BIOCOMPATIBILITY OF DENTAL MATERIALS DR RAMASHANKER Associate professor

BIOCOMPATIBILITY OF DENTAL MATERIALS DR. RAMASHANKER Associate professor Deptt. of prosthodontic 3 rd nov 2014 time-10 -11 am

CONTENTS Ø Introduction Ø History Ø Definition Ø Requirements Ø Tests for evaluation Ø Allergic responses to dental materials Ø Materials considered for biocompatibility Ø Physical factors affecting pulp health Ø Summary Ø References
Introduction Ø Biocompatibility : - interaction between body & material Ø Body ↔ Material Ø Placement of material creates interface : dynamic Ø Interface activity depends on: - location of material - its duration in body - its properties - health of host 3

History Ø Mid 1800’ s dentists tried new materials for first time by directly putting them in patient’s mouth eg. Fox : fusible metal-bismuth, lead & tinmelted & poured in cavity preparation at appx. 100 o C Ø G. V. Black tried his new ideas of restorative materials, like early amalgams in patients’ mouth

Ø Concept of protecting patients- early 1960’s Ø Regulations & ethics introduced Ø Organisations like FDA, ANSI, ADA and ISO.

Definition Ø Being harmonious with life & not having toxic or injurious effects on biologic function. (G. P. T. 8 th edn. -2005) Ø Ability of the material to elicit an appropriate biological response in a given application in the body (Kenneth J. A).

Requirements for Dental Material Biocompatibility Ø Should not be harmful to pulp & soft tissues Ø Should not contain toxic diffusible substances Ø Should not produce allergic responses Ø Should not be carcinogenic 7

Biomaterial Any substance, other than a drug, that can be used for any period as a part of a system that treats, augments, or replaces any tissue, organ or function of the body. (G. P. T. 8 th edn. -2005) 8

Classification of Biomaterials from perspective of Biocompatibility Ø Those which contact soft tissues within the oral cavity eg. Acrylic resin Ø Those which could affect health or vitality of pulp eg. Liner, bases Ø Those which are used as root canal filling materials eg. Gutta percha 9

Classification of Biomaterials from perspective of Biocompatibility Ø Those which affect hard tissues of oral cavity eg. Implants Ø Those used in dental laboratory eg. Nickel, chromium, cobalt 10

ADVERSE EFFECTS FROM DENTAL MATERIALS • Classical biological reactions to materials are : TOXICITY INFLAMMATION ALLERGY MUTAGENICITY 11

TOXICITY Ø Earliest response studied Ø Earlier material containing LEAD posed a risk to patient due to toxic property of lead 12

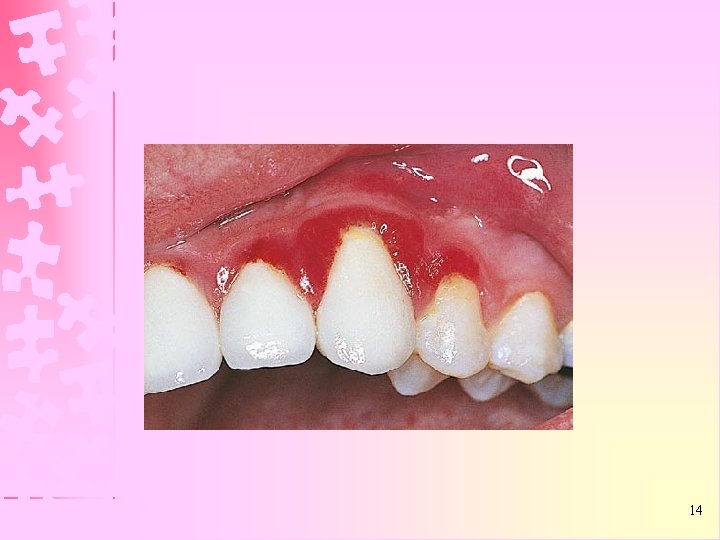
INFLAMMATION Ø Involves activation of the host immune system Ø Histologically it is characterized by edema of the tissue with infiltration of acute & chronic inflammatory cells 13
14

