BIOENERGETICS By Ms Preeti S Salve KLE College

BIOENERGETICS By: Ms. Preeti S. Salve KLE College of Pharmacy, Belagavi A constituent Unit of KLE Academy of Higher Education and Research Nehru Nagar, Belagavi – 590 010, Karnataka, India Phone: 0831 -2471399; Fax: 0831 -2472387; Web: http: //www. klepharm. edu. E-mail: principal@klepharm. edu
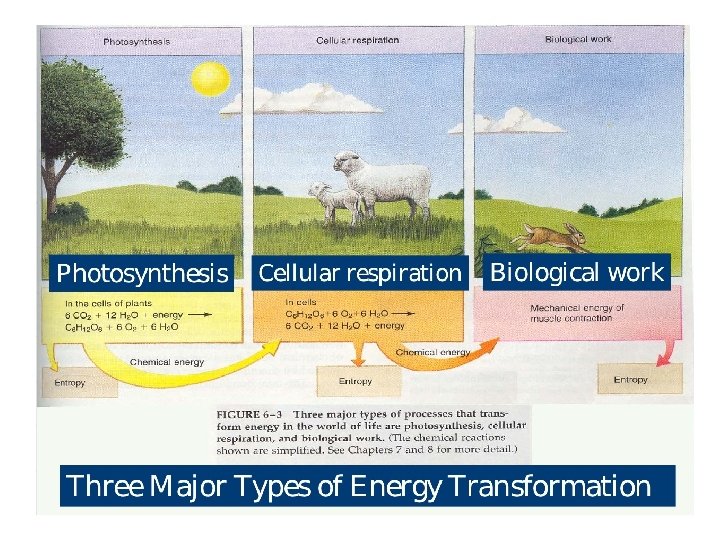

Bioenergetics / Biochemical thermodynamics § It is the study of energy changes accompanying biochemical reactions § It describes the transfer and utilization of energy in biological systems § It is concerned with the initial and final energy states of reaction components and not the reaction mechanism or the time required for the chemical reaction

Free energy § It is the energy actually available to do work (utilizable energy) § Change in free energy (∆G) predicts whether a chemical reaction is feasible/favourable § Reactions can occur simultaneously if they are accompanied by decrease in free energy § ∆G approaches zero as reaction proceeds to equilibrium

§ During a chemical reaction, heat may be released or absorbed § Enthalpy (∆H) is a measure of the change in heat content of the reactants, compared to products § Entropy (∆S) represents a change in the randomness or disorder of reactants and products § Entropy reaches a maximum as the reaction approaches equilibrium § The reactions of biological systems involve a temporary decrease in entropy

Laws of thermodynamics § 1 st law: Energy can neither be created nor be destroyed, but can be converted from one form to another § 2 nd law: Total entropy of a system must increase is a process has to occur simultaneously

§ Combining the two laws of thermodynamics, Gibbs in 1878, came up with the following equation § The relation between the change in free energy (∆G), enthalpy (∆H) and entropy (∆S) is expressed as – ∆G = ∆H – T ∆S where, ∆G = the change in free energy of a reacting system ∆H = the change in heat content or enthalpy of this system T = the absolute temperature in Kelvin at which the process is taking place ∆S = the change in entropy of this system

Free energy change § ∆G predicts the change in free energy and thus the direction of the reaction, at any specified concentration of the reactants and products § Standard free energy ∆G° is the energy change when the reactants and products are at a concentration of 1 mol/L

Negative and positive ∆G Consider the reaction: § If ∆G is a negative number --- Net loss of energy --- Reaction proceeds spontaneously --- A is converted to B --- Reaction is exergonic § If ∆G is a positive number --- Net gain of energy --- Reaction does not proceed spontaneously from B to A --- Reaction is endergonic --- Energy must be supplied to the reactants

