Biochemistry THE CHEMISTRY OF LIFE Chemistry I thought

Biochemistry THE CHEMISTRY OF LIFE

Chemistry? I thought this was Biology! Chemistry is the central science. To understand life, we must understand chemistry. Examples: All cells are made of molecules, and molecules are made of atoms Animals breathe in oxygen and eat sugar, and this creates energy for them Plants “breathe” in carbon dioxide and absorb water, and they make oxygen and sugar DNA has the genes of all living things Chemicals act as “messengers” in living organisms. Water is the molecule of life Etc. etc. !

PART I: Atoms & Elements
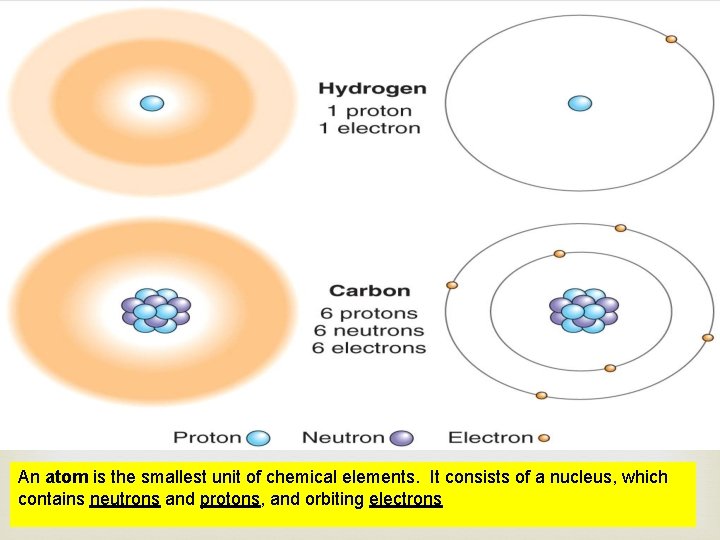
An atom is the smallest unit of chemical elements. It consists of a nucleus, which contains neutrons and protons, and orbiting electrons
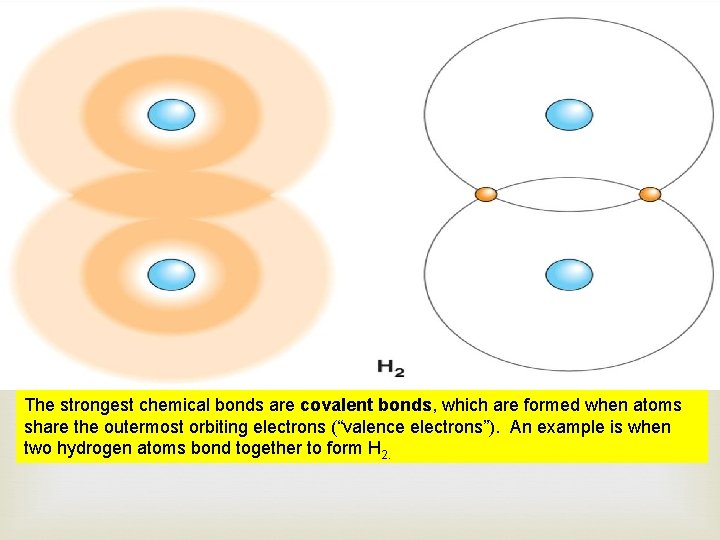
The strongest chemical bonds are covalent bonds, which are formed when atoms share the outermost orbiting electrons (“valence electrons”). An example is when two hydrogen atoms bond together to form H 2.

“Ionic bonds” are those that are formed between atoms when one or more valence electrons are completely transferred from on atom to another. A good example is table salt (Na. Cl).
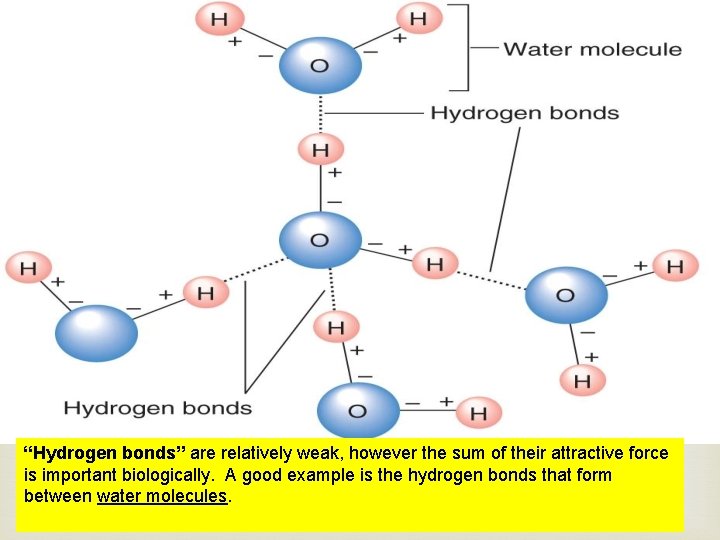
“Hydrogen bonds” are relatively weak, however the sum of their attractive force is important biologically. A good example is the hydrogen bonds that form between water molecules.

Atoms & Elements There are 118 “elements” Fe (Iron), Au (gold), H (hydrogen), O (oxygen), etc. Element = a pure substance that consists of only 1 kind of atom Cannot be broken down by chemical reactions Atom – comes from Greek word atomos, means “unable to be cut. ” Is this true? Placed side by side, 100 million atoms would make a row only the width of your little finger.

Subatomic Particles Atoms are made up of A nucleus, which has Protons – positive charge Neutrons – zero charge (neutral) Electrons – negative charge Spin around nucleus Has a mass that is 1/1840 that of a proton

Atom Vocabulary Isotopes = an atom with a different number of neutrons Radioactive isotopes = isotopes with nuclei that are unstable Can be dangerous But are important to science Compound = 2 or more elements in certain proportions H 2 O, CH 4, C 6 H 12 O 6 Molecule = 2 or more atoms in certain proportions H 2, O 2, … and H 2 O, CH 4, C 6 H 12 O 6

What elements are in cells? All life on earth is made up of the same 25 chemical elements

What elements are in cells? “HONC” Hydrogen (H) Oxygen (O) Nitrogen (N) Carbon (C) Make up 96% of the mass of every organism on Earth.

What elements are in cells? Calcium (Ca) Phosphorus (P) Potassium (K) Sulfur (S) Sodium (Na) Chlorine (Cl) Magnesium (Mg) These make up most of the remaining 4%


PART II: Macromolecules
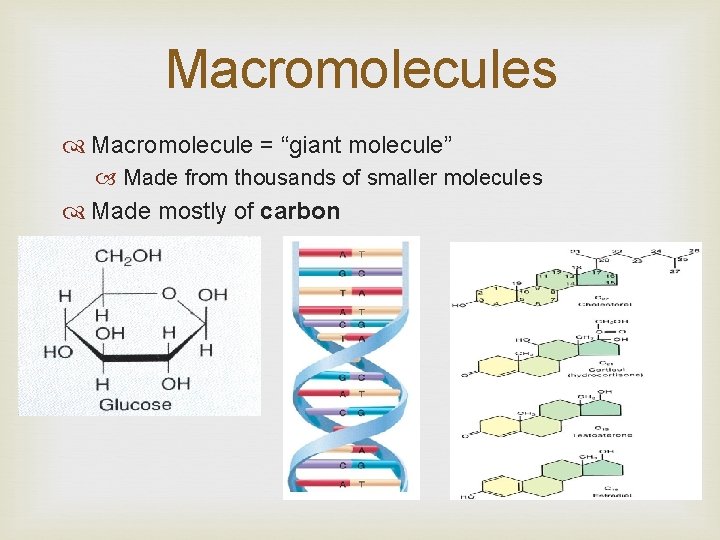
Macromolecules Macromolecule = “giant molecule” Made from thousands of smaller molecules Made mostly of carbon

Four major groups Carbohydrates Lipids Nucleic Acids Proteins These are the BIOMOLECULES

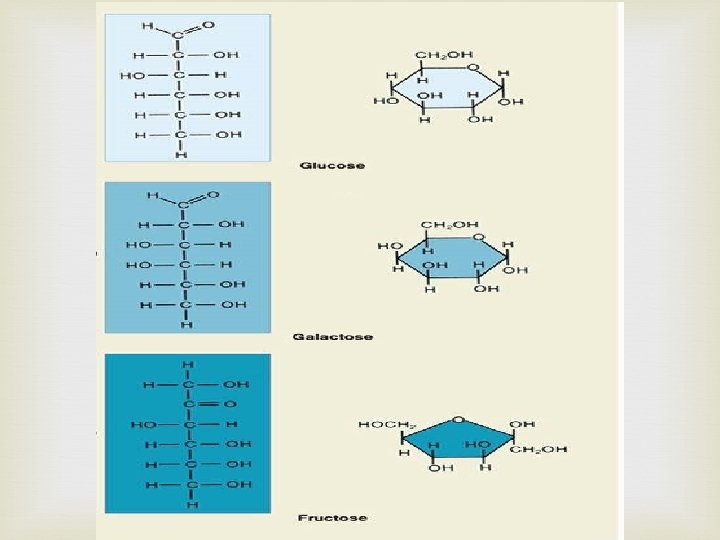
Carbohydrates Made of C, H, and O Usually a 1: 2: 1 ratio Used by living things as their main source of energy Plants and some animals use them for structure Also called “sugars” Two types Monosaccharides – single sugar molecules Polysaccharides – a chain of monosaccharides Monosaccharide Glucose Galactose Fructose Polysaccharide Glycogen Starch Cellulose


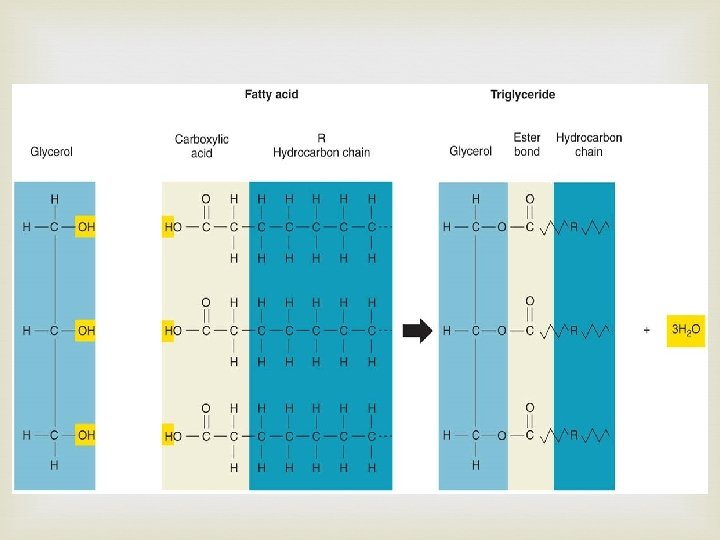
Lipids Made mostly of C and H Types Fats Oils Waxes Used to store energy Important parts of biological membranes and waterproof coverings Steroids are lipids also – some are chemical messengers Many are formed when a glycerol molecule joins with fatty acids.

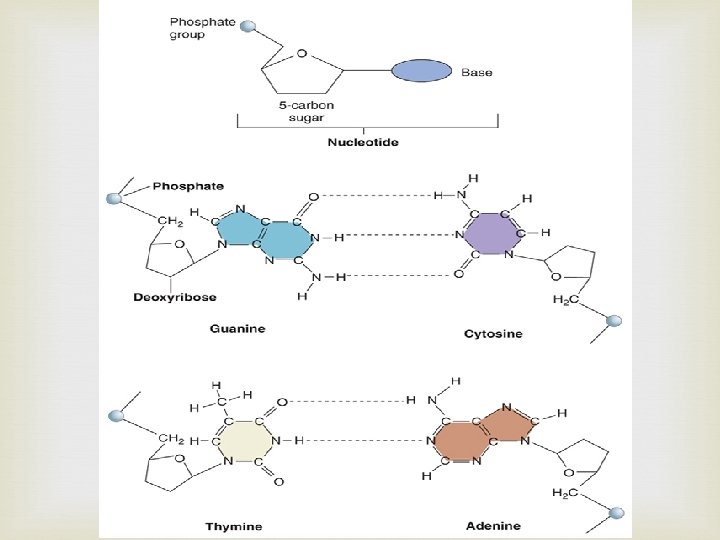
Nucleic Acids Contain H, O, N, C, and P Built from individual units called nucleotides Nucleotides have 3 parts: 5 -carbon sugar Phosphate group Nitrogenous base Nucleic acids store and transmit genetic information Two kinds: DNA RNA
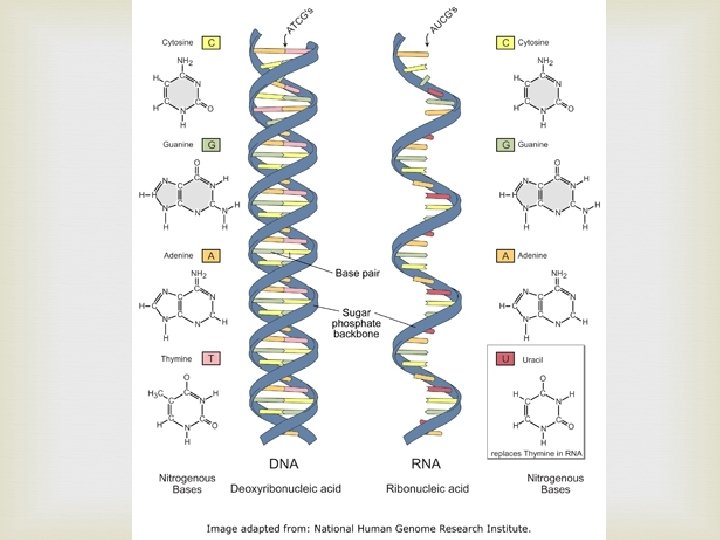

