Biochemistry The Chemistry of Life Basic Chemistry Atom

Biochemistry The Chemistry of Life

Basic Chemistry • Atom – Simplest unit of matter – Made up of three different “subatomic” particles

Subatomic Particles • Protons – Have a positive charge (+) – Have mass – Located in nucleus • Neutrons – Are neutral, have no charge (0) – Have mass – Located in nucleus • Electrons – Have a negative charge (-) – Almost no mass – Located in “energy levels” outside of nucleus

• Atomic Number: – Tells you the number of protons an atom has, and also the # of electrons Atoms are neutral # Protons (+) = # Electrons (-)

• Atomic Mass: – Add together the number of protons and neutrons in nucleus of atom – Electrons hardly contribute any mass

• Isotopes: – Atoms of one type of element with different atomic masses – Same # of protons – Different # neutrons – Changes the mass only, the properties are the same

• Radioactive Isotopes: – The nucleus of some isotopes is unstable – The atom with emit radiation – Radiation can be measured with tools • ex. Geiger counter – These isotopes can be used as useful tools in science – Ex: • Carbon-14 dating • Radiation treatment for cancer

• Elements – Substance made of only one type of atom – Each element has a unique atomic # – Elements most commonly found in living things • • Carbon (C) Hydrogen (H) Oxygen (O) Nitrogen (N)

• Compounds – Two or more elements chemically combined – Atoms held together by bonds. – Once bonds form, compounds will have new, and different properties

– Organic Compounds • Contains carbon and hydrogen (and often O, N) • Ex: Glucose (C 6 H 12 O 6), carbohydrate, lipids, proteins – Inorganic Compounds • Doesn’t contain carbon and hydrogen together • Ex: H 2 O, CO 2, NH 3, Na. Cl

• Why do atoms form compounds? – Bonds that form between atoms give atoms a stable outer electron level. – Called a “stable octet” of valence electrons (8)

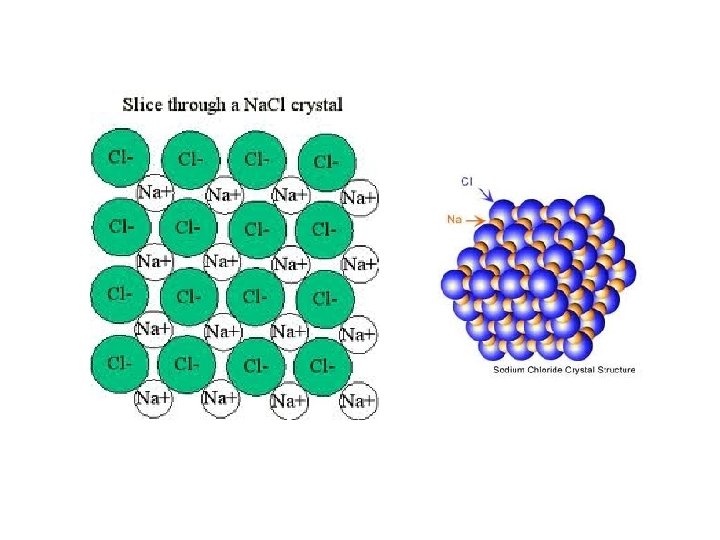
Types of Chemical Bonds • Ionic Bonds: – One or more electrons are transferred from one atom to another. – Neutral atoms become positive & negative ions – Forms salts • (Ex: Na. Cl) Ionic bond = attraction between (+) and (-) ions
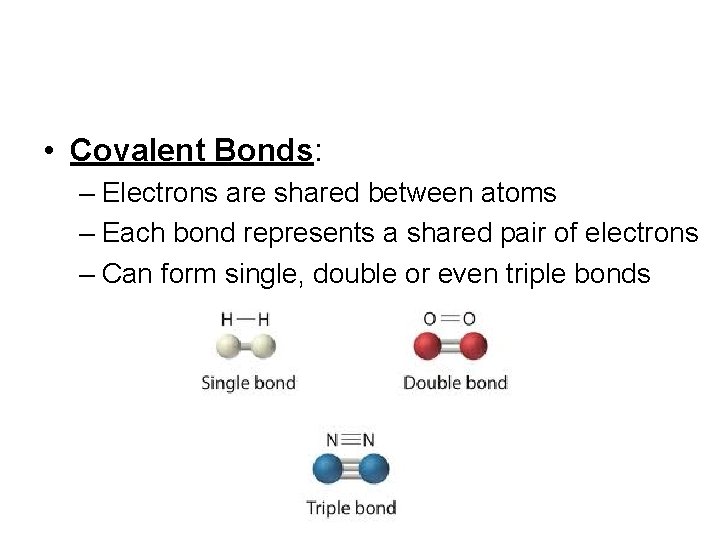
• Covalent Bonds: – Electrons are shared between atoms – Each bond represents a shared pair of electrons – Can form single, double or even triple bonds
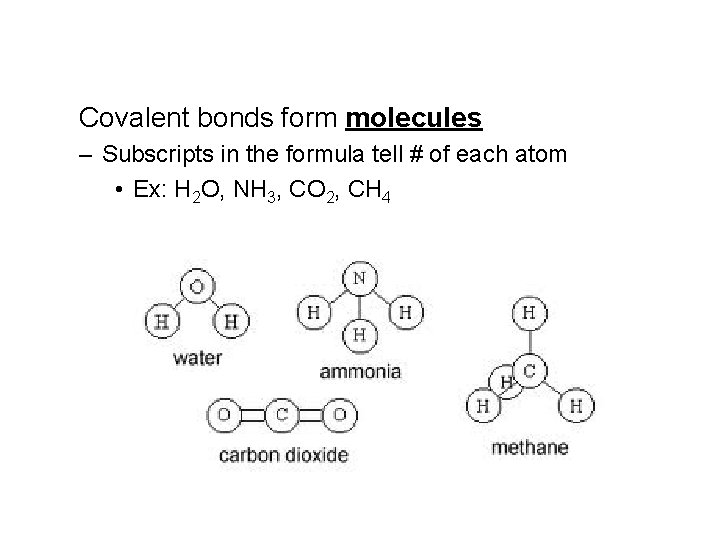
Covalent bonds form molecules – Subscripts in the formula tell # of each atom • Ex: H 2 O, NH 3, CO 2, CH 4
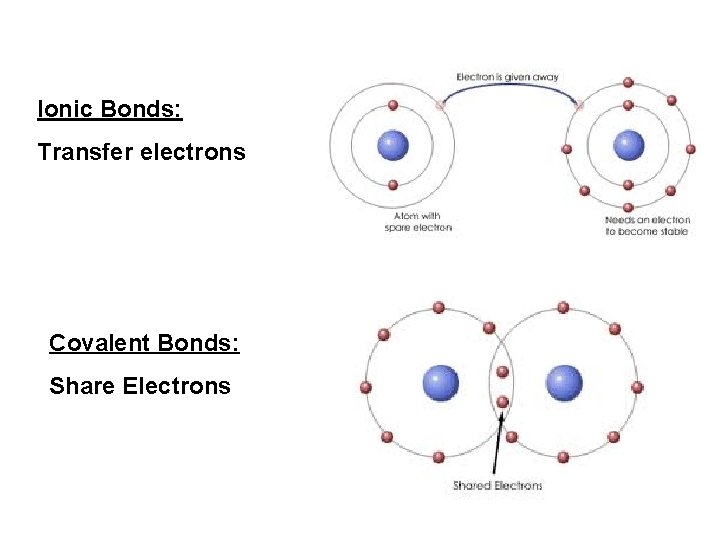
Ionic Bonds: Transfer electrons Covalent Bonds: Share Electrons

