Biochemistry The Chemistry of Life Is chemistry really

Biochemistry The Chemistry of Life

Is chemistry really that important to learn about? • Of course it is!!! • Chemicals make up EVERYTHING in our bodies, as well as everything we eat! • Chemicals are Ev. ERYw. HEr. E!!! • 4 important types of organic molecules are: Carbohydrates, Lipids, Proteins and Nucleic Acids

But first…. a little review • Elements: simplest form of a substance - cannot be broken down any further without changing what it is • Atom: the actual basic unit composed of protons, neutrons, and electrons

Atomic structure • Just like cells are the • • basic unit of life, the ATOM is the basic unit of matter. They are very small. If placed side by side one million would stretch a distance of 1 cm. The atom is made up of 3 particles. Particle Charge PROTON + NEUTRON NEUTRAL ELECTRON -

Isotopes • atoms of the same element that HAVE A • • DIFFERENT NUMBER OF NEUTRONS Some isotopes are radioactive. This means that their nuclei is unstable and will break down at a CONSTANT RATE over time. There are several practical uses for radioactive isotopes: 1. CARBON DATING 2. TRACERS 3. KILL BACTERIA / CANCER CELLS

Ionic Bonds • Occur when 1 or more electrons are • • • TRANSFERRED from one atom to another. When an atom loses an electron it is a POSITIVE charge. When an atom gains an electron it is a NEGATIVE charge These newly charged atoms are now called IONS – Example: Na. Cl (SALT)

Covalent bonds • Occur when electrons are SHARED by atoms. • These new structures that result from covalent bonds are called MOLECULES • ** In general, the more chemical bonds a molecule has the more energy it contains

Water—The Mysterious Molecule • Because water (H 20) is a polar molecule, it has many unique properties. • Slightly +Hydrogens are attracted to slightly –Oxygens • These attractions are called hydrogen bonds and are the reason water: – is the only substance that is less dense as a solid – Has a high specific heat/heat capacity – Is attracted to itself (cohesion) and other substances (adhesion)

p. H scale • In water, some H 20 breaks apart • • into H+ and OHIf the amount of each ion is equal, the solution has a p. H of 7 The more H+, the more acidic the solution – 0 – 6 acid – 6 -7 neutral – 8 -14 base

Carbon chemistry • 4 valence electrons, • Can form 4 bonds • Readily bonds with other carbons • Can form long carbon chains, or carbon rings • Can form single, double or even triple bonds

