BIOCHEMISTRY The chemical basis of life 1 ATOMS

BIOCHEMISTRY The chemical basis of life 1

ATOMS n n Basic unit of matter Two regions n n n Nucleus Electron cloud ic m Ato 6 er b Num Atomic particles n Protons • Positively charged particles • Located in the nucleus n C Carbon 12. 011 Neutrons • Neutral particles • Located in the nucleus & act as the glue there n Electrons • Negatively charged particles • Located in the electron cloud • These are the particles involved in chemical reactions 2

Elements Contain all of the same type of atom within the element n Elements of Life n • 96% Hydrogen (H), Oxygen (O), Nitrogen (N), and Carbon (C) … or as Mr. Curtis calls them “HONC” • 3% P, S, Ca, K, Na, Mg, Fe, Cl • 1% other trace elements 3

Chemical Bonds form between elements n Involve electrons n n Examples: n Ionic bonds • Electrons from one element are taken away and given to another element. n Covalent bonds • Electrons are shared between elements n Hydrogen bonds: • Bonds that hold one hydrogen-containing molecule to another. • Water molecules connect to one another this way. 4

Water (H 2 O) Is the most abundant compound in organisms n Has many unique properties that make water so versatile & so important to life n 5

Properties of Water” n 1. ) Polarity n Electrons are shared unequally between hydrogen & oxygen atoms • Like a Sumo wrestler and a 5 year-old playing tug-owar. n n Important because polarity makes all of the other properties possible. Water has hydrogen bonds, which allow for: n 2. ) Adhesion • H 2 O sticking to other substances n 3. ) Cohesion • H 2 O molecules sticking to other H 2 O molecules n 4. ) Surface tension n • Measure of how difficult it is to stretch or break the surface of a liquid • Allows animals to “walk on water. ” 4. 5) Capillary action • Combination of cohesion & adhesion • This is how trees get water from deep in the ground up to the highest leaves. 6

Water (H 2 O) n n 5. ) High specific heat n Water resists temperature changes when it absorbs or releases heat n Why important: it keeps oceans at constant temperatures. 6. ) High heat of vaporization n Water (liquid) can absorb a lot of heat before it changes to a gas. n Why important: it prevents oceans from boiling away. n Why important: it stabilizes earth’s climate. n Human impact: • Evaporative cooling • As a liquid evaporates, the surface of the liquid that remains behind cools down. • This is why sweating cools us down. 7
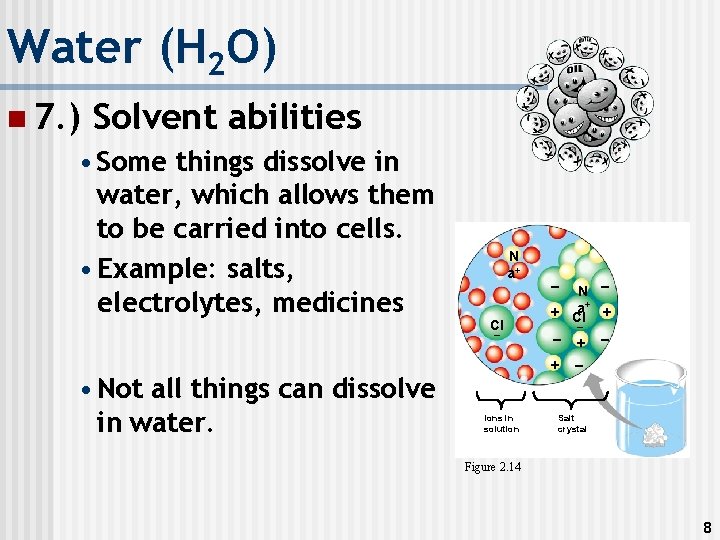
Water (H 2 O) n 7. ) Solvent abilities • Some things dissolve in water, which allows them to be carried into cells. • Example: salts, electrolytes, medicines N a+ Cl – • Not all things can dissolve in water. – N – a+ + + Cl – + Ions in solution – + – – Salt crystal Figure 2. 14 8