Properties of Water • Water is necessary for life • Contains covalent bonds
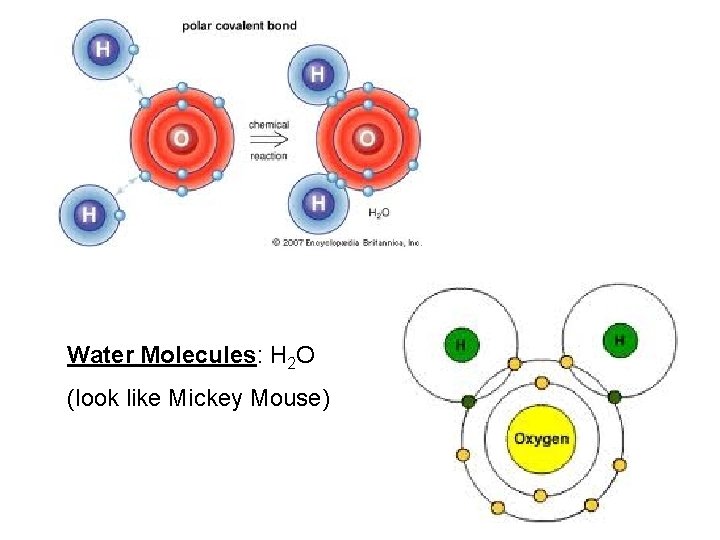
Water Molecules: H 2 O (look like Mickey Mouse)
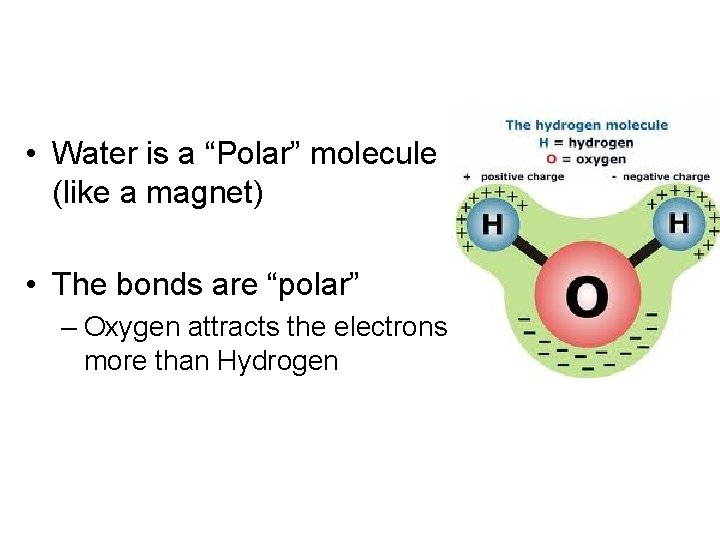
• Water is a “Polar” molecule (like a magnet) • The bonds are “polar” – Oxygen attracts the electrons more than Hydrogen
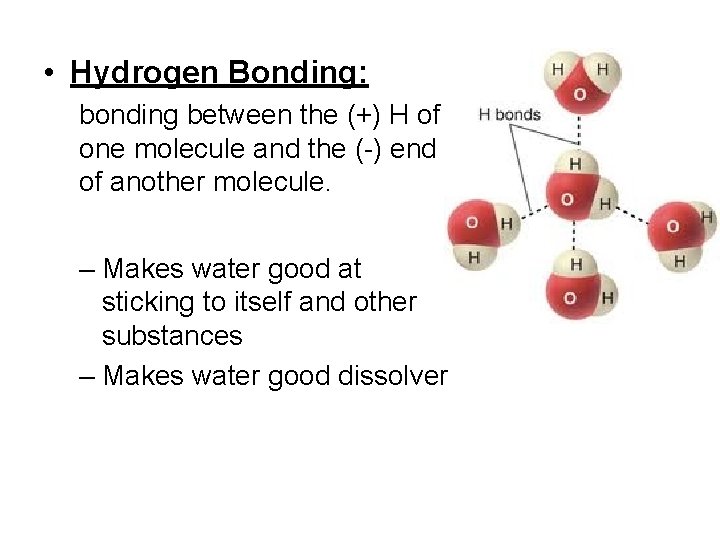
• Hydrogen Bonding: bonding between the (+) H of one molecule and the (-) end of another molecule. – Makes water good at sticking to itself and other substances – Makes water good dissolver

• Cohesion: water sticks to itself – Ex: • Water forms “beads” on smooth surface • Surface Tension allows insect to “skate”
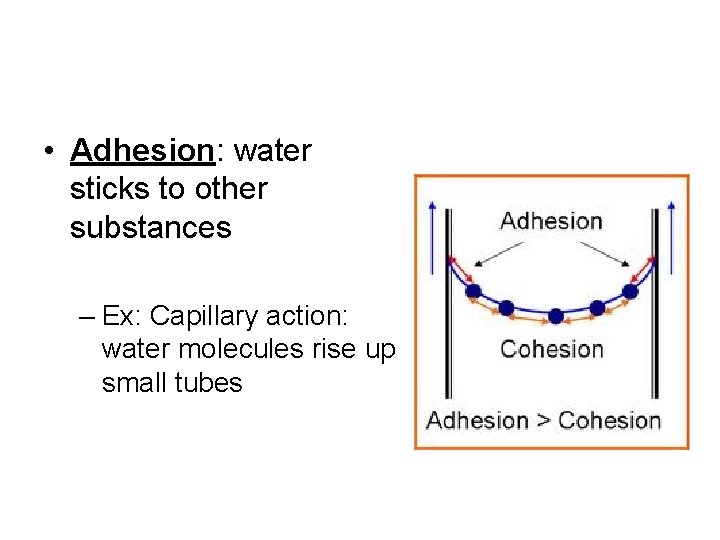
• Adhesion: water sticks to other substances – Ex: Capillary action: water molecules rise up small tubes

• Heat Capacity: – Water has a relatively high heat capacity – Ex: • Lakes and oceans can absorb a lot of heat from sun without a drastic temperature change

• Water as a Solvent: – Dissolves most ionic and covalent substances – “Universal Solvent” = many things can dissolve in it.

– Solute: substance being dissolved – Solvent: substance in which solute dissolves – Solution: evenly disbursed mixture – Suspensions: material in the water but just suspended not dissolved (ex: blood cells in blood)
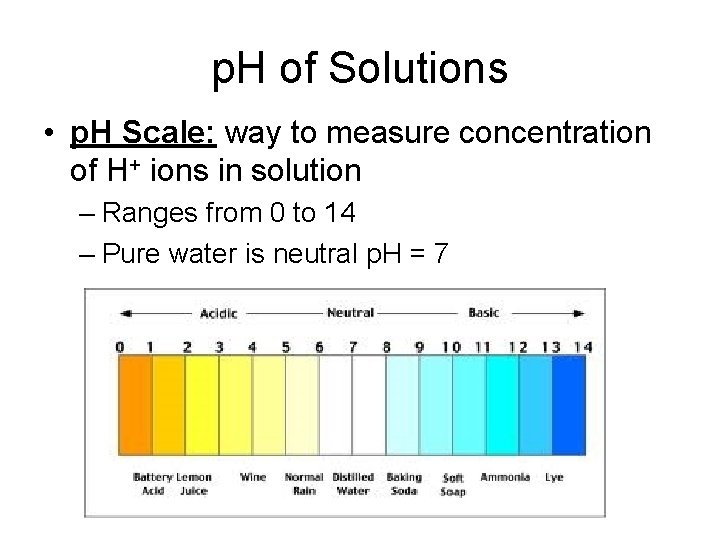
p. H of Solutions • p. H Scale: way to measure concentration of H+ ions in solution – Ranges from 0 to 14 – Pure water is neutral p. H = 7

