BIOCHEMISTRY The Chemistry of Life PACKET 2 NFL

BIOCHEMISTRY (The Chemistry of Life) PACKET 2 NFL Video Link
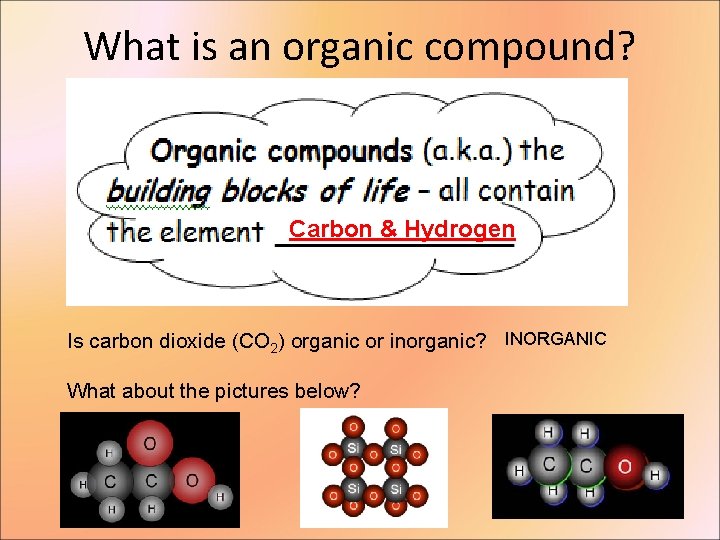
What is an organic compound? Carbon & Hydrogen Is carbon dioxide (CO 2) organic or inorganic? INORGANIC What about the pictures below?

Organization of Life Atoms (Carbon or Oxygen) __________ (carbon dioxide, water) MOLECULES ___________ (carbs) MACROMOLECULES ___________ (chloroplast) ORGANELLES ___________ (plant/animal) CELLS TISSUES ___________ (muscle) ___________ (stomach) ORGANS ___________ (digestive) ORGAN SYSTEMS ___________ (person) ORGANISM
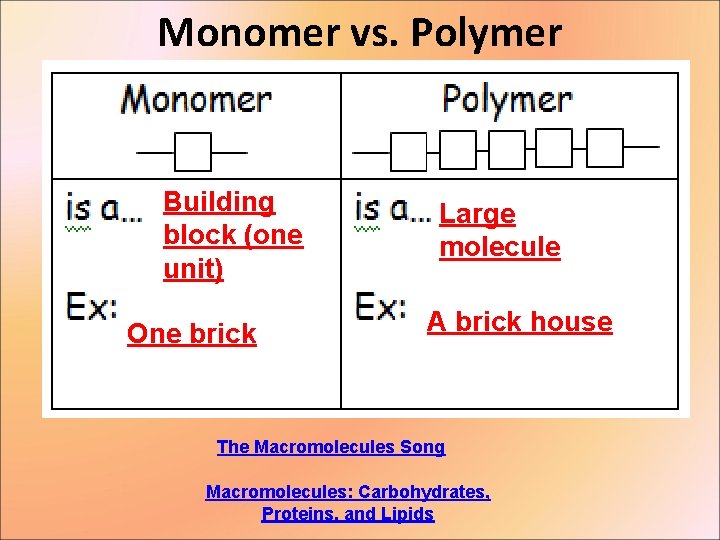
Monomer vs. Polymer Building block (one unit) One brick Large molecule A brick house The Macromolecules Song Macromolecules: Carbohydrates, Proteins, and Lipids

The p. H Scale: Neutral 0 --------------------- 7. 0 -------------------- 14 STRONG acid WEAK base STRONG base • BUFFERS __________ help maintain PH homeostasis by minimizing changes in ____. Most body systems function optimally at a p. H of 7. 4 near _______. As the p. H changes ENZYMES __________ may stop working, nerve and muscle activity weakens, and finally all chemical reactions are affected.

