BIOCHEMISTRY THE CHEMISTRY OF LIFE BIOCHEMISTRY In simple

BIOCHEMISTRY THE CHEMISTRY OF LIFE

BIOCHEMISTRY �In simple terms: the chemistry of life �The study of what living things are made of and what “makes them work. ”

THE BASICS: ELEMENTS �Element : single substances which cannot be broken down any more. There are 118 different elements that are known to man. �List of all elements: Periodic Table
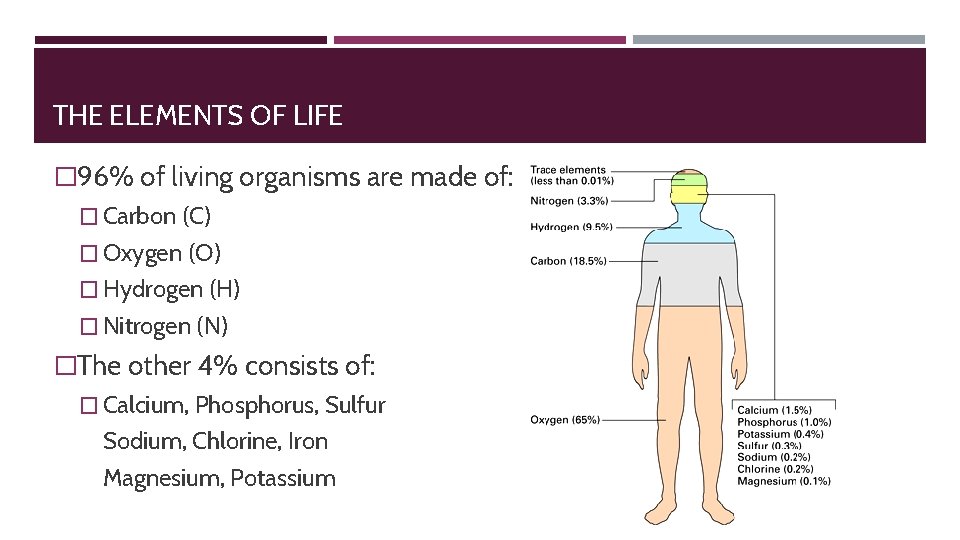
THE ELEMENTS OF LIFE � 96% of living organisms are made of: � Carbon (C) � Oxygen (O) � Hydrogen (H) � Nitrogen (N) �The other 4% consists of: � Calcium, Phosphorus, Sulfur Sodium, Chlorine, Iron Magnesium, Potassium
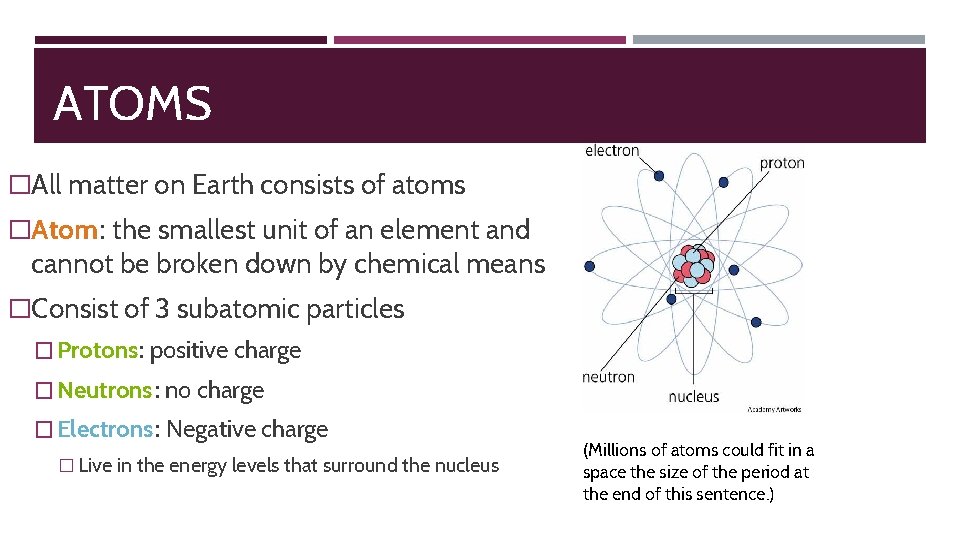
ATOMS �All matter on Earth consists of atoms �Atom: the smallest unit of an element and cannot be broken down by chemical means �Consist of 3 subatomic particles � Protons: positive charge � Neutrons: no charge � Electrons: Negative charge � Live in the energy levels that surround the nucleus (Millions of atoms could fit in a space the size of the period at the end of this sentence. )

CHEMICAL BONDING: HOW ATOMS JOIN �Atoms can form stable substances when joining together through a chemical bond �Chemical bond: force that holds 2 or more atoms together

MOLECULES � Formed from the atoms that have been joined by chemical bonds � Examples: � O 2 (Oxygen gas) � Consist of two oxygen atoms that share two pairs of electrons � N 2 (Nitrogen gas)

COMPOUNDS a molecule that contains at least two different elements. � Compound: � All compounds are molecules, but not all molecules are compounds � Examples: � H 20 (water) � CO 2 (carbon dioxide) � Na. Cl (Sodium Chloride aka: salt)

CHEMICAL REACTIONS �How molecules and compounds form �Processes of breaking chemical bonds and forming new ones Reactants �Written like so: ______ Starting material Products Newly formed substances _______

CHEMICAL REACTIONS �During reactions: energy is absorbed or released, supplying energy for the organism �Examples: �Water: 2 H 2 + O 2 → 2 H 2 O Reactants Product �Photosynthesis: 6 CO 2 + 6 H 2 O → C 6 H 12 O 6 + 6 O 2 Reactants Products

MACROMOLECULES THE MOLECULES OF LIFE

MACROMOLECULES (LARGE, COMPLEX MOLECULES) Monomer → simple building block (one lego) Polymer → large complex molecule made of many monomers (house built of legos) Polymers are made from monomers

MACROMOLECULES: THE MOLECULES OF LIFE 4 main macromolecules found in organisms: � Carbohydrates � Lipids � Nucleic Acids � Proteins
11/15 -16 30 Reflection: My Favorite Macromolecules Draw your favorite (or favorites) source for proteins, carbohydrates, and lipids. Include: Color Detail Title Label Name of polymer: Name of monomer:
- Slides: 14