Biochemistry The Chemistry of Life Biochemistry The life

Biochemistry The Chemistry of Life

Biochemistry • The life processes (Chapter 1) are chemical in nature. • Chemical reactions occur in life. • Living things are made of chemical compounds.

The Atom- The Basic Unit of Matter • Atom= smallest part of an element that retains the properties of that element • Contain subatomic particles: ▫ ▫ Protons= + charge (p+) Neutrons= 0 charge (n) Electrons= - charge (e-) When protons and electrons are in equal numbers, the atoms are neutral
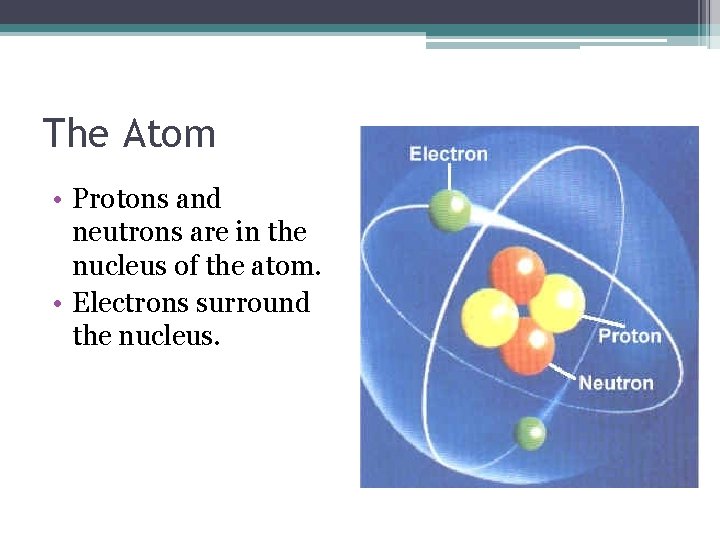
The Atom • Protons and neutrons are in the nucleus of the atom. • Electrons surround the nucleus.

Elements, Molecules, and Compounds • An element is a substance made entirely of 1 type of atom. Ex: C, H, N, O, S • Molecule/Compound- Combination of 2 or more atoms. Ex: H 20