• Litmus Paper: – Used to test p. H of a solution • Red = acidic • Blue = basic

• Acids: Form H+ ions – p. H is <7 – Ex: HCl (stomach acid), lemon juice

• Bases: Produces OH- (hydroxide ions) – Also called “alkaline” – p. H >7 – Ex: Lye (Na. OH) used as drain cleaner

• Buffers: – Maintain p. H at a certain level – Usually between 6. 5 – 7. 5 (close to neutral) – Helps to maintain homeostasis in organism

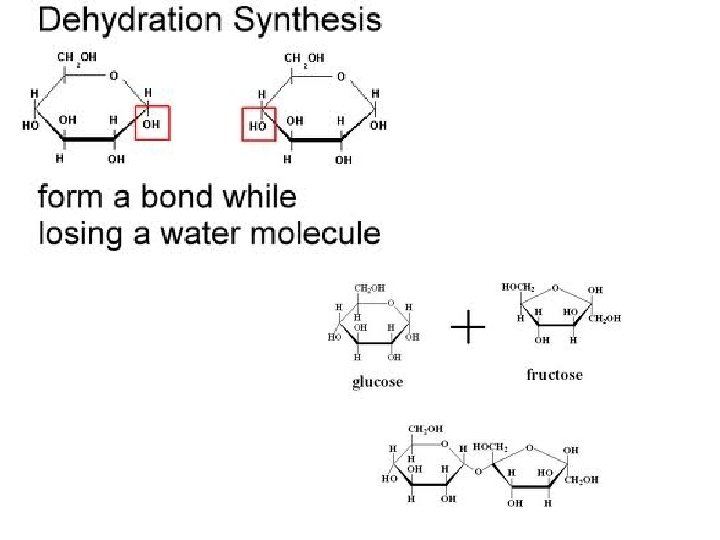
Major Types of Chemical Reactions • Dehydration Synthesis: (Condensation) – Chemically combine two smaller molecules – Water is removed – “Dehydrate” = remove water – “Synthesis” = to make

• Hydrolysis: – Break apart large molecule into smaller pieces – Water is added – “Hydro” = water “Lysis” = to break

• Polymerization: – Create a large molecule (polymer) – Join up smaller “monomer” units – Often a dehydration synthesis reaction Ex: Join amino acids (monomer) to make protein (polymer) Join glucose (monosaccharide) to make starch (polysaccharide)

Organic Compounds
Carbohydrates • Sugars and starches • FUNCTION: – Used as an energy source – Energy released during cellular respiration • Made of carbon, hydrogen and oxygen Ex: C 6 H 12 O 6, C 12 H 22 O 11 • Ratio of H of O is always 2: 1

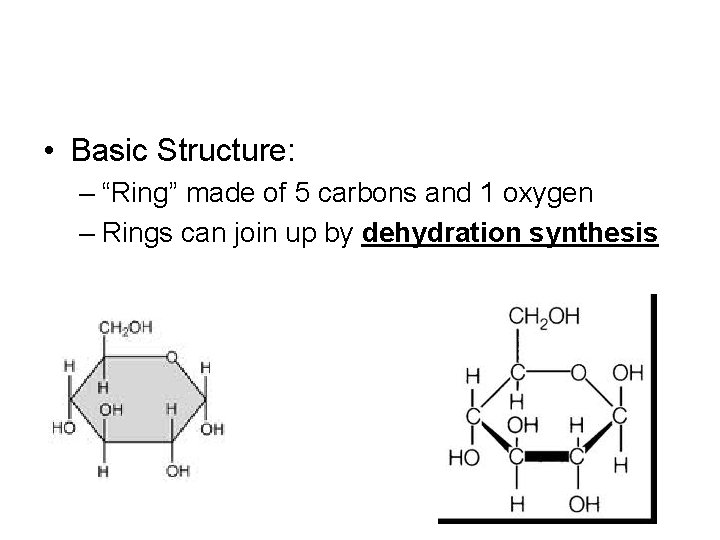
• Basic Structure: – “Ring” made of 5 carbons and 1 oxygen – Rings can join up by dehydration synthesis