Enzymes • PROTEIN A special type of ____________ • SPEED UP Function - to _______________ a chemical reaction by LOWERING ____________ the energy needed to start the reaction ACTIVATION ENERGY (_____________________) • Enzyme Animation Link SPECIFIC Enzymes are __________ since they only work on one type of substrate. • REUSABLE Enzymes are __________ since they can be used over and over. • TEMP PH Enzymes are affected by ________ and ___________. SHAPE Their _________ changes so they can no longer fit with the substrate. • PHOTOSYNTHESIS Equation for a chemical reaction (_______________): ENZYME _____ 6 CO 2 + 6 H 2 O + light • REACTANTS __________ C 6 H 12 O 6 + 6 O 2 PRODUCTS _________
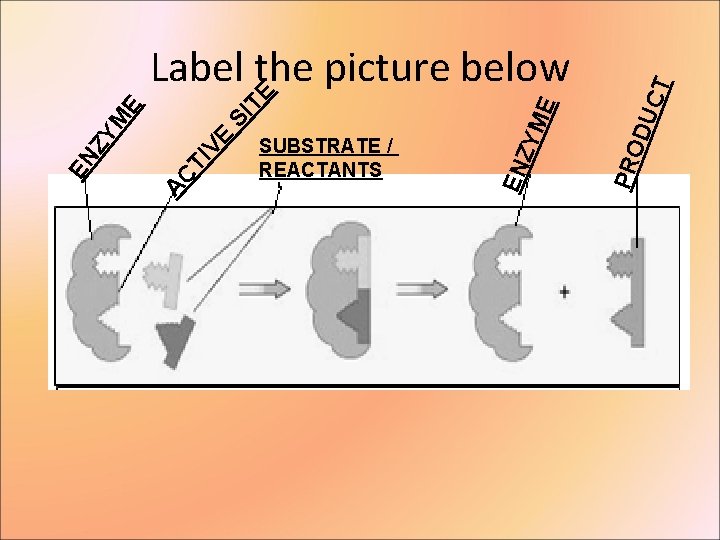
CT DU PRO E ZYM SUBSTRATE / REACTANTS EN TI VE AC EN ZY M E SI TE Label the picture below
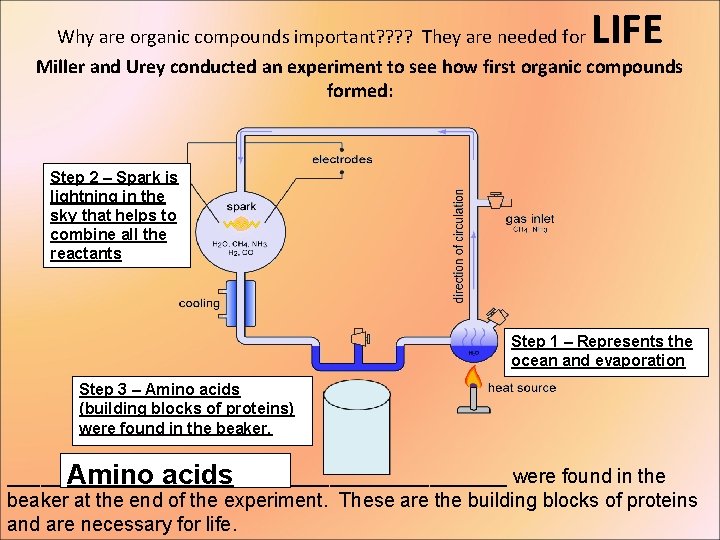
Why are organic compounds important? ? They are needed for LIFE Miller and Urey conducted an experiment to see how first organic compounds formed: Step 2 – Spark is lightning in the sky that helps to combine all the reactants Step 1 – Represents the ocean and evaporation Step 3 – Amino acids (building blocks of proteins) were found in the beaker. __________ Amino acids_____________ were found in the beaker at the end of the experiment. These are the building blocks of proteins and are necessary for life.