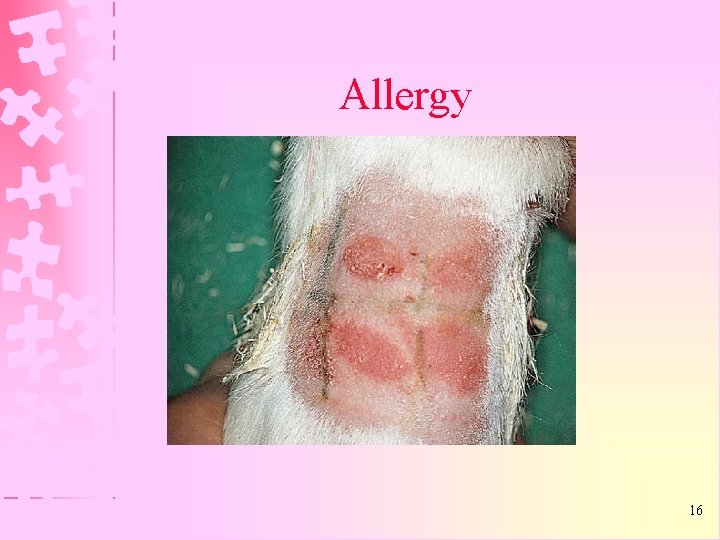
ALLERGY Ø Most common response that occurs when the body recognizes a material as foreign Ø Reactions involves all dimensions of immune system Ø An allergic reaction results histologically in an inflammatory response that can be difficult to differentiate between non allergic inflammation or low grade toxicity.
Allergy 16

Allergic Responses to Dental Materials Ø Allergic Contact Dermatitis Ø Allergic Contact Stomatitis Ø Allergy to Latex products
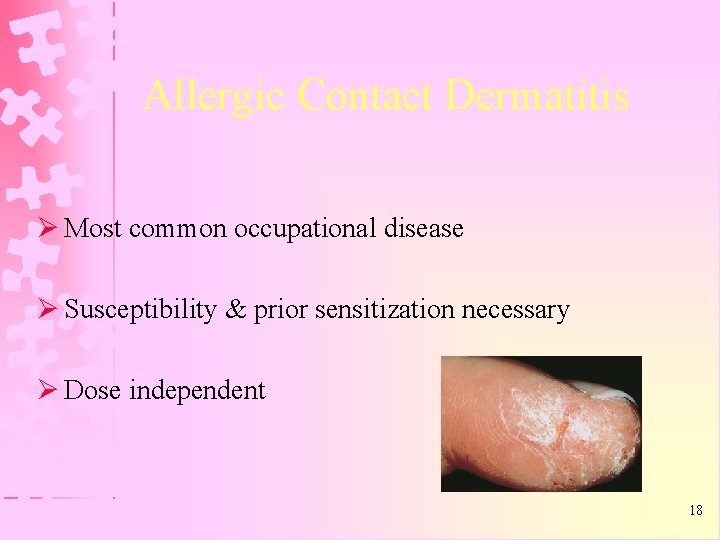
Allergic Contact Dermatitis Ø Most common occupational disease Ø Susceptibility & prior sensitization necessary Ø Dose independent 18
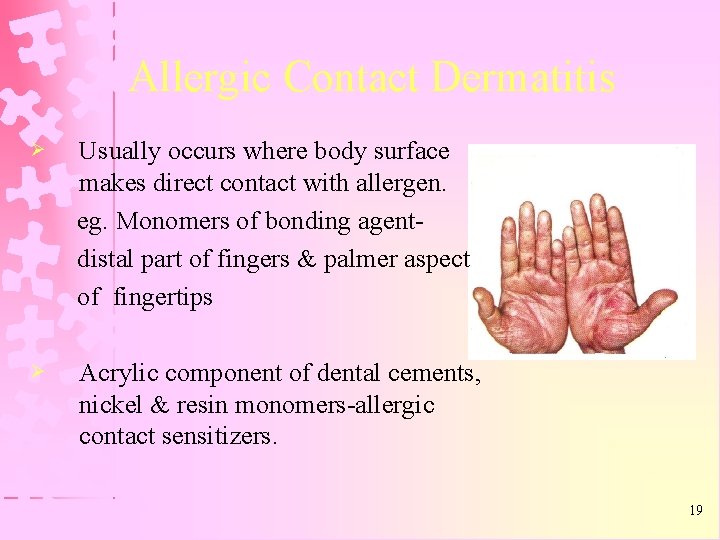
Allergic Contact Dermatitis Ø Usually occurs where body surface makes direct contact with allergen. eg. Monomers of bonding agentdistal part of fingers & palmer aspect of fingertips Ø Acrylic component of dental cements, nickel & resin monomers-allergic contact sensitizers. 19