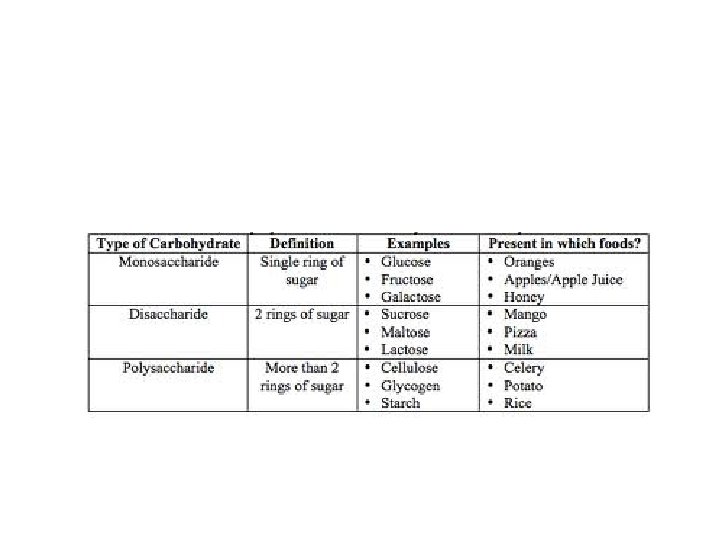
3 Types of Carbohydrates • Monosaccharide's: 1 sugar ring • Disaccharides: 2 sugar rings • Polysaccharides: many sugar rings
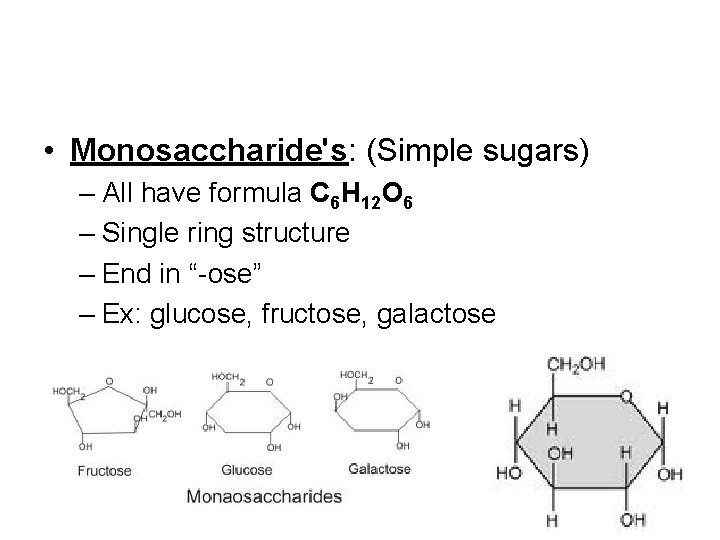
• Monosaccharide's: (Simple sugars) – All have formula C 6 H 12 O 6 – Single ring structure – End in “-ose” – Ex: glucose, fructose, galactose
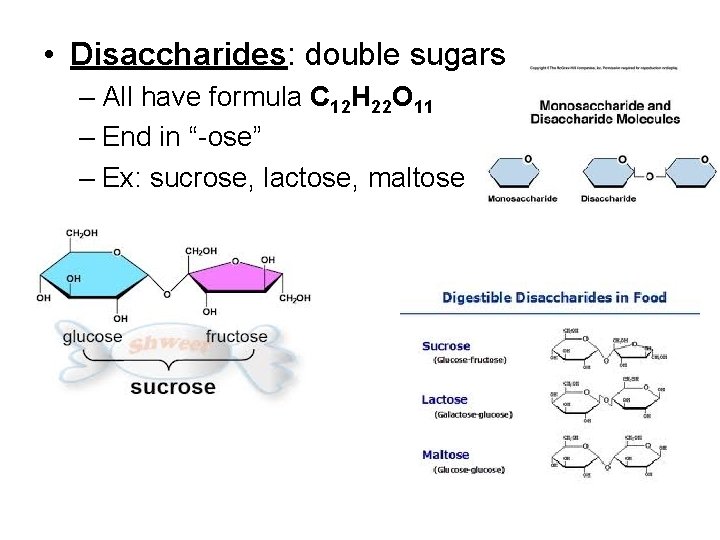
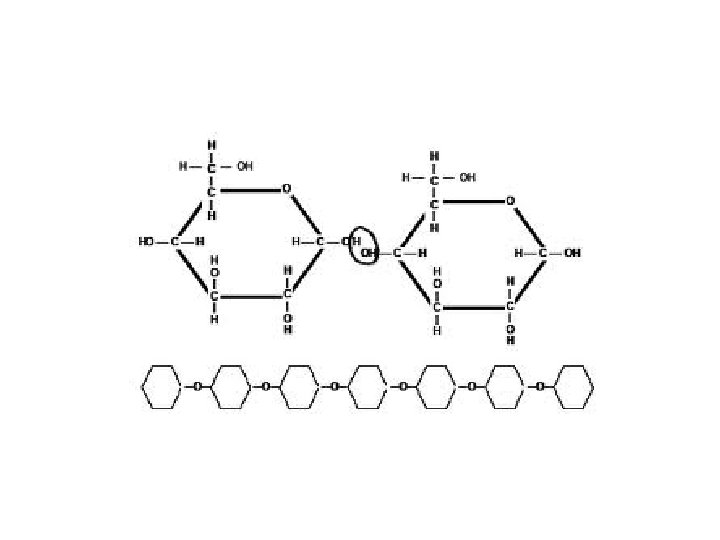
• Disaccharides: double sugars – All have formula C 12 H 22 O 11 – End in “-ose” – Ex: sucrose, lactose, maltose
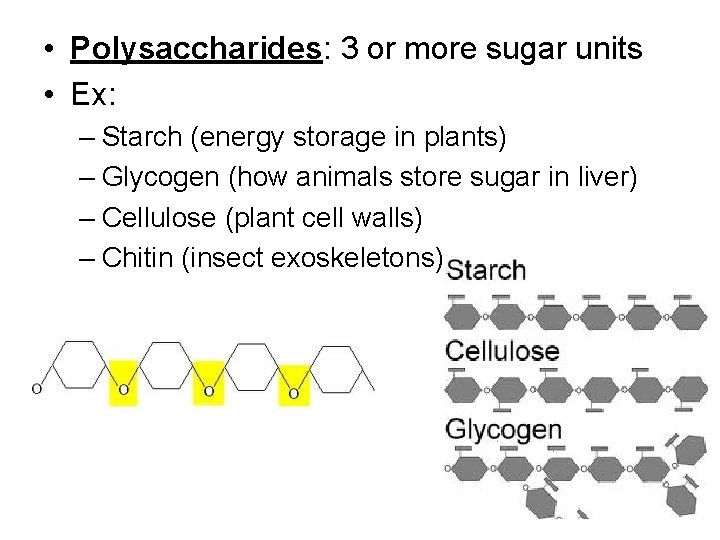
• Polysaccharides: 3 or more sugar units • Ex: – Starch (energy storage in plants) – Glycogen (how animals store sugar in liver) – Cellulose (plant cell walls) – Chitin (insect exoskeletons)
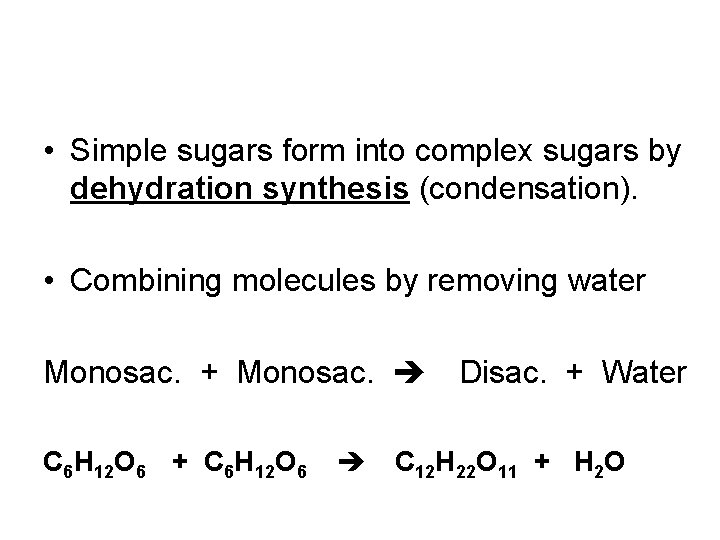
• Simple sugars form into complex sugars by dehydration synthesis (condensation). • Combining molecules by removing water Monosac. + Monosac. C 6 H 12 O 6 + C 6 H 12 O 6 Disac. + Water C 12 H 22 O 11 + H 2 O
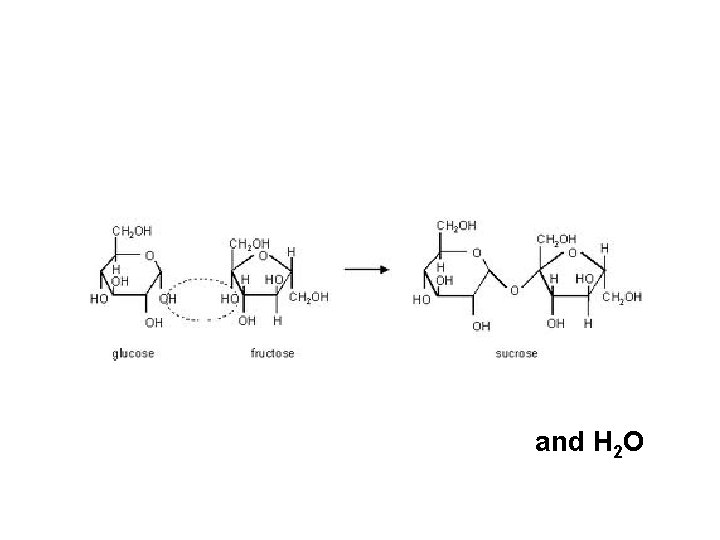
and H 2 O
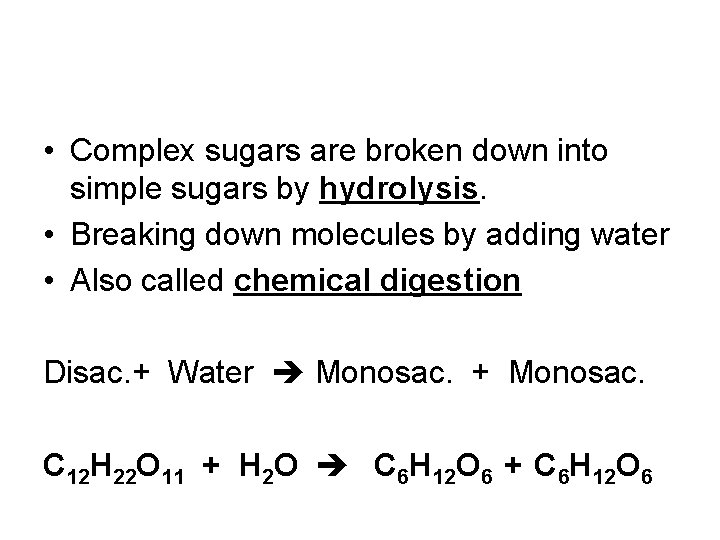
• Complex sugars are broken down into simple sugars by hydrolysis. • Breaking down molecules by adding water • Also called chemical digestion Disac. + Water Monosac. + Monosac. C 12 H 22 O 11 + H 2 O C 6 H 12 O 6 + C 6 H 12 O 6
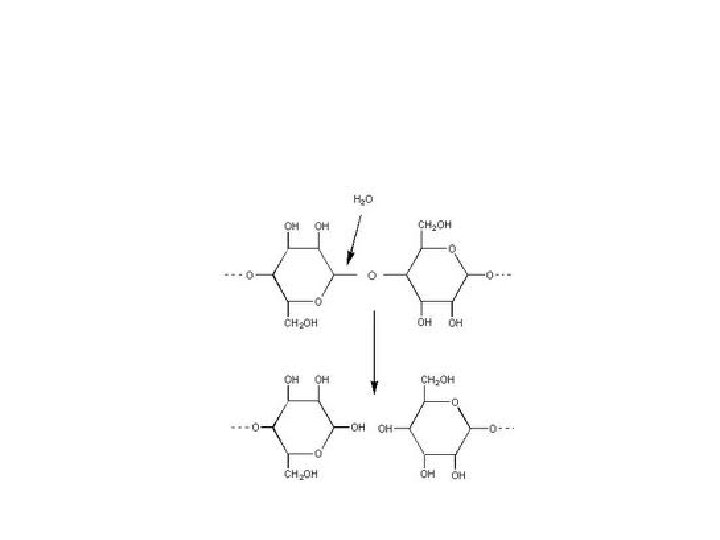
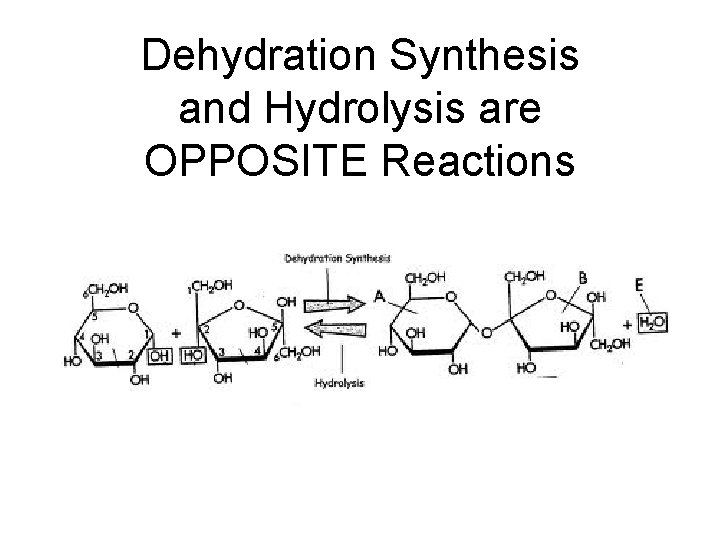
Dehydration Synthesis and Hydrolysis are OPPOSITE Reactions
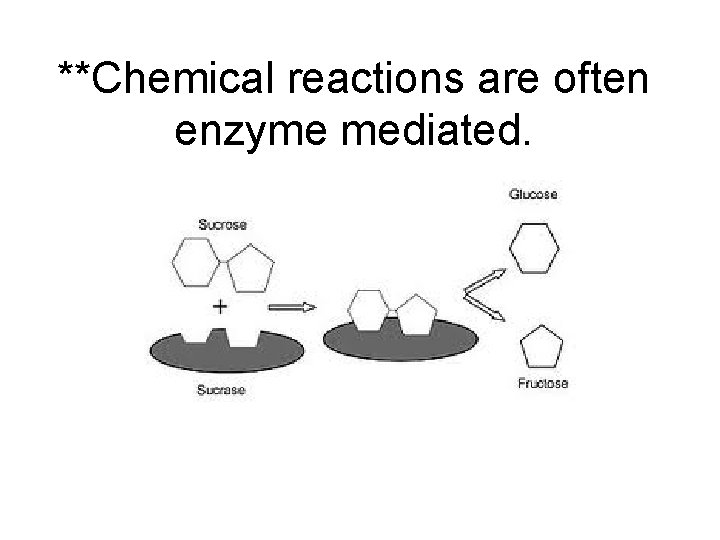
**Chemical reactions are often enzyme mediated.

