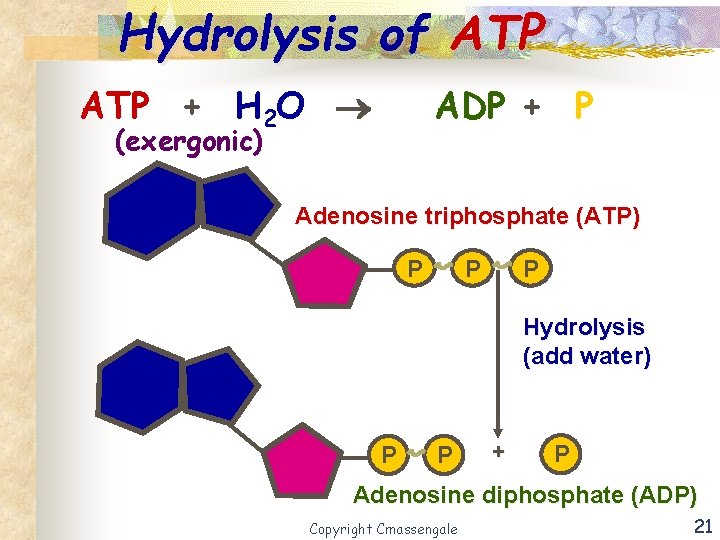
Hydrolysis of ATP + H 2 O ADP + P (exergonic) Adenosine triphosphate (ATP) P P P Hydrolysis (add water) P P + P Adenosine diphosphate (ADP) Copyright Cmassengale 21
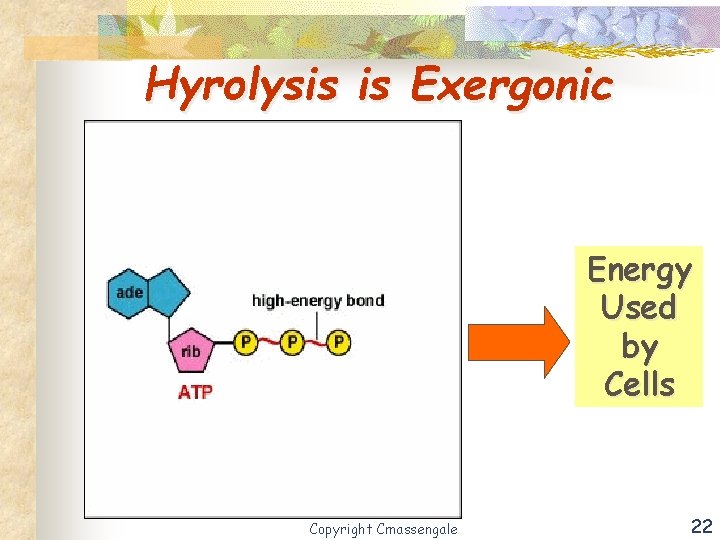
Hyrolysis is Exergonic Energy Used by Cells Copyright Cmassengale 22
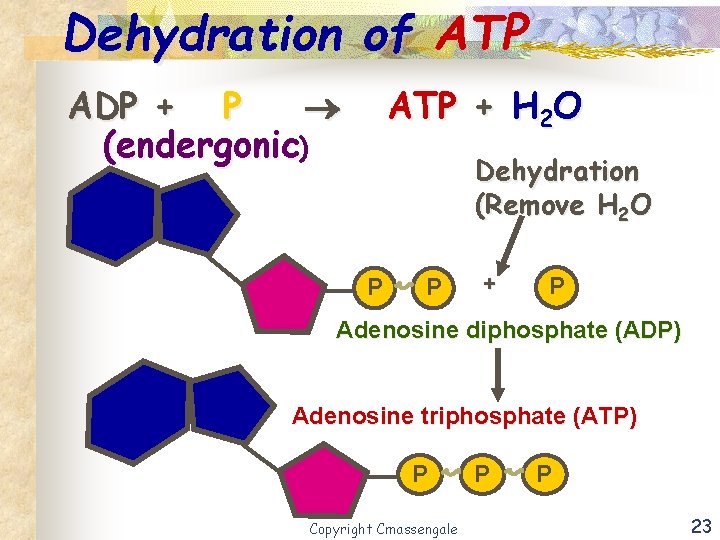
Dehydration of ATP ADP + P (endergonic) ATP + H 2 O Dehydration (Remove H 2 O P P + P Adenosine diphosphate (ADP) Adenosine triphosphate (ATP) P Copyright Cmassengale P P 23
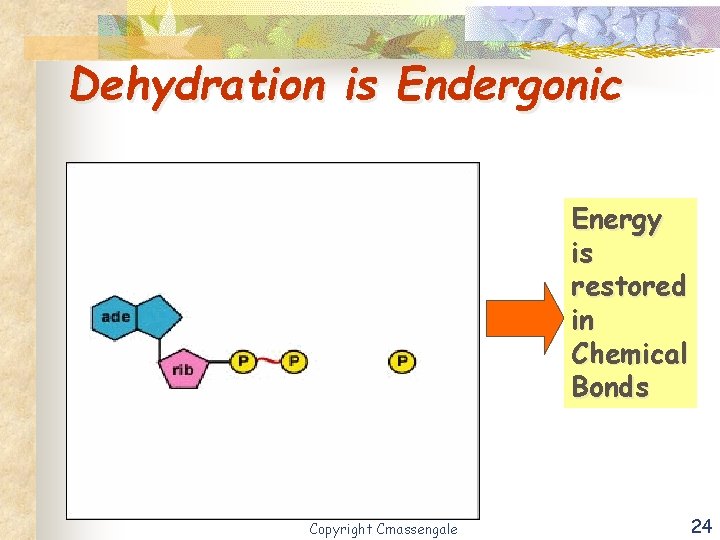
Dehydration is Endergonic Energy is restored in Chemical Bonds Copyright Cmassengale 24

Review Copyright Cmassengale 25

How many high energy phosphate bonds does ATP have? Copyright Cmassengale 26

Which is true of photosyntheis? Anabolic or Catabolic Exergonic Or Endergonic Copyright Cmassengale 27

The breakdown of ATP is due to: Dehydration or Hydrolysis H 2 O added or H 2 O removed Copyright Cmassengale 28

Which Reactions are often Coupled in Organisms Hydrolysis Anabolism Endergonic or BOTH Dehydration or Catabolism or Exergonic BOTH Copyright Cmassengale 29

Ready for the test? If not, review and try again!!! Copyright Cmassengale 30
- Slides: 30