Patch Test Ø Most definitive diagnostic test Ø Suspected allergen applied to skin to produce small area of allergic contact dermatitis After 48 to 96 hrs hyperemia, edema, vesicle formation & itching Positive reaction (Slavin and Ducomb, 1989) 20
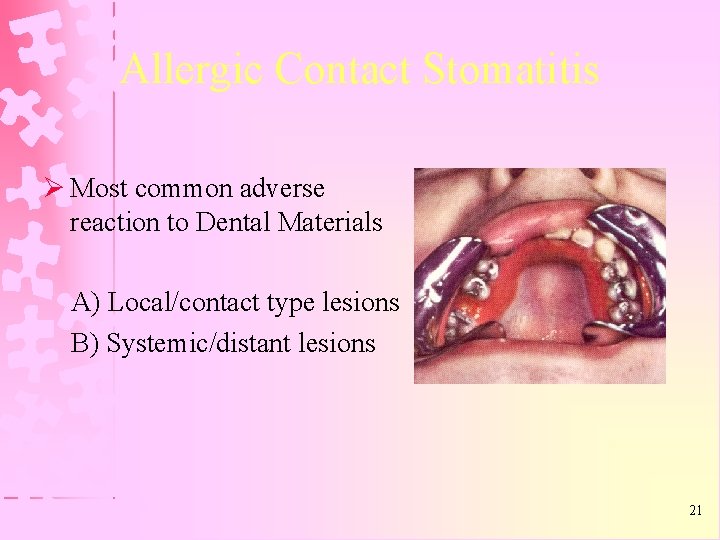
Allergic Contact Stomatitis Ø Most common adverse reaction to Dental Materials A) Local/contact type lesions B) Systemic/distant lesions 21

Allergic Contact Stomatitis Ø Common allergens : - chromium, cobalt, mercury, eugenol, components of resin based materials, & formaldehyde Ø Mouthwashes, dentifrices, lozenges, & cough drops cause burning, swelling & ulceration of oral tissues. Ø Lichenoid reactions : - Long-term effect in oral mucous membrane adjacent amalgam & composite resins. (Bratel and Johntell, 1994) 22
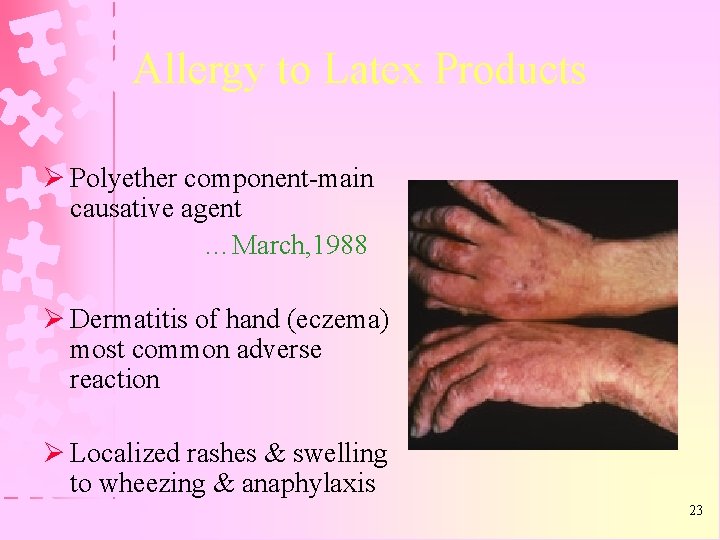
Allergy to Latex Products Ø Polyether component-main causative agent …March, 1988 Ø Dermatitis of hand (eczema) most common adverse reaction Ø Localized rashes & swelling to wheezing & anaphylaxis 23

Ø Repeated exposure & duration plays important role. Ø Most serious systemic reactions occur when gloves or rubber dam contact mucous membrane - generalized angioneurotic edema, chest pain, rash on neck or chest region and respiratory distress …Blinkhorn and Leggate, 1984 24

Prevention: Use Vinyl gloves or gloves made of other synthetic polymer gloves: Polythene gloves. Powder free gloves. Nitrile gloves. 25

MUTAGENIC REACTIONS • Mutagenicity results when the components of the material alter the base pair sequences of the DNA in cells • Dental materials or components such as nickel, copper, beryllium, some components of root canal sealers & resin based materials are mutagens

KEY PRINCIPLES THAT DETERMINE ADVERSE EFFECTS • Two key factors have paramount importance : Ø Ø Metal Corrosion or Metal degradation Surface Characterstics 27

CORROSION • Biocompatibility depends on degradation process • Biological response of corrosion products depends on: Amount Composition Form Location in tissues 28

CORROSION • Biological environment in contact also determines the corrosion property for eg: Ø salivary esterases accelerate breakdown of dental resins Ø Ingestion of acidic substances may alter corrosion of alloys or ceramics 29