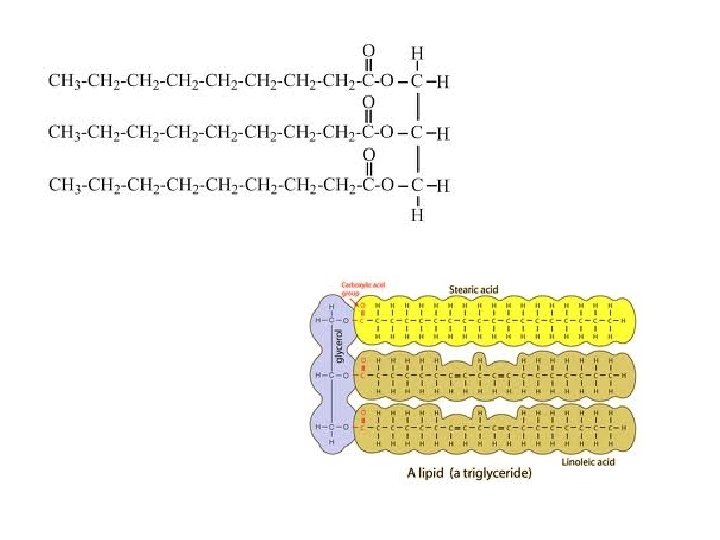
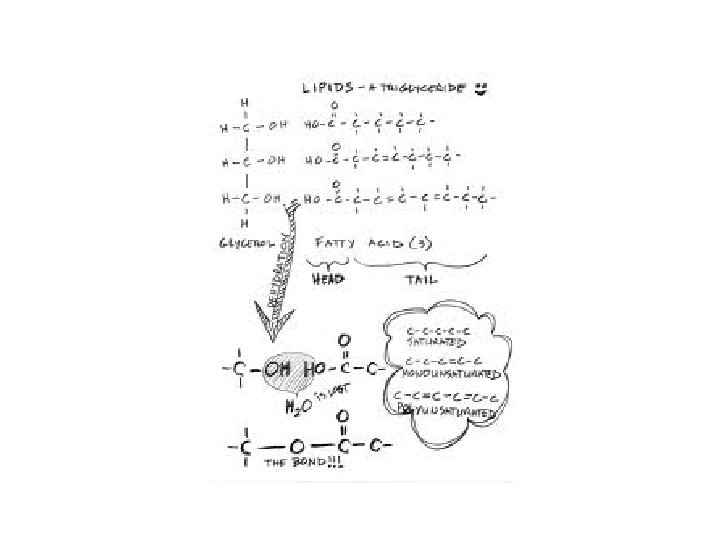
Lipids • Fats, oils and waxes • FUNCTION: – Long term energy storage – Insulation – Protective coating around cells – Cell membranes • Made of carbon, hydrogen, and oxygen • Ratio of H to O is not 2: 1 – Ex: C 15 H 26 O 6 (fewer oxygens)