Molecules of Life • Carbohydrates (CHO) • Sugars and starches • Lipids (CHO) • Fats, oils, waxes and steroids • Proteins (CHONS) • Amino acids and enzymes • Nucleic Acids (CHONP) • DNA and RNA Polymer: Large molecule made up of repeating subunits Monomer: smaller molecules that make up polymers
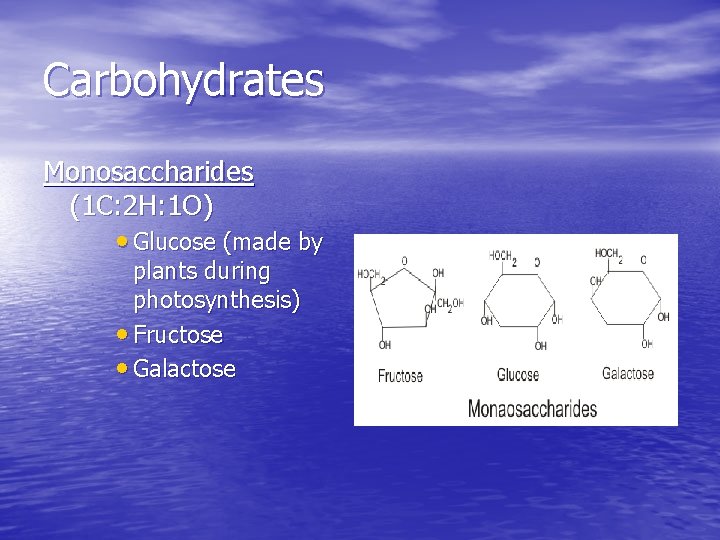
Carbohydrates Monosaccharides (1 C: 2 H: 1 O) • Glucose (made by plants during photosynthesis) • Fructose • Galactose
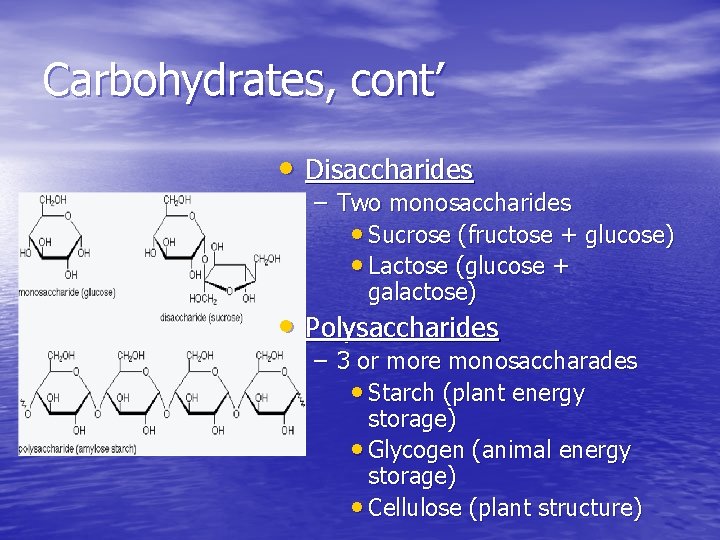
Carbohydrates, cont’ • Disaccharides – Two monosaccharides • Sucrose (fructose + glucose) • Lactose (glucose + galactose) • Polysaccharides – 3 or more monosaccharades • Starch (plant energy storage) • Glycogen (animal energy storage) • Cellulose (plant structure)

Lipids • More C, less H and O • Insoluble in water • Used for long term energy storage and insulation • Fatty Acids, Triglycerides, Waxes and Steroids

Lipids, cont’ • Fatty Acids – Make up most lipids – Long hydrocarbon chain with a carboxyl group (COOH) on one end – Carboxyl group is polar (hydrophilic) – Hydrocarbon chain is nonpolar (hydrophobic) – In a cell membrane, fatty acids are arranged in a double layer, with chains pointing towards eachother

Saturated fats vs Unsaturated fats • Saturated fats Hydrocarbon chain is ‘saturated’ with hydrogens (all single bonds) – Solid at room temperature – Usually animal based –

• Unsaturated Fats – Contain one or more double bonds – Liquid at room temperature – Usually plant based

Proteins • Contain H, O, C and Nitrogen • Main component of our body tissue • Made up of chains of amino acids – Amino acids = monomers – Polypeptide/protein = polymer
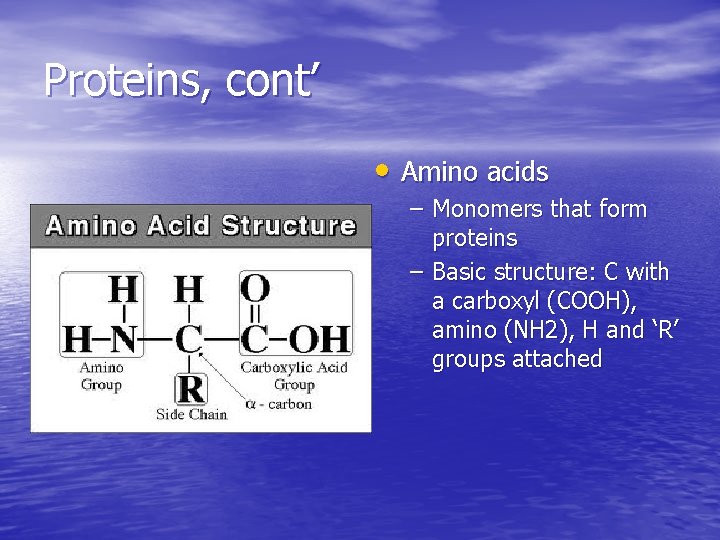
Proteins, cont’ • Amino acids – Monomers that form proteins – Basic structure: C with a carboxyl (COOH), amino (NH 2), H and ‘R’ groups attached