§ e. g. Hydrolysis of ATP --- Exergonic reaction § Reversal of the reaction (ADP + Pi ATP) is endergonic and occurs only when there is a supply of energy of atleast 7. 3 cal/mol (∆G° is positive) § If ∆G = 0, reaction is in equilibrium --- Free energy of the forward reaction ( ) is equal in magnitude but opposite in sign to that of backward reaction ( )

§ ∆G is dependent on the actual concentration of reactants and products § At constant temperature and pressure, the following relation can be derived

∆G is an additive value for pathways § Biochemical pathways involve a series of reactions § Free energy change is an additive value for such reactions § The sum of ∆G determines whether a particular pathway will proceed or not. § If the sum of ∆G of individual reactions is negative, the pathway can operate. § This happens despite the fact that some of the individual reactions may have positive ∆G

Determination of ∆G from equilibrium constant § For a reaction, § The equation can be written as § At equilibrium, there is no net conversion of A to B. Hence, ∆G = 0 § Where, Keq = Equilibrium constant

§ By evaluating the constants we get, ü R = 1. 987 cal/mol-degree, ü 25°C = 298°T ü ln X = 2. 303 log 10 x § Substituting these values in previous equation, ∆G° = – (1. 987) (298) (2. 303) log 10 Keq = – 1363 log 10 Keq § If the concentration of both reactants and products at equilibrium can be measured, the Keq and in turn the ∆G° of the reaction can be calculated

Determination of ∆G from reduction potential § ∆G of a reaction which involves an oxidation-reduction process may be related to the difference in oxidation-reduction potentials (∆E°) of the reactants § Reducing agent – Substance that donates an electron and gets oxidized § Fe+++ is an oxidising agent – Accepts an electron and gets reduced

§ These reactions in which the electrons are indicated as being accepted or donated but the donor or acceptor have not been mentioned are called as half reactions § Various oxidation-reduction systems have certain reduction potential values

§ The relation between change in free energy and reduction potential is given as – ∆G = – n. F. ∆E° Where, § n = Number of electrons transferred in a redox reaction § F = Faraday’s constant = 23, 063 cal/v equiv § ∆E° = difference in reduction potential of oxidising and reducing agents i. e. (E° of half reaction containing oxidising agent) – (E° of half reaction containing reducing agent)

High energy compounds § Certain compounds are encountered in the biological system which, on hydrolysis, yield energy § High-energy compounds or energy rich compounds – substances which possess sufficient free energy to liberate at least 7 cal/mol at p. H 7. 0 § Low-energy compounds – Compounds which liberate less than 7. 0 cal/mol (lower than ATP hydrolysis to ADP + Pi)