• Animals store energy mostly as fats • Plants store energy mostly as oils
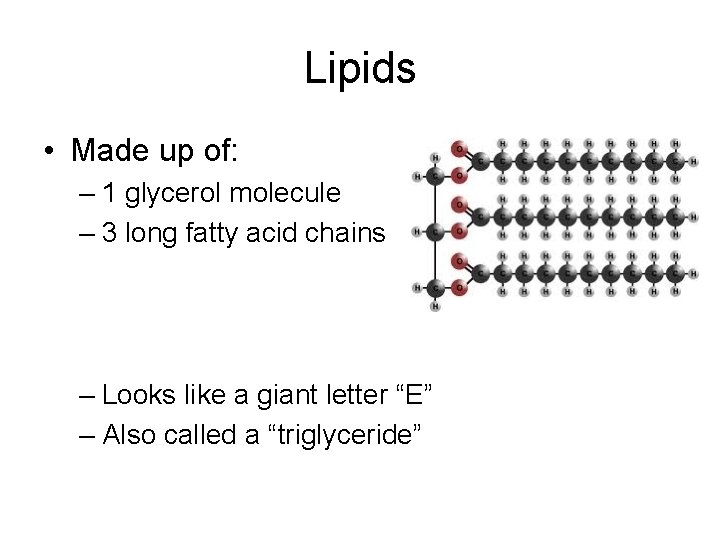
Lipids • Made up of: – 1 glycerol molecule – 3 long fatty acid chains – Looks like a giant letter “E” – Also called a “triglyceride”
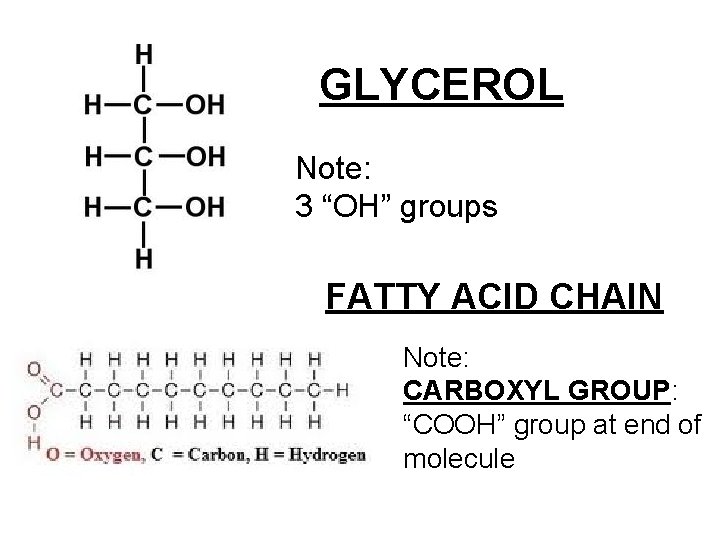
GLYCEROL Note: 3 “OH” groups FATTY ACID CHAIN Note: CARBOXYL GROUP: “COOH” group at end of molecule
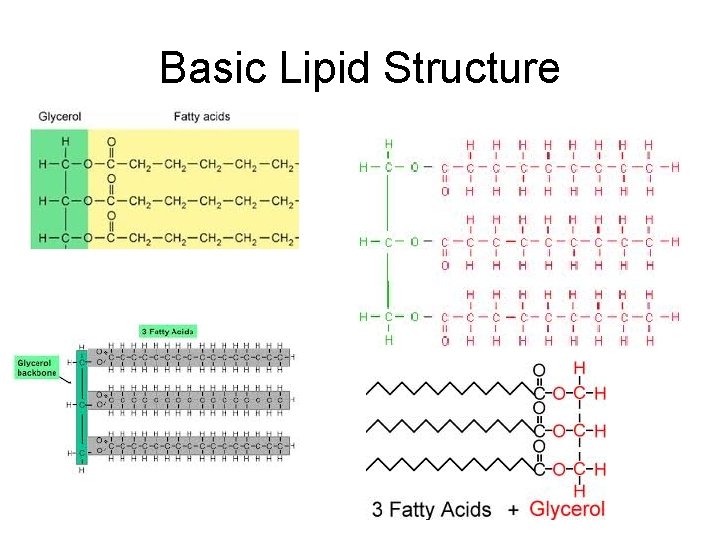
Basic Lipid Structure

• Lipids are: – formed by dehydration synthesis – broken down by hydrolysis
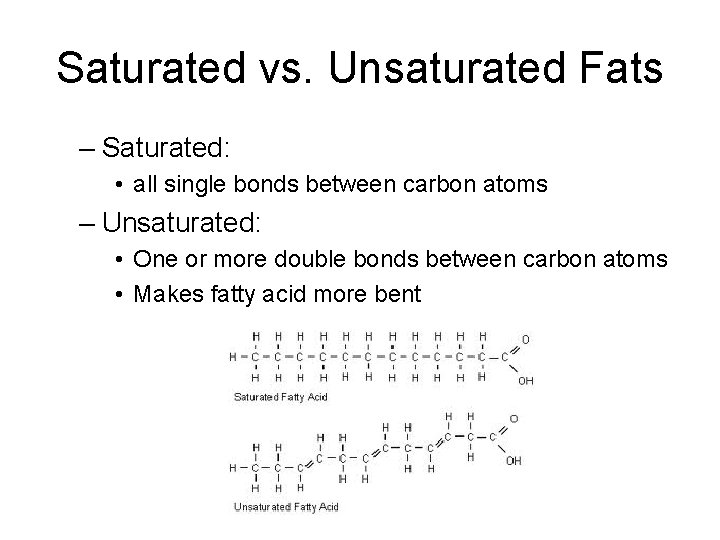
Saturated vs. Unsaturated Fats – Saturated: • all single bonds between carbon atoms – Unsaturated: • One or more double bonds between carbon atoms • Makes fatty acid more bent

Proteins • Protein Structure: – Made of carbon, hydrogen, oxygen and NITROGEN – Large complex polymer molecules that can have a large range of sizes, shapes and properties

Proteins • FUNCTIONS: – Enzymes: speed up chemical reactions – Hormones: chemical messengers – Antibodies: defend against disease – Hemoglobin: binds oxygen to red blood cells – Cell growth and repair – Cell Membrane Channels
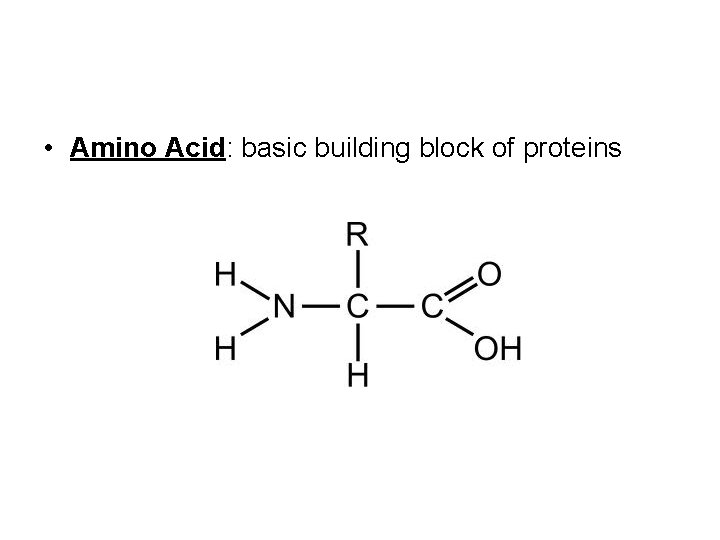
• Amino Acid: basic building block of proteins
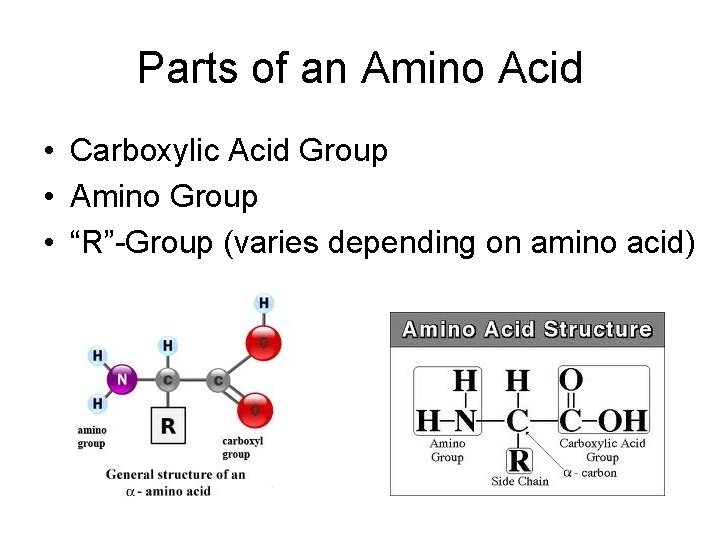
Parts of an Amino Acid • Carboxylic Acid Group • Amino Group • “R”-Group (varies depending on amino acid)

Examples of Different Amino Acids • There are 20 different amino acids • All have different “R” groups
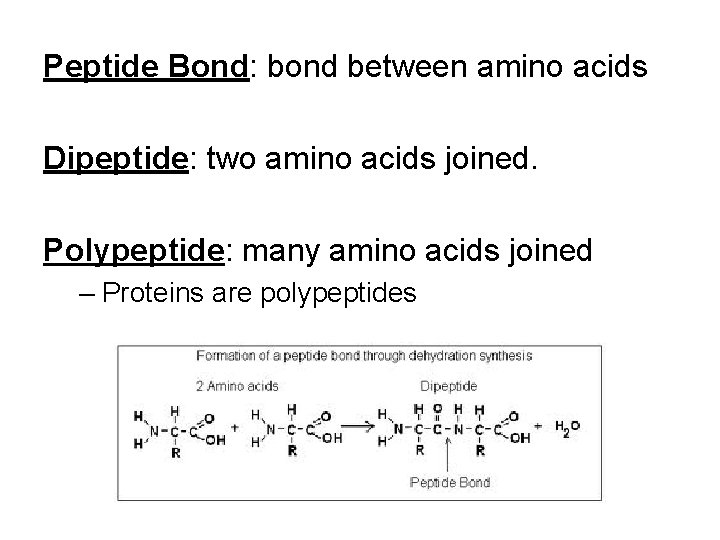
Peptide Bond: bond between amino acids Dipeptide: two amino acids joined. Polypeptide: many amino acids joined – Proteins are polypeptides
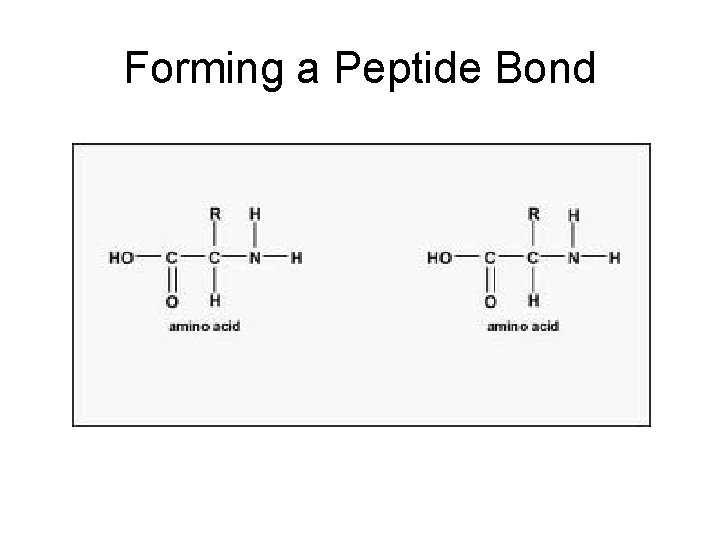
Forming a Peptide Bond
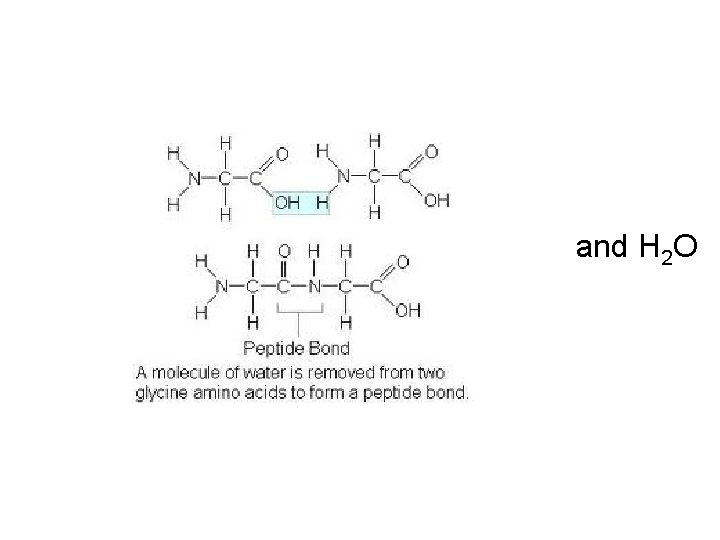
and H 2 O
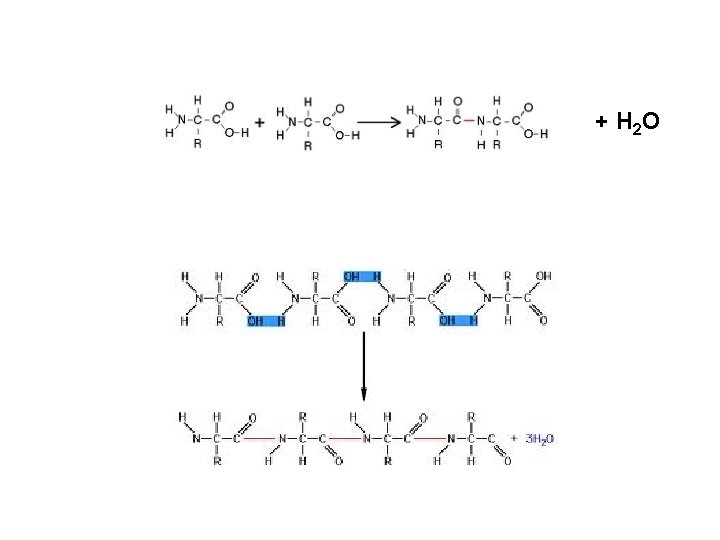
+ H 2 O
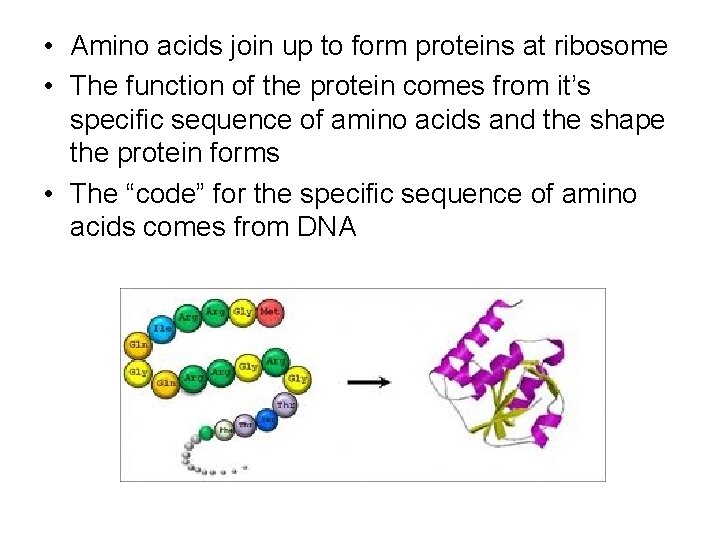
• Amino acids join up to form proteins at ribosome • The function of the protein comes from it’s specific sequence of amino acids and the shape the protein forms • The “code” for the specific sequence of amino acids comes from DNA
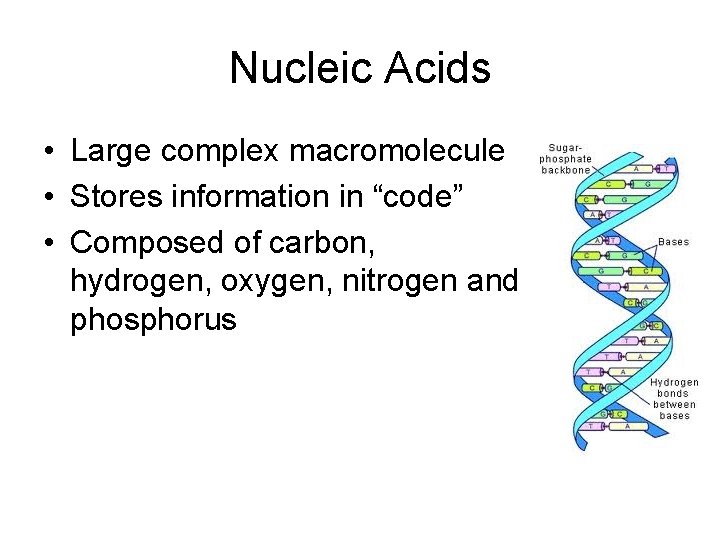
Nucleic Acids • Large complex macromolecule • Stores information in “code” • Composed of carbon, hydrogen, oxygen, nitrogen and phosphorus
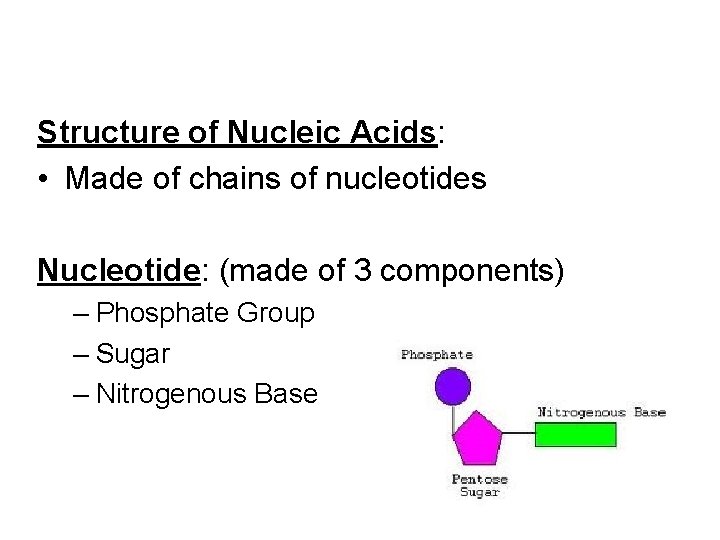
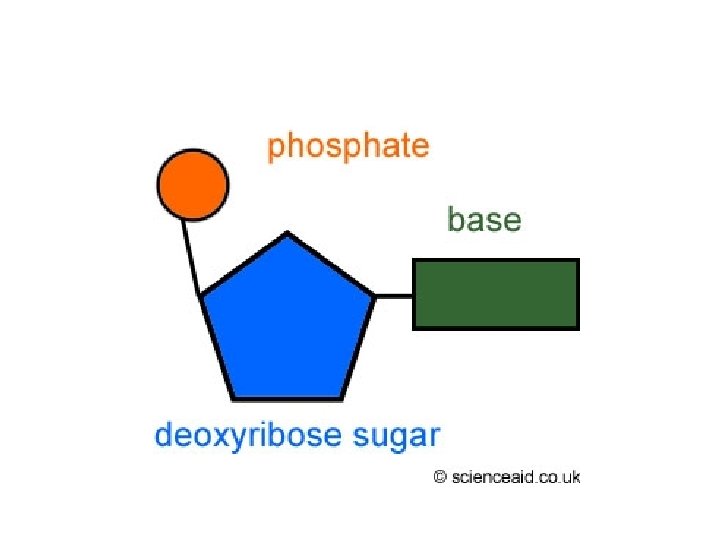
Structure of Nucleic Acids: • Made of chains of nucleotides Nucleotide: (made of 3 components) – Phosphate Group – Sugar – Nitrogenous Base