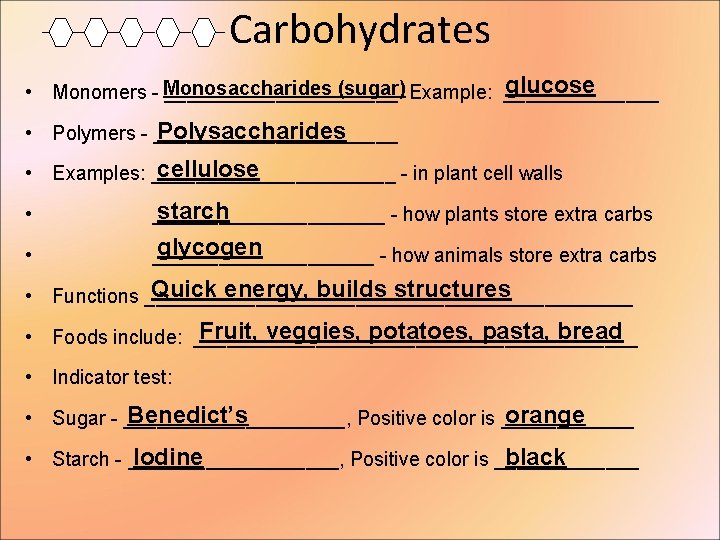
Carbohydrates glucose (sugar) Example: _______ • Monomers - Monosaccharides ___________ Polysaccharides • Polymers - ___________ cellulose • Examples: ___________ - in plant cell walls • starch ___________ - how plants store extra carbs • glycogen __________ - how animals store extra carbs Quick energy, builds structures • Functions ______________________ Fruit, veggies, potatoes, pasta, bread • Foods include: ____________________ • Indicator test: Benedict’s orange • Sugar - __________, Positive color is ______ Iodine black • Starch - __________, Positive color is _______

Lipids • Monomers - Fatty ___________ acids & glycerol Fats, Oils & Waxes • Polymers - ___________ steroids Examples: ___________ - such as cholesterol that makes hormones and parts of cells phospholipids ___________ - make up the cell membrane Long-term energy, insulation, water-proofing • Functions ______________________ • Foods include: ____________________ Butter, mayo, oil, salad dressing Brown paper towel Positive color is • Indicator test: ___________, Grease stain a ____________ triglyceride
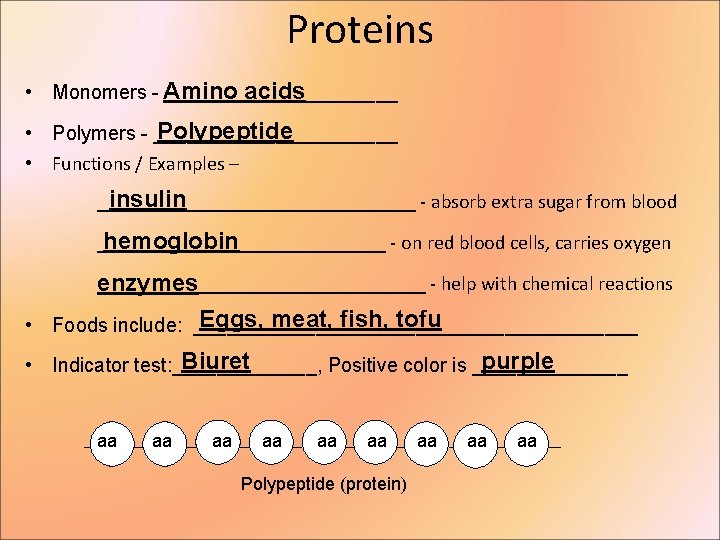
Proteins • Monomers - Amino ___________ acids Polypeptide • Polymers - ___________ • Functions / Examples – insulin ________________ - absorb extra sugar from blood _______________ - on red blood cells, carries oxygen hemoglobin _________________ - help with chemical reactions enzymes Eggs, meat, fish, tofu • Foods include: ____________________ Biuret purple • Indicator test: _______, Positive color is _______ aa aa aa Polypeptide (protein) aa aa aa
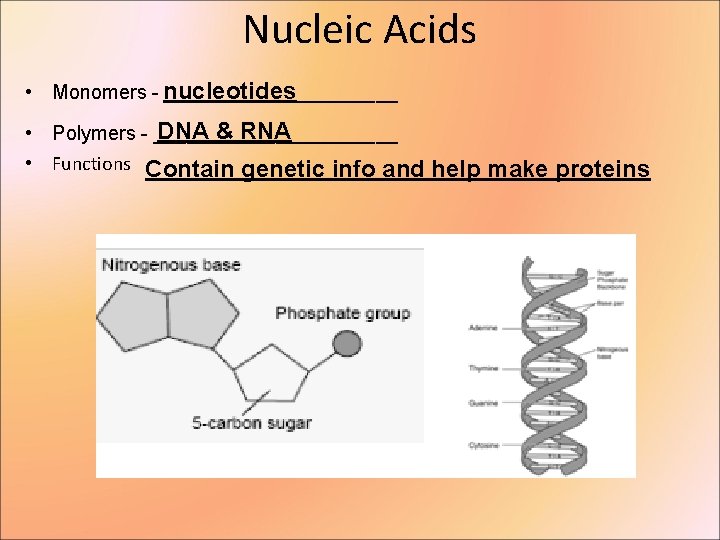
Nucleic Acids • Monomers - nucleotides ___________ DNA & RNA • Polymers - ___________ • Functions Contain genetic info and help make proteins
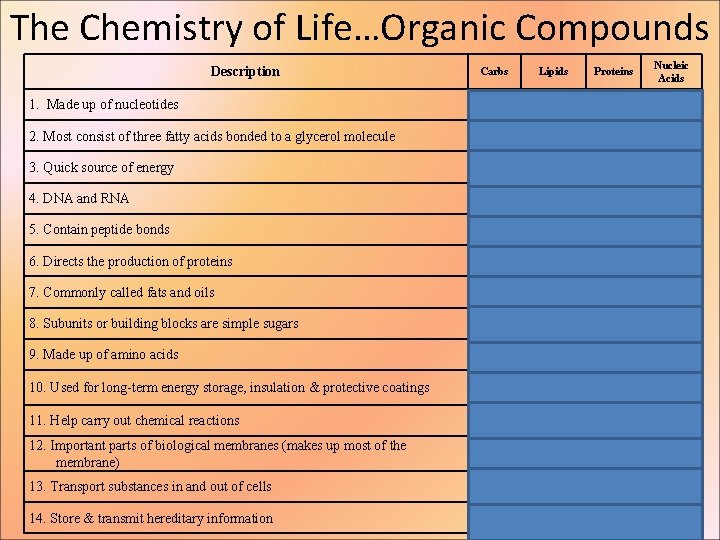
The Chemistry of Life…Organic Compounds Description Carbs Lipids Proteins X 1. Made up of nucleotides X 2. Most consist of three fatty acids bonded to a glycerol molecule 3. Quick source of energy X X 4. DNA and RNA X 5. Contain peptide bonds X 6. Directs the production of proteins X 7. Commonly called fats and oils 8. Subunits or building blocks are simple sugars X X 9. Made up of amino acids 10. Used for long-term energy storage, insulation & protective coatings X X 11. Help carry out chemical reactions 12. Important parts of biological membranes (makes up most of the membrane) 13. Transport substances in and out of cells 14. Store & transmit hereditary information Nucleic Acids X X X

STARCH 15. ______is how plants store excess sugar and animals store excess sugar as. GLYCOGEN ______. Both are complex carbs. 16. This is a protein in red blood cells HEMOGLOBIN _________. 17. A protein that can change the rate of a reaction is an ENZYME _______. 18. Triglyceride is lipid made up of a glycerol molecule and 3 (#) FATTY ACIDS ______________. 19. The monomers that make up nucleic acids are known as NUCLEOTIDES __________ DNA & RNA 20. The two basic kinds of nucleic acids are _______ 21. If you see a word end in –ose (Ex: glucose, sucrose) then SUGAR think ______. If the word ends in –ase (Ex: cellulase, ENZYME amylase) then think ______.
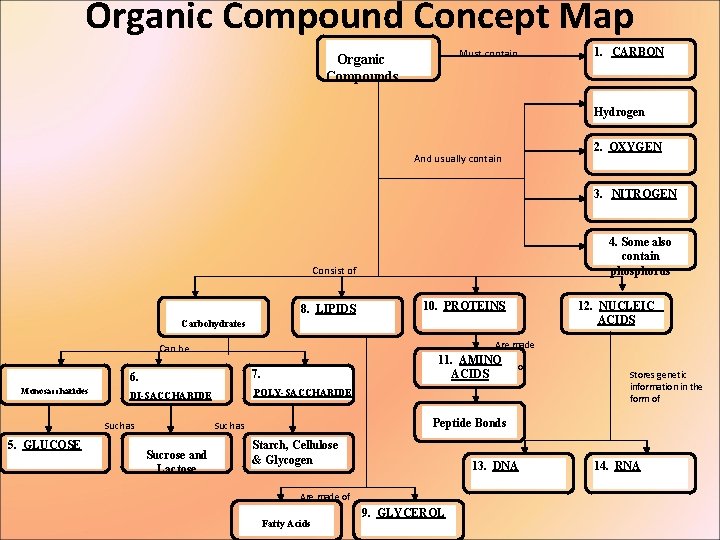
Organic Compound Concept Map 1. CARBON Must contain Organic Compounds Hydrogen 2. OXYGEN And usually contain 3. NITROGEN 4. Some also contain phosphorus Consist of 8. LIPIDS 10. PROTEINS 12. NUCLEIC ACIDS Carbohydrates Are made Can be Monosaccharides 6. DI-SACCHARIDE POLY-SACCHARIDE Such as 5. GLUCOSE 11. AMINO ACIDS 7. Stores genetic information in the form of Peptide Bonds Such as Sucrose and Lactose of Starch, Cellulose & Glycogen 13. DNA Are made of Fatty Acids 9. GLYCEROL 14. RNA
- Slides: 15