Proteins, cont’ • Enzymes – – – Specific proteins that speed up chemical reactions Not affected by the reaction “Induced fit” model (used to be called ‘lock and key’)

Nucleic Acids • Complex polymers made of monomers called nucleotides • Store important information in the cell – Deoxyribonucleic acid (DNA) • Stores most of the information necessary for cell function – Ribonucleic acid (RNA) • Stores and transfers information for making proteins

Vocabulary check… • Macromolecule • Polymer • Carbohydrate • Lipid • Protein • Amino acid • Nucleic acid • Nucleotide

FUNCTIONAL GROUPS • Functional groups are structural building blocks that determine the characteristics of a compound. • One functional group important to living things is the hydroxyl, represented by --OH. (Table 3 -1) • An alcohol is an organic compound with a Hydroxyl Group attached to one of its Carbon Atoms. • The hydroxyl group makes alcohol a polar molecule, which means it is soluble in water • Other functional groups important to living things are shown in Table 3 -1.

A little more about enzymes… • Most of the reactions that take place in our body require enzymes.


**EXTRA CREDIT OPPORTUNITY** • Speaking of fatty acids… • Find out: – What trans-fatty acids are (trans fats) – How they are formed – Why they are not good for us – What kind of foods have them – Turn in by Friday for 5 EC points
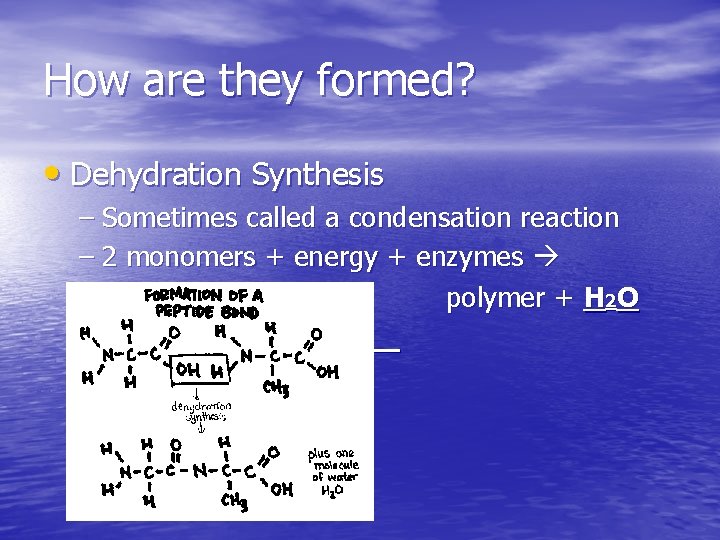
How are they formed? • Dehydration Synthesis – Sometimes called a condensation reaction – 2 monomers + energy + enzymes polymer + H 2 O

How are they broken down? • Hydrolysis reaction – Reverse of a dehydration synthesis reaction – Hydro = water – Lysis = breaking apart – Requires enzymes – Releases energy (heat)

Lipids, cont’ • Triglycerides – Three fatty acid chains joined to one molecule of glycerol – Either oil or fat depending on state at room temperature

Lipids, Cont’ • Waxes – Long fatty acid chain joined to a long alcohol chain – Highly waterproof – Protective barrier in plants and animals (earwax)

Lipids, cont’ • Steroids – Unlike other lipids, composed of 4 carbon rings – Considered lipids because they are insoluble in water – Ex. Hormones, nerve tissues, venoms, plant poisons
- Slides: 31