• Atomic Number= Number of protons • Atomic Mass= Total of protons and neutrons (since atoms are neutral, # of protons = # of electrons) • Isotopes- varieties of an element that differ only in the # of neutrons. ▫ All have the same # protons ▫ Ex: Isotopes of Oxygen- Oxygen 16, Oxygen 17, Oxygen 18 (number indicates mass)
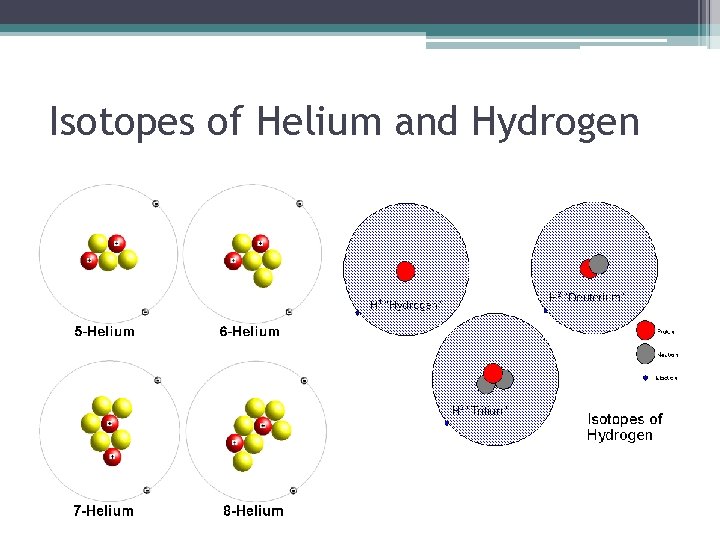
Isotopes of Helium and Hydrogen
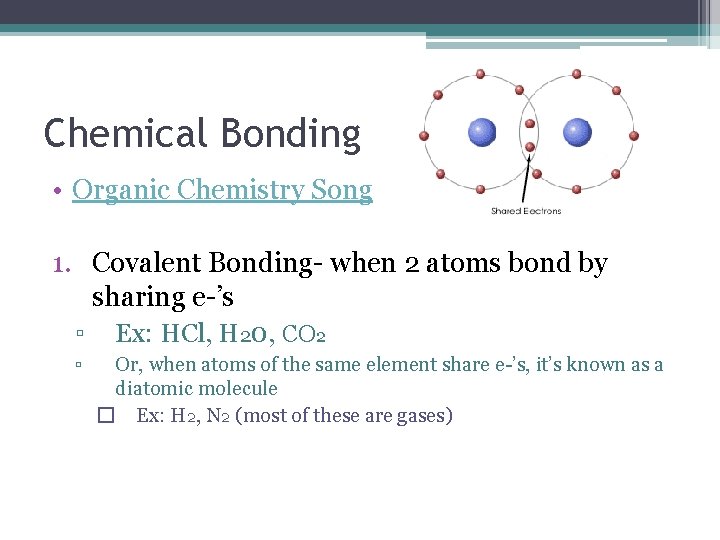
Chemical Bonding • Organic Chemistry Song 1. Covalent Bonding- when 2 atoms bond by sharing e-’s ▫ Ex: HCl, H 20, CO 2 ▫ Or, when atoms of the same element share e-’s, it’s known as a diatomic molecule � Ex: H 2, N 2 (most of these are gases)
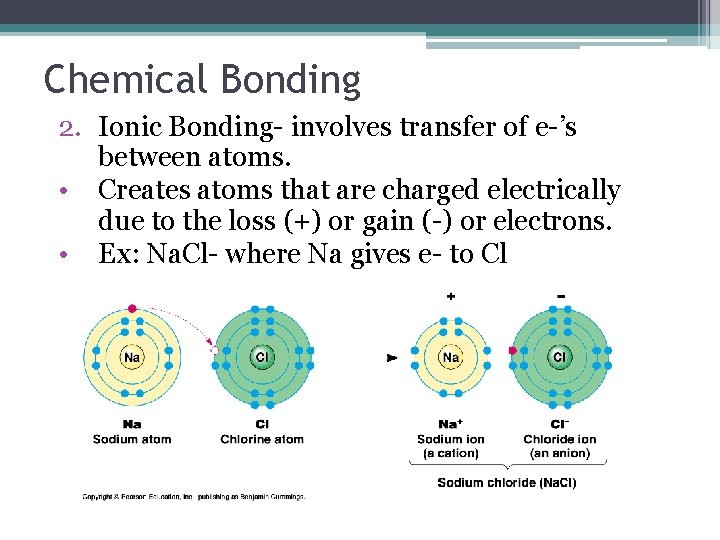
Chemical Bonding 2. Ionic Bonding- involves transfer of e-’s between atoms. • Creates atoms that are charged electrically due to the loss (+) or gain (-) or electrons. • Ex: Na. Cl- where Na gives e- to Cl

Chemical Formulas 1. Molecular Formula- describes the # and type of atoms in a compound: H 2 O, H 2 SO 4 2. Structural Fromula- shoes the #, type, and arrangement of atoms in a compound Water

Isomers • Same molecular formula, but atoms are arranged differently (different structural formula) • Ex: Glucose, Galactose, and Fructose ▫ All are C 6 H 12 O 6, but all have different arrangements/structures

Functional Groups • Groups of atoms added to a molecule that give the molecule certain properties.

Chemical Compounds in Living Things Inorganic Organic No C-H bonds Contain C-H bonds Salts Part of or product of a living thing Acids Bases Water • all living things need water to survive • approx. 65% of body is water • many life processes occur in water (ex: digestion) 4 Groups: 1) Carbohydrates 2) Lipids 3) Proteins 4) Nucleic Acids (in DNA/RNA)

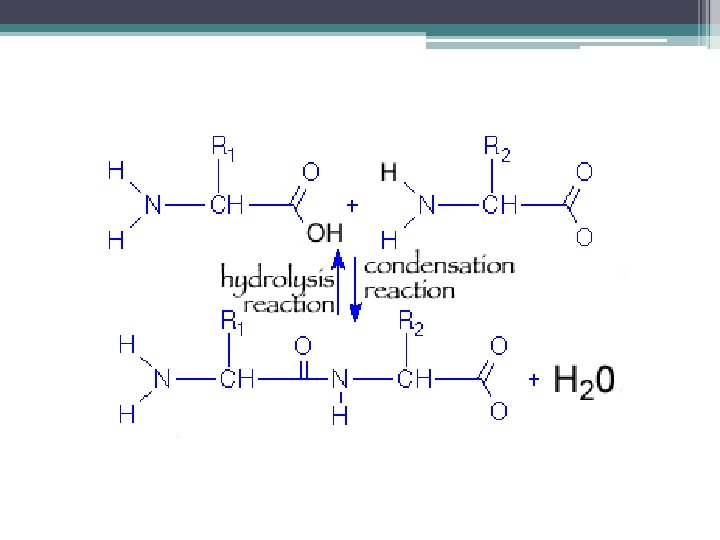
Synthesis and Digestion of Organic Compounds 1. Synthesis- to make a carb, lipid, or protein by combining certain elements ▫ ▫ Water must be removed Called Dehydration Synthesis (DS) Small + Small = Large + Water Molecule Dehydration Synthesis