Two Types of Nucleic Acids • DNA: (Deoxyribonucleic Acid) – Contain genetic information – Found in nucleus – Divides when cell divides • RNA: (Ribonucleic Acid) – Copies code from DNA – Takes code to ribosomes for protein synthesis – Found in nucleus and cytoplasm

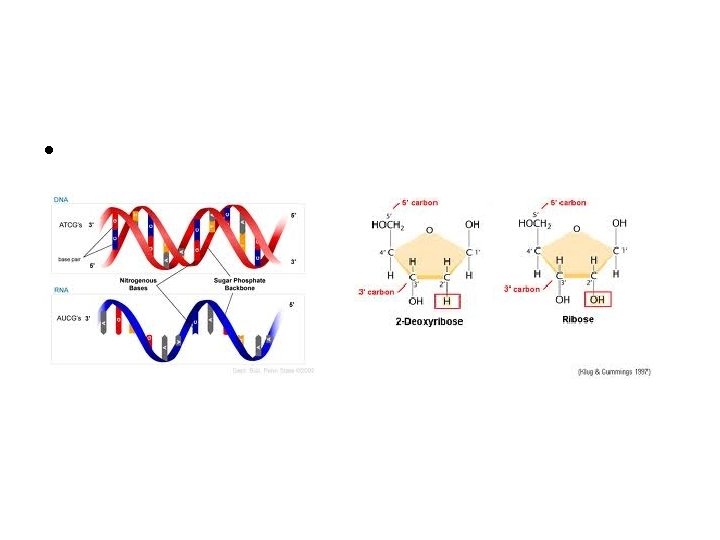
Differences Between DNA & RNA – DNA: • Double strand of nucleotides • Sugar is deoxyribose • Nitrogenous Bases – Adenine, Thymine, Cytosine, Guanine – RNA: • Single strand of nucleotides • Sugar is ribose • Nitrogenous bases – Adenine, Uracil, Cytosine, Guanine
- Slides: 75