Classification of high energy compounds
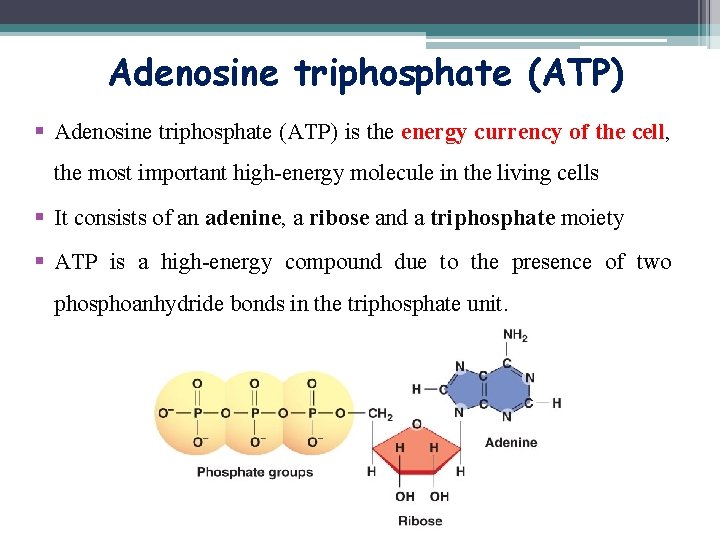
Adenosine triphosphate (ATP) § Adenosine triphosphate (ATP) is the energy currency of the cell, the most important high-energy molecule in the living cells § It consists of an adenine, a ribose and a triphosphate moiety § ATP is a high-energy compound due to the presence of two phosphoanhydride bonds in the triphosphate unit.
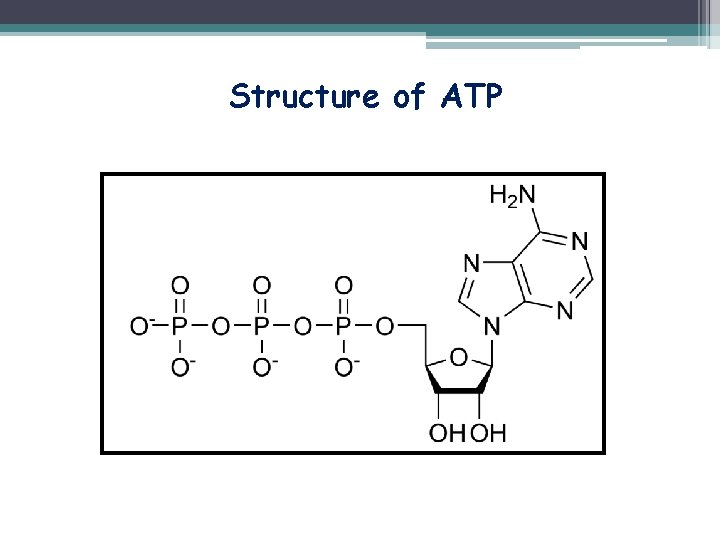
Structure of ATP
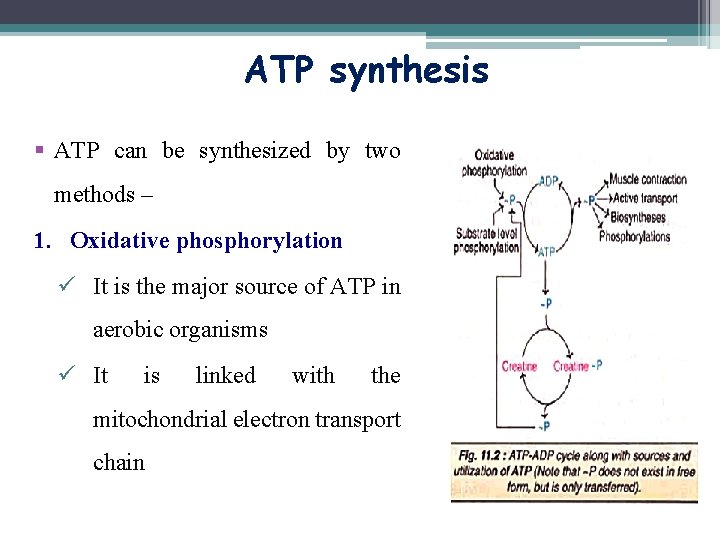
ATP synthesis § ATP can be synthesized by two methods – 1. Oxidative phosphorylation ü It is the major source of ATP in aerobic organisms ü It is linked with the mitochondrial electron transport chain

ATP synthesis 2. Substrate level phosphorylation ü ATP can be directly synthesized during substrate oxidation in the metabolism ü High-energy compounds like phosphoenolpyruvate and 1, 3 bisphoglycerate (intermediates of glycolysis) and succinyl Co. A (of citric acid cycle) can transfer high-energy phosphate to finally produce ATP

Biological significance of ATP § The hydrolysis of ATP is associated with the release of large amount of energy – ATP + H 2 O ADP + Pi + 7. 3 Cal The energy liberated is utilized for various processes like muscle contraction, active transport etc § ATP can also act as a donor of high-energy phosphate to lowenergy compounds, to make them energy rich ADP can accept high-energy phosphate from the compounds possessing higher free energy content to form ATP

§ ATP is required for synthesis of nucloside triphosphates § The cyclic phosphodiester c. AMP is formed from ATP in a reaction catalysed by adenylyl cyclase. c. AMP, which is a second messenger participates in different regulatory functions of the cell

Thank you
- Slides: 27