2. Digestion • Carbs, lipids, and proteins also need to be broken down into smaller pieces so that our cells can utilize them • Water must be added (exact opposite process of DS!) • Called hydrolysis (to lyse= to cut using water) Large + Molecule Water = Small Molecule Hydrolysis + Small Molecule

The 3 Organic Nutrients: 1. • • • Carbohydrates (sugars and starches) Contain C, H, O H: O = 2: 1 Building blocks are monosaccharides (simple sugar) ▫ ▫ ▫ 1 sugar= monosaccharide (glucose, fructose, ribose) 2 sugars= disaccharide (maltose, sucrose) More than 2 sugars= polysaccharide (starch, cellulose, glycogen, chitin)
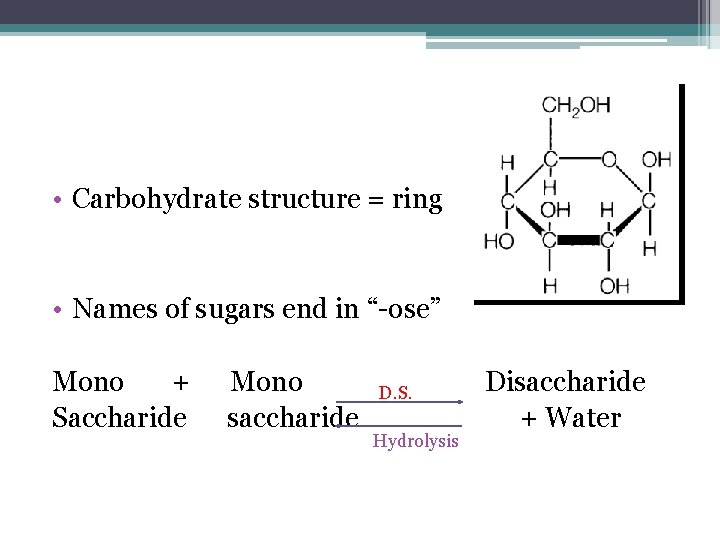
• Carbohydrate structure = ring • Names of sugars end in “-ose” Mono + Saccharide Mono saccharide D. S. Hydrolysis Disaccharide + Water

Carbohydrates • More than 2 monosaccharides joined together form a polysaccharide, generically known as a polymer (large molecules consisting of repeating units). • Functions: energy source, component of cell structures (cell wall, membrane) • Indicators used to test for presence of carbs ▫ Benedict’s (sugar indicator solution) turns orange after heat- for simple/monosaccharides ▫ Lugol’s Iodine (starch indicator solution) turns blackfor polysaccharides/starch
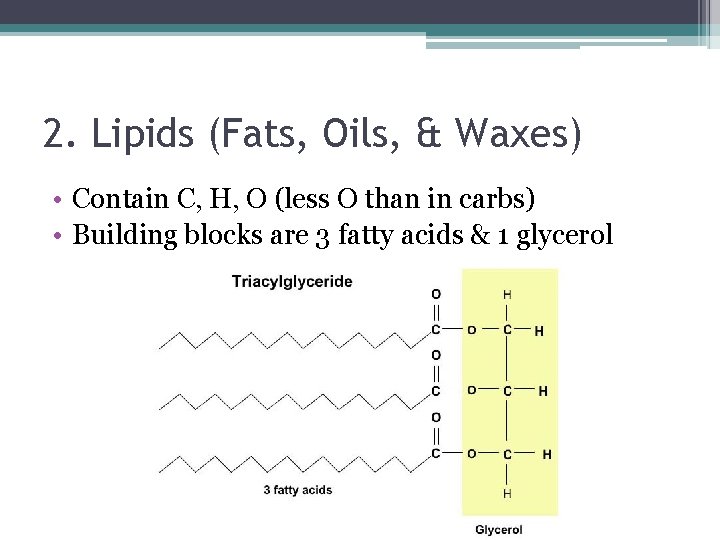
2. Lipids (Fats, Oils, & Waxes) • Contain C, H, O (less O than in carbs) • Building blocks are 3 fatty acids & 1 glycerol