Biochemistry 9

Organic vs. Inorganic n All compounds can be separated into two groups: n Inorganic • Doesn’t contain carbon • Non-living • Examples: Oxygen gas, metals, rocks, water n Organic • Contains carbon • Living (or dead) • Examples: wood, grass, petroleum 10

Polymerization n Monomers n One unit of a compound n Polymers n Many monomers combine to make a polymer n Macromolecules n Many large molecules combined 11

n Example: n Bricks in a wall n Bricks = monomers n The whole wall = polymer 12

Biochemistry The 4 Organic Macromolecules 13

1. ) Carbohydrates n n Made of C, H, & O Functions n Main energy source in organisms n Structural component in plants 14

Types of Carbohydrates n 1. ) Monosaccharides • AKA: Simple sugars • give off energy when broken down • Example: glucose (blood sugar) & fructose (fruit sugar) n 2. ) Disaccharides • Double sugars…two monosaccharides put together. • Example: sucrose (table sugar) n 3. ) Polysaccharides • Structural polysaccharides • Provide support & structure to cells • Example: cellulose (in plants) & chitin (in fungi) • Storage polysaccharides • Store sugar • Ex: starch (plants) and glycogen (in animals) 15

2. ) Lipids n n n Made of C, H, O Common types: fats, oils, & waxes Functions n n n Storage of energy Parts of biological membranes Waterproof coverings Chemical messengers (steroids) Insoluble in water (because they are non-polar) Ex. Lard, butter, oil, hormones, steroids 16
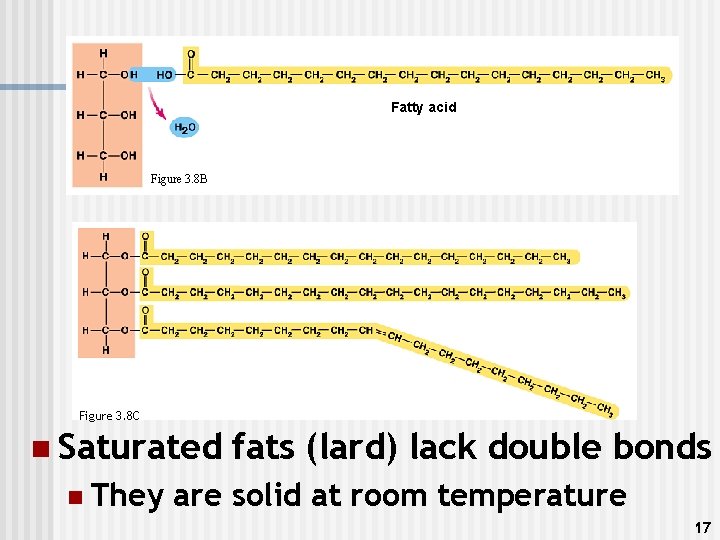
Fatty acid Figure 3. 8 B Figure 3. 8 C n Saturated n They fats (lard) lack double bonds are solid at room temperature 17
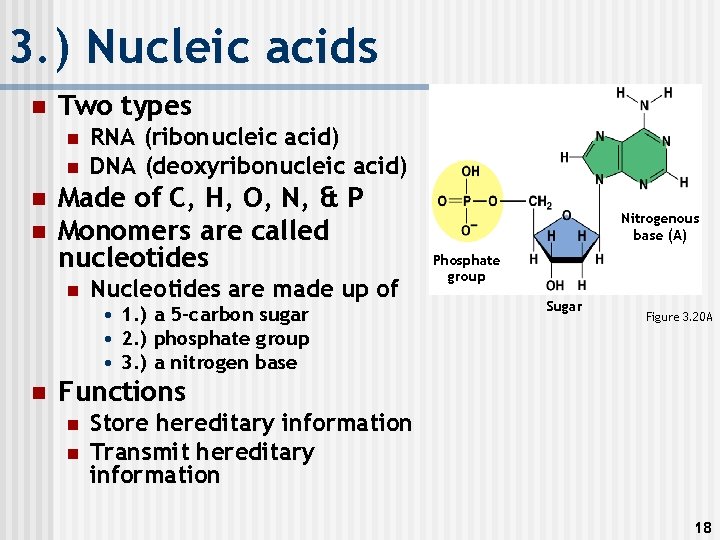
3. ) Nucleic acids n Two types n n RNA (ribonucleic acid) DNA (deoxyribonucleic acid) Made of C, H, O, N, & P Monomers are called nucleotides n Nucleotides are made up of • 1. ) a 5 -carbon sugar • 2. ) phosphate group • 3. ) a nitrogen base n Nitrogenous base (A) Phosphate group Sugar Figure 3. 20 A Functions n n Store hereditary information Transmit hereditary information 18