SURAFCE CHARATERISTICS • Surface different from the Interior region • For eg: casting alloy sealant • EFFECTS OF SURFACE : Ti alloys promote osseointegration Rough surface promotes corrosion 30

Tests for Evaluation of Biocompatibility Aim Ø To eliminate any potential harm or damage to oral or maxillofacial tissues from a product or any component of a product Ø To modify or control the use by manufacturer & operator to prevent cytotoxicity. 31

Tests for Evaluation of Biocompatibility Ø Biocompatibility tests are classified on three levels (tiers) : 1. Group I : Primary tests 2. Group II : Secondary tests 3. Group III : Usage tests 32

Group I : Primary Tests Ø Advantages : § in vitro test, done in controlled experimental condition § Most rapid, economical & easily standardized § Large scale screening Ø Disadvantages : § Lack of relevance to in vivo use of material § Lack of immune, inflammatory & circulatory system 33

Cytotoxicity Tests Ø Material in a fresh or cured state → placed directly on tissue culture cells or on membranes overlying them. e. g. . Agar (Agar overlay technique) Barriers like dentin disks

Genotoxicity Tests Ø Determines carcinogenic/mutagenic potential Ø Carried out on mammalian or non-mammalian cells, bacteria, yeasts, or fungi. Ø Evaluates gene mutations, changes in chromosomal structure & other DNA or genetic changes caused by dental materials.

Genotoxicity Tests Ø Ames Test : -Material is tested with mutant histidine dependant bacteria -Agent is added to culture medium consisting salmonella typhimurium mutant gene which cannot produce histidine -If carcinogenic : - salmonella species reversed to original state, i. e. . start producing histidine again

Group II : Secondary Tests § § § Advantages : Intact biologic system to respond to a material Provide important bridge between in vitro environment & clinical use of material Disadvantages : More expensive & difficult to control Time consuming Ethical concerns

Group II : Secondary Tests 1. Systemic toxicity test : - Ø Material administered to test animals e. g. . Ratsorally or i. v. Ø If > 50% animals survive safe material is

Group II : Secondary Tests 2. Skin irritation test : Ø Irritation is inflammation without intervention of antibody or immune system. Ø Material held in contact with shaved skin of rats for 24 to 90 days Ø Erythema & edema are examined & confirmed.

Group II : Secondary Tests 3. Skin Sensitization test : Ø Sensitization is inflammatory response requiring participation of an antibody system specific for material allergen. Ø Done similar to irritation tests

Group II : Secondary Tests 4. Inhalation toxicity test : Ø Performed on rats, rabbits or guinea pigs in exposure chamber with aerosol preparations by releasing spray material around head & upper trunk of animals. Ø Death within 2 to 3 min. No death very toxic safe for human application

Group II : Secondary Tests 5. Implantation test : Ø Only used for testing implants & endodontic materials. Ø Material placed subcutaneously, intramuscularly, or as a bone implant at lateral cortex of femur or tibia or both Ø Histopathological examination has to be done Ø Observation period may be upto 1 year.
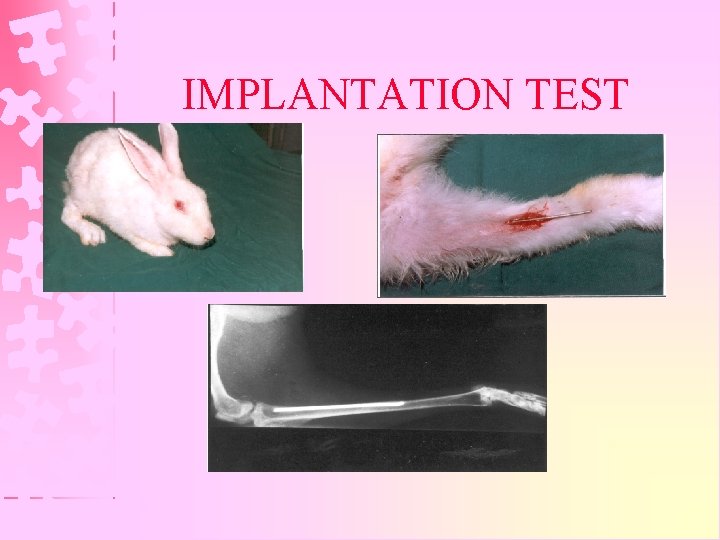
IMPLANTATION TEST