Lipids • Unsaturated fats have double or triple C=C bonds. • Saturated fats have single C-C bonds. D. S. 1 Glycerol + 3 Fatty Acids Hydrolysis 1 Fat + 3 Water Molecule

Lipids • Functions: ▫ ▫ Stored energy Components of cell structures (membrane) Insulation Cushioning • Indicator Used: ▫ Brown Paper- Turns translucent in presence of lipids
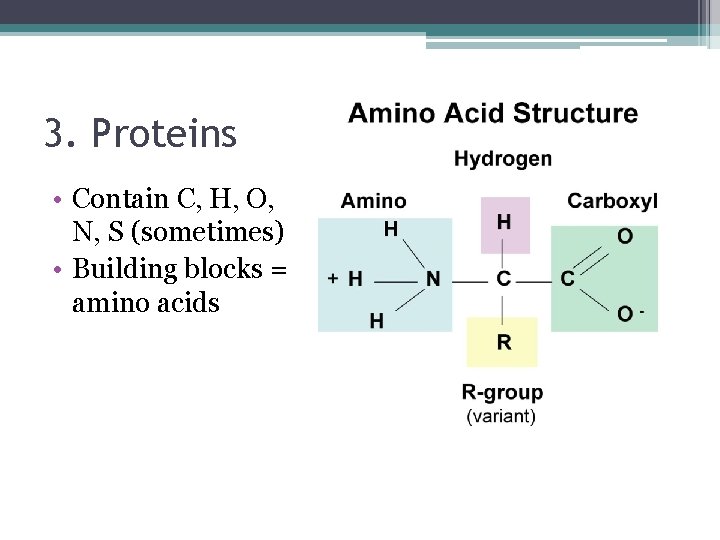
3. Proteins • Contain C, H, O, N, S (sometimes) • Building blocks = amino acids

Proteins • There are 20 amino acids in nature. • Peptide bonds: The C-N bond between 2 amino acids ▫ Dipeptide= 2 amino acids ▫ Polypeptide= more than 2 amino acids

Proteins • Examples: meat, fish, eggs, beans, nuts • Functions: ▫ Components of cell structures (membranes) ▫ Build body tissues (blood, muscle, insulin, hemoglobin, enzymes, antibodies, hair, nails, cartilage). • Indicator Used: Biuret’s Reagent, turns light purple • Proteins are considered to be the “machinery of life. ” They are found throughout living organisms. A characteristic shared by all proteins is that their function is determined by the shape of the protein molecule.
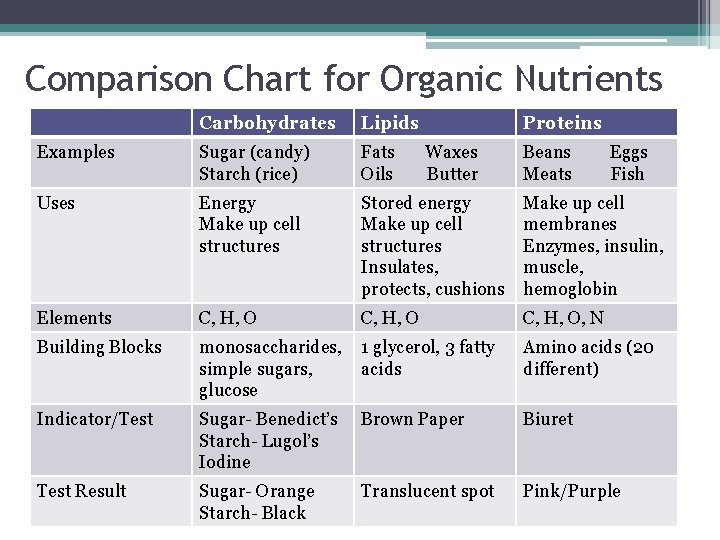
Comparison Chart for Organic Nutrients Carbohydrates Lipids Proteins Examples Sugar (candy) Starch (rice) Fats Oils Uses Energy Make up cell structures Stored energy Make up cell structures Insulates, protects, cushions Make up cell membranes Enzymes, insulin, muscle, hemoglobin Elements C, H, O, N Building Blocks monosaccharides, simple sugars, glucose 1 glycerol, 3 fatty acids Amino acids (20 different) Indicator/Test Sugar- Benedict’s Starch- Lugol’s Iodine Brown Paper Biuret Test Result Sugar- Orange Starch- Black Translucent spot Pink/Purple Waxes Butter Beans Meats Eggs Fish
Enzymes • “Protein catalysts” that are necessary for most of the chemical reactions (DS and hydrolysis) that occur in living cells. • Each chemical reaction requires a specific enzyme!

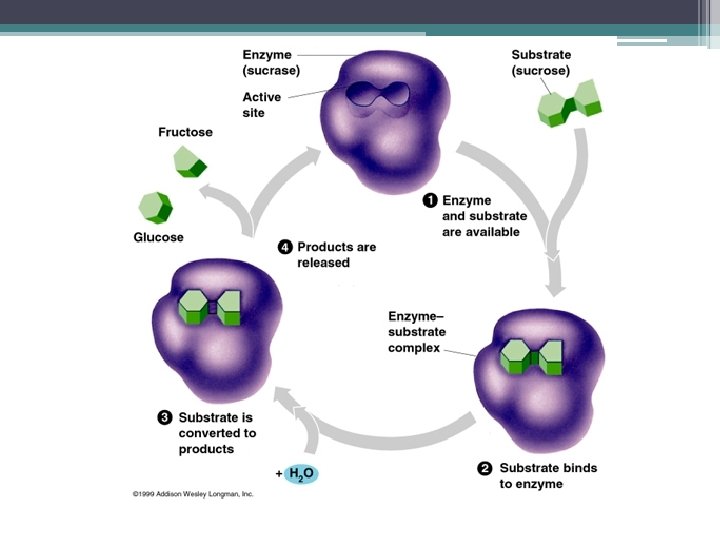
Enzyme Structure • Large complex proteins • Names usually end in “-ase. ” • May contain a non-protein portion caled a coenzyme. Vitamins are coenzymes. • They are usually larger than the substance (substrate) they act on. • The part of the enzyme that carries out the activity/ binds with the substrate is called the active site.

Enzyme-Substrate Complex • Close physical association • No chemical bonding • After reaction (DS, hydolysis) is done, enzyme and substrate separate • Enzyme is unchanged during the reaction and is reused in future reactions
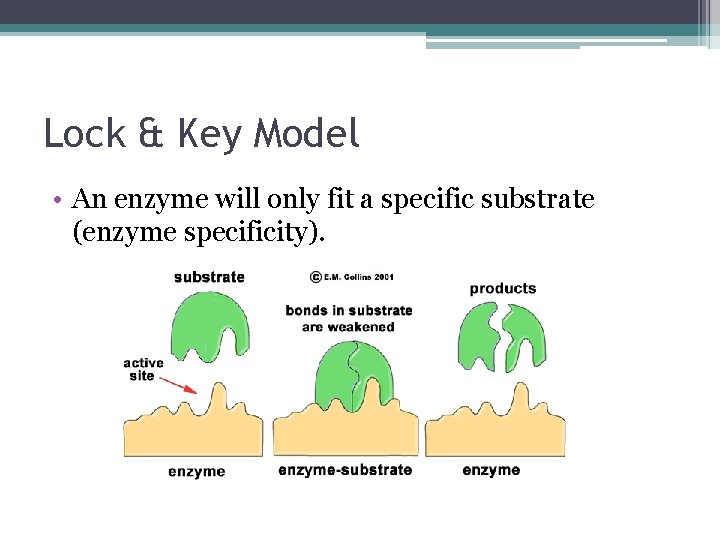
Lock & Key Model • An enzyme will only fit a specific substrate (enzyme specificity).
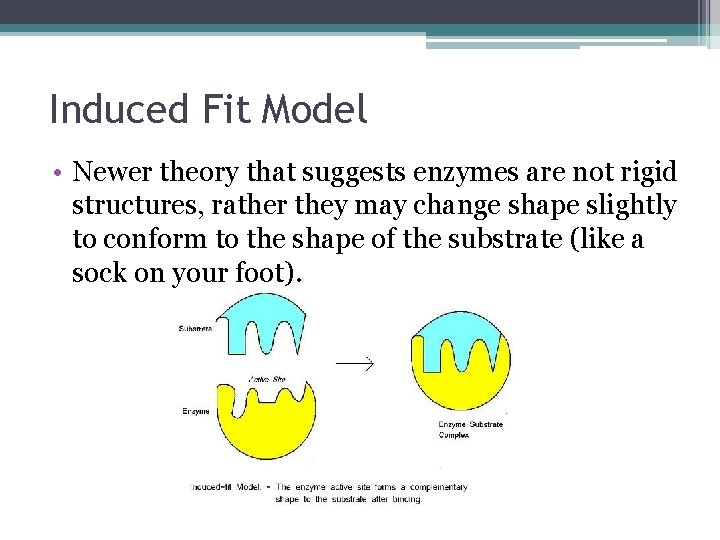
Induced Fit Model • Newer theory that suggests enzymes are not rigid structures, rather they may change shape slightly to conform to the shape of the substrate (like a sock on your foot).

Factors Affecting Enzyme Action 1. Temperature ▫ ▫ ▫ Generally, as temperature increases, enzyme action increases. However, at very high temperature, the shape of the enzyme is altered (“melts”) and is no longer effective = denaturation In humans, denaturation occurs near 40˚C (104˚F).
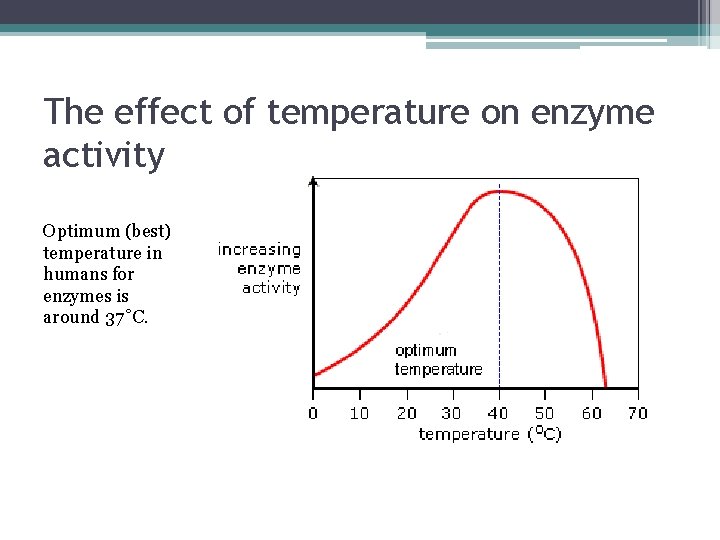
The effect of temperature on enzyme activity Optimum (best) temperature in humans for enzymes is around 37˚C.

Factors Affecting Enzyme Action 2. Amount of Enzyme & Substrate • The amount of available substrate molecules affects enzyme action: ▫ When an excess of substrate is added to a fixed amount of enzyme, the rate of enzyme action increases to a point, then remains fixed/plateaus.

Factors Affecting Enzyme Action 3. p. H- Hydrogen ion concentration • Scale 1 -14 ▫ p. H 7= neutral ▫ p. H<7= acid ▫ p. H>7= base • Optimum p. H= p. H that an enzyme works best at (optimum p. H in stomach is 2, in large intestine is 9).

Effect of p. H on Enzyme Action
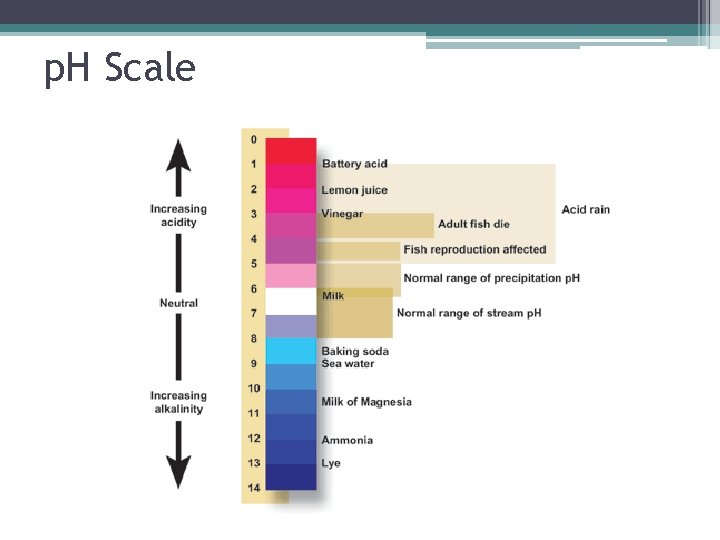
p. H Scale
- Slides: 38