4. ) Proteins n n Made of C, H, O, & N Monomers are amino acids n n n There are 20 different amino acids that combine in different ways to make millions of proteins Proteins are the most diverse group of macromolecules Amino group Carboxyl (acid) group Figure 3. 12 A Functions n n n Control the rates of chemical reactions (enzymes) Regulate cell processes Form bone & muscles Transport substances into or out of cells Help fight diseases (antibodies) 19
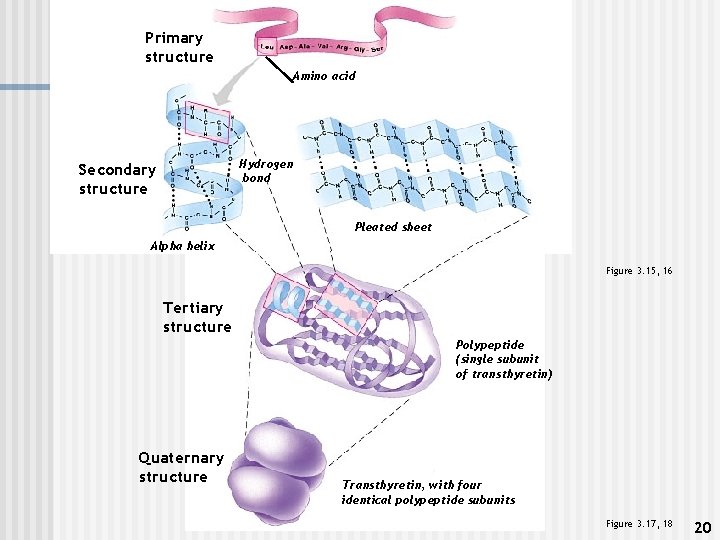
Primary structure Amino acid Hydrogen bond Secondary structure Pleated sheet Alpha helix Figure 3. 15, 16 Tertiary structure Polypeptide (single subunit of transthyretin) Quaternary structure Transthyretin, with four identical polypeptide subunits Figure 3. 17, 18 20

3 – 2 -1 Exit Ticket n Tell me n 3 things you learned today n 2 - Questions you have n 1 – Connection you made ( Why/ how will you remember this? ) Place your paper in the tray by the door on your way out 21

Opener 1/30/13 n What is the difference between a Saturated and Unsaturated fat? (hint * Has something to do with bonding) n Why does this make a difference in the food you eat? n What types of foods do you find Saturated fats? Give two examples. 22

Enzymes The “ 411” on biological catalysts. 23

Vocab: n Reactant: The starting material in a reaction. n similar to “ingredients” in a recipe. n Usually on the left side of the reaction. n n Product: What is produced (made) at the end of the reaction. n Usually on the right side of the reaction. n 24

Example: n Glucose n What + O 2 CO 2 + H 20 are the reactants? n Glucose n What n CO 2 and O 2 are the products? and H 20 25
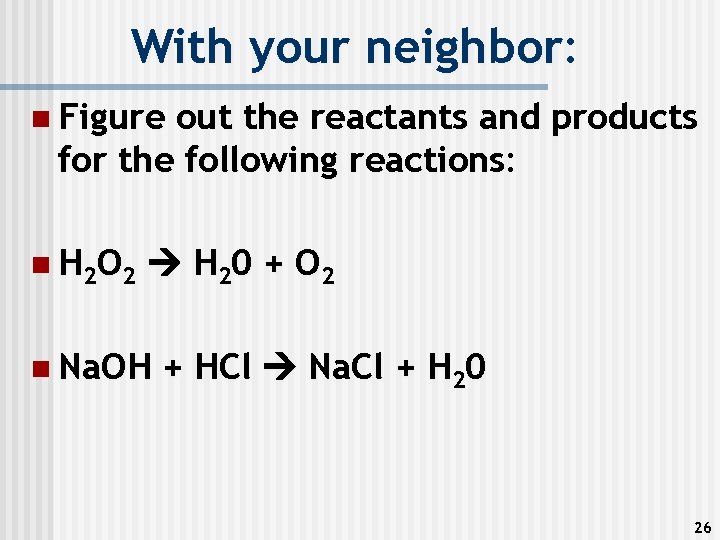
With your neighbor: n Figure out the reactants and products for the following reactions: n H 2 O 2 H 20 + O 2 n Na. OH + HCl Na. Cl + H 20 26