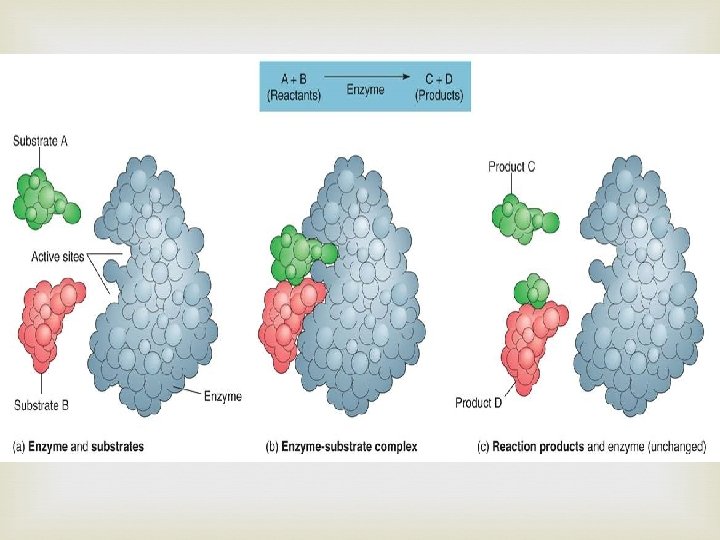
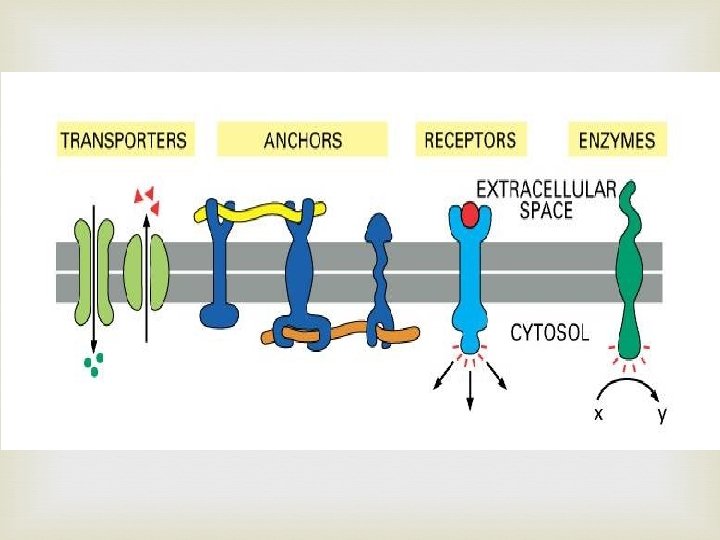
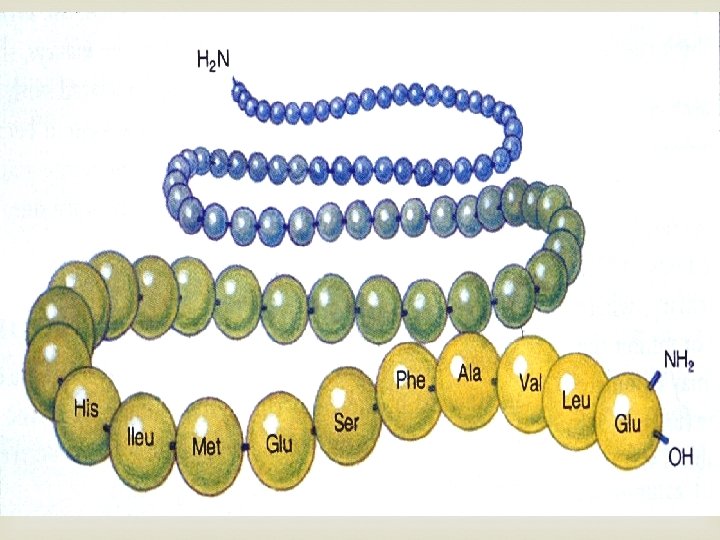
Proteins Contain N, C, H, and O Built from individual units called amino acids More than 20 amino acids are found in nature Think of all the possible combinations of 20 amino acids!!! Proteins have a variety of jobs Control how fast chemical reactions happen Control processes in cells Form bones and muscles Transport substances into or out of cells Help fight disease
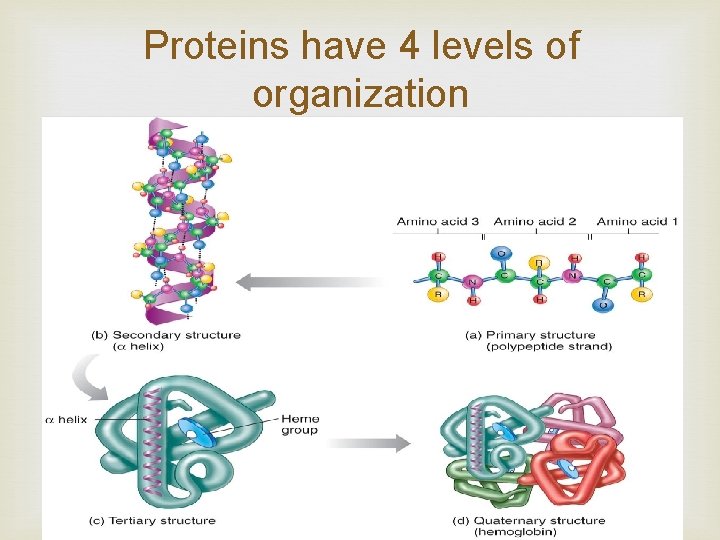
Proteins have 4 levels of organization

Hold up number of fingers … All biomolecules are mostly made of 1. Water 2. Nitrogen 3. Graphite 4. Carbon 5. Phosphorus

4 - Carbon

Tell a neighbor … These store energy in organisms, and form part of membranes and coverings. 1. Proteins 2. Lipids 3. Carbohydrates 4. Nucleic acids

2 - Lipids

1 -2 -3 SING! These are involved in chemical reactions, cell processes, bones and muscles, transporting substances, and fighting disease. 1. Proteins 2. Lipids 3. Carbohydrates 4. Nucleic acids

1 - Proteins

Four Corners These store and transfer genetic information. 1. Proteins 2. Lipids 3. Carbohydrates 4. Nucleic acids

4 - Nucleic Acids

Write the number in the air … These are the main energy source for all living things. They also provide structure in plants and some animals. 1. Proteins 2. Lipids 3. Carbohydrates 4. Nucleic acids

3 - Carbohydrates
- Slides: 41