Group III : Usage Tests Advantage : § Material placed in an environment clinically relevant to its use in clinical practice Disadvantages : § Extremely complex & difficult to perform § Exceptionally expensive & very time consuming § Ethical concerns q In animals : usage tests q In humans : clinical trials 44
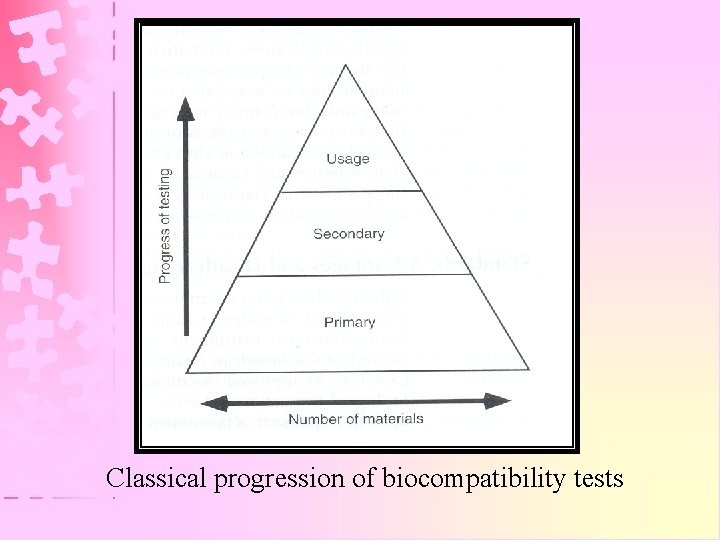
Classical progression of biocompatibility tests
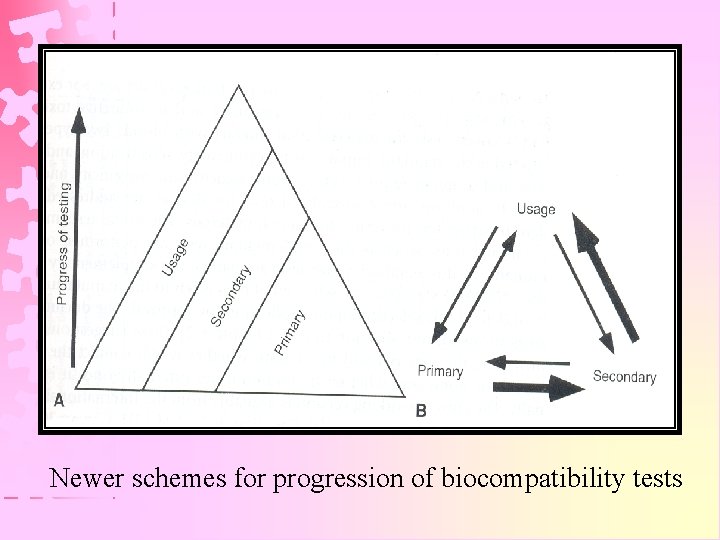
Newer schemes for progression of biocompatibility tests

VARIOUS DENTAL MATERIALS CONSIDERED FOR BIOCOMPATIBILITY

Ø Metals : Amalgam & mercury Nickel Beryllium Gold Ø Resins : Acrylic Resins Chemically cured composite resins Light cured composite resins

Ø Cements : Silicate cement Zinc Phosphate cement Glass ionomer cement Zinc oxide Eugenol cement Ø Miscellaneous : Impression materials Implant materials

Amalgam & Mercury Ø Mercury itself has no effect on pulp Ø Pulp response related to condensation pressure Ø Rate of diffusion into enamel & dentin-inversely related to degree of mineralization.

Mercury Ø Elemental mercury, Inorganic ion & Methyl mercury Ø Methyl mercury : -formed by biologic action of elemental mercury; Absorbed 100% in gut; Most toxic Ø Elemental mercury absorbed less than 0. 01% Ø 65% to 85% mercury vapor that is inhaled is retained in body

Mercury Levels in Blood Ø Subjects with amalgam restoration Ø Subjects without amalgam restoration Ø Lowest level at which earliest non-specific symptoms occur 0. 7 ng/m. L 0. 3 ng/m. L 35 ng/m. L

Mercury Hazard to Dental Personnel Ø Via inhalation & skin contact (allergic contact dermatitis) Ø Accidental spillage Ø Handling with bare fingers Ø Improper storage Ø Improper retrieval of spilled mercury or waste amalgam Ø Faulty equipment

Ø Acute mercury poisoning : Rare; stomatitis & diarrhoea Ø Chronic mercury poisoning : Weakness, fatigue, anorexia, wt. loss, insomnia, irritability, shyness, dizziness & tremors in extremities. Ø Methyl mercury poisoning : Paresthesia of extremities, lips & tongue; ataxia (gait disturbance), & concentric constriction of visual fields (Tunnel Vision)

Recommendations in Mercury Hygiene 1. 2. 3. 4. 5. 6. 7. Store in unbreakable tightly sealed containers Clean-up spilled mercury immediately Do not handle with bare hands Salvage all amalgam scrap & store it under water Use water spray & suction while grinding Do not use ultrasonic condensers Periodic mercury vapor level determination in clinic 8. Alert health personnel about hazards of mercury 9. Use of rubber dam 10. Provide adequate ventilation
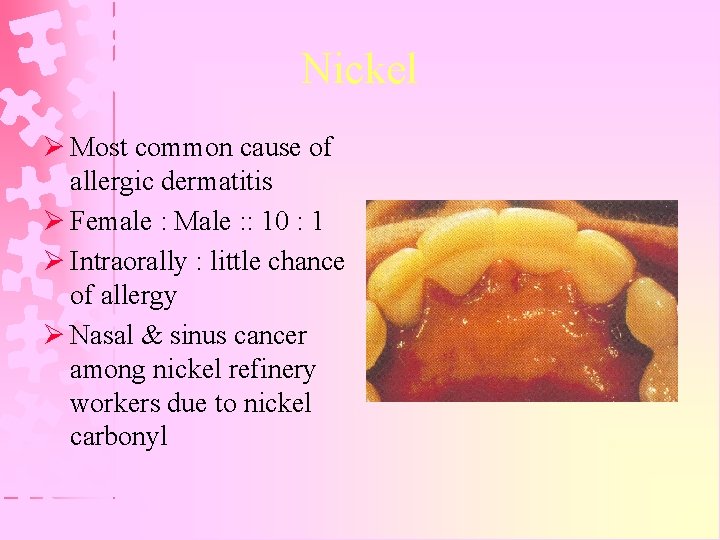
Nickel Ø Most common cause of allergic dermatitis Ø Female : Male : : 10 : 1 Ø Intraorally : little chance of allergy Ø Nasal & sinus cancer among nickel refinery workers due to nickel carbonyl

Beryllium Ø Component of base metal alloys Ø Highest risk to dental technicians during melting & trimming of alloy Ø Berylliosis : inflammatory lung disease due to inhalation of beryllium dust or fumes

Beryllium Ø Prevention : • Confirm allergy by Patch test • Avoid base metal restorations in patients with known allergy • Good ventilation & exhaust

Gold Ø Pure gold is inert Ø Allergy to gold is very rare (1 in 1 million)
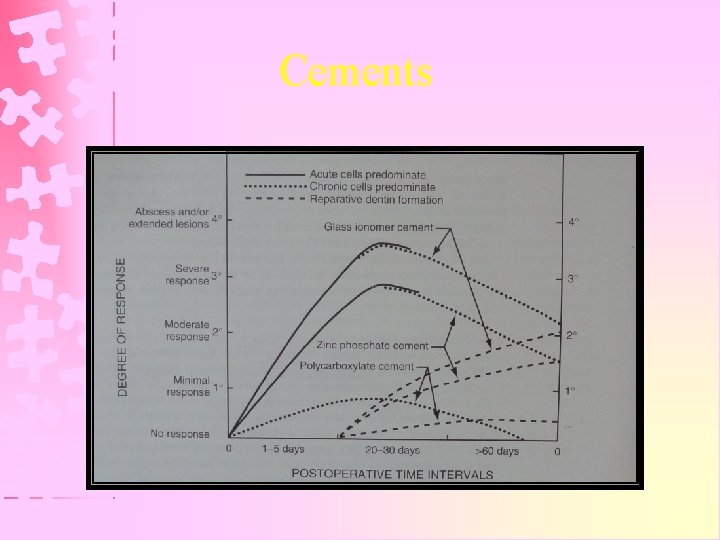
Cements
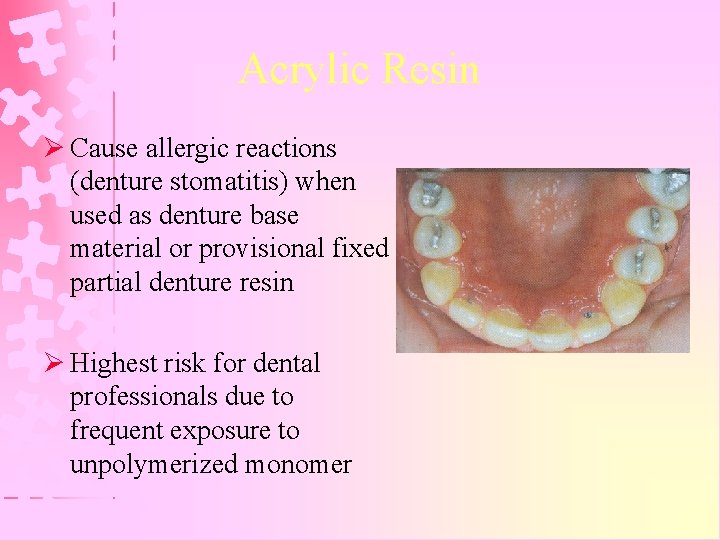
Acrylic Resin Ø Cause allergic reactions (denture stomatitis) when used as denture base material or provisional fixed partial denture resin Ø Highest risk for dental professionals due to frequent exposure to unpolymerized monomer

Chemically Cured Resin Composites Ø Require use of matrix pressure to enhance adaptation to cavity walls : Potential pulp irritant Ø Chronic pulpitis : persists for indefinite period, after 2 to 3 weeks, develop massive pulp lesion Ø Thin coating of hard setting Ca(OH)2 cement recommended for deep cavities

Light-Cured Resin Composites Ø Visible light cured systems : greater depth of cure, shorter curing time, less porosity & more wear-resistant restorations than UV light cured systems less pulp response Ø Use twice the recommended time exposure to light Ø Conservative cavity preparation & incremental curing; no need for matrices & pressure, to gain adaptation less toxicity to pulp

Impression Materials Ø Irreversible hydrocolloids : - Inhaling fine airborne particles (dust) can cause silicosis & pulmonary hypersensitivity. Dustless/Dustfree alginate is preferred Ø Elastomers : - Cellular toxicity levels Polyether > Addition Silicone > Polysulphide

Implant Materials Ø Commercially pure Titanium & its alloys are the most biocompatible restorative materials Ø Bio-glass ceramics used as implant materials also exhibit good biocompatibility
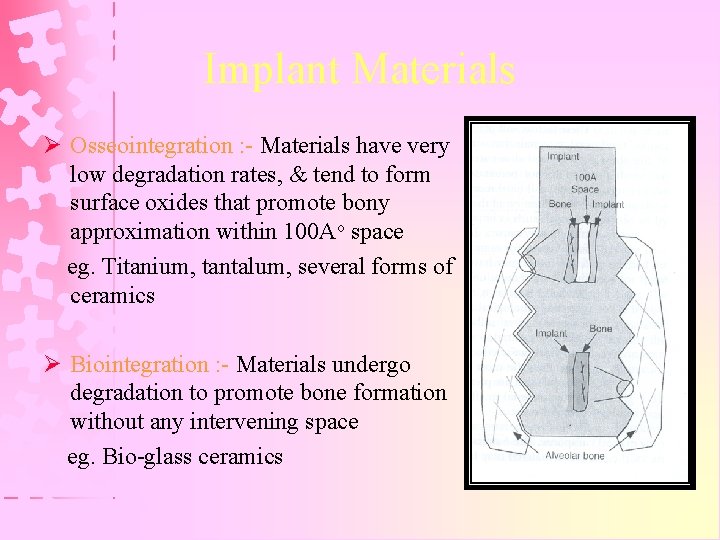
Implant Materials Ø Osseointegration : - Materials have very low degradation rates, & tend to form surface oxides that promote bony approximation within 100 Ao space eg. Titanium, tantalum, several forms of ceramics Ø Biointegration : - Materials undergo degradation to promote bone formation without any intervening space eg. Bio-glass ceramics

PHYSICAL FACTORS AFFECTING PULP HEALTH

Microleakage Ø Free penetration of fluids, micro-organisms & oral debris along interface between restoration & tooth, progressing down the walls of cavity preparation Ø It can result in : 1. Secondary/Recurrent caries acute/chronic pulpitis, pulp abscess, etc. 2. Staining or discoloration 3. Sensitivity due to continuing Pulpal irritation
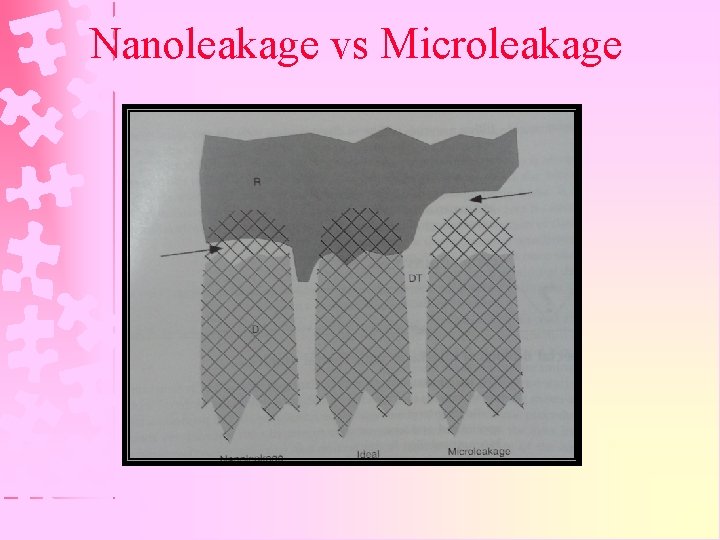
Nanoleakage vs Microleakage

Prevention: 1. Use bonding/adhesive techniques for better adaptation of restoration to tooth surface 2. Regular monitoring of restoration 3. Use cavity varnish below amalgam restoration (leakage space filled by corrosion products thereby sealing cavity : but requires much time)

Thermal Changes Ø Temperature fluctuations in oral cavity may crack restorative material or produce undesirable dimensional changes Microleakage Ø Thermal conductivity & coefficient of thermal expansion Ø Metals are good conductors of heat, causing sensitivity with large metallic restorations eg. Amalgam or gold inlays Provide suitable base
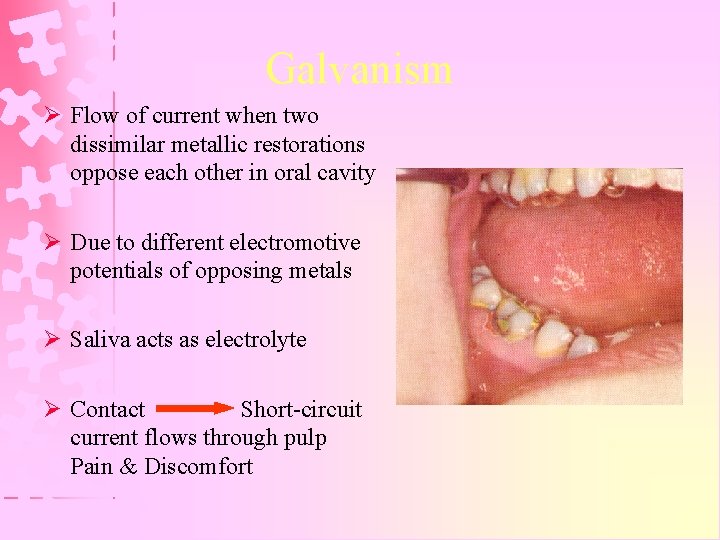
Galvanism Ø Flow of current when two dissimilar metallic restorations oppose each other in oral cavity Ø Due to different electromotive potentials of opposing metals Ø Saliva acts as electrolyte Ø Contact Short-circuit current flows through pulp Pain & Discomfort

Galvanism Ø Current falls off if fillings are maintained in contact due to polarization of cell Ø Pain perception depends on patient sensitivity rather than magnitude of current Ø Magnitude of current depends on composition & surface area of metals eg. Alloy of stainless steel develop higher current density than gold or cobalt-chromium alloys when in contact with amalgam

Galvanism Ø As size of cathode (eg. gold alloy) increase relative to anode (eg. Amalgam), current density increases Ø Larger cathode enhances corrosion of smaller anode Ø Current density in non-gamma 2 containing amalgam is less than gamma 2 containing amalgam Ø Prevention : Placement of insulating base Applying varnish on cavity walls Proper planning of restoration

Estrogenicity • Ability of a chemical to act in the body in a manner similar to that of an estrogen. • Bisphenol A –xenoestrogen may act on estrogenic receptors in cells. • E-screen assay –relies on growth response of breast cancer cells that are estrogen sensitive.

Summary Ø Clinical Guidelines for selecting biocompatible materials : q Define the use of material q Define how the material has been tested q Think in terms of Risk & Benefit
Conclusion Benefits Clinical Judgement Risks

References: 1) Philips’ Science of Dental Materials - Kenneth J. Anusavice 2) Dental Material Sciences - Combe 3) Dental Materials – Properties & Manipulation - Craig 4) Color Atlas of Oral Pathology – 4 th Edition - Robinson & Miller 5) Essentials of Oral Pathology & Oral Medicine - Cawson & Odell

Thank You For a Patient Listening
- Slides: 80