CATALYST n Is anything that speeds up a chemical reaction. 27

Enzyme n Is a protein that acts as a catalyst in living organisms. 28

How do enzymes speed up chemical reactions in cells? n They speed up reactions by lowering the activation energy for the reaction. n The activation energy is the amount of energy that must be absorbed by the reactants for the reaction to start. n General rule: the higher the activation energy, the harder it is to do the reaction. 29
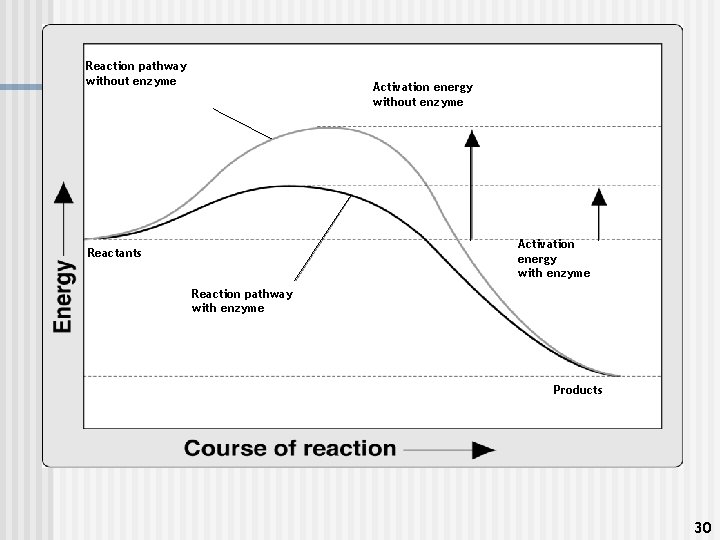
Reaction pathway without enzyme Activation energy with enzyme Reactants Reaction pathway with enzyme Products 30

Parts of the Enzymecatalyzed Reaction 31

Substrate n This is the molecule being changed by the enzyme-catalyzed reaction (it is the reactant in the reaction). n Enzymes are specific as to the substrate they work on. (L&K/IF) n The place on the enzyme where the substrate bonds is called the Active Site. 32
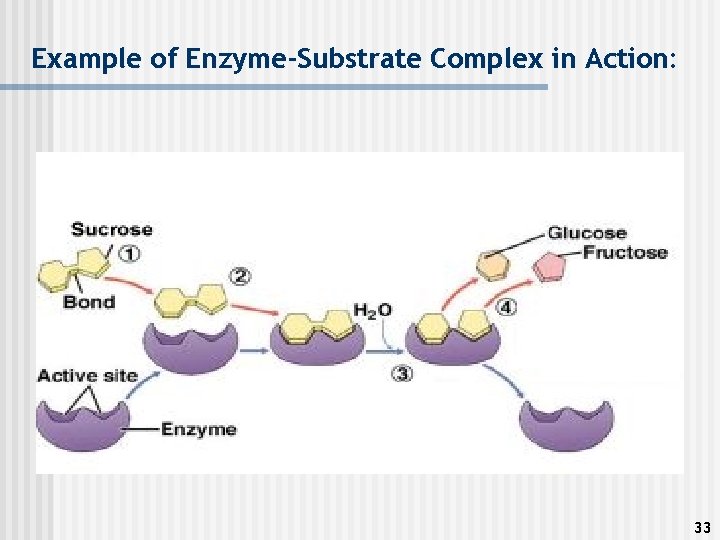
Example of Enzyme-Substrate Complex in Action: 33

See page 56 in your textbook for a picture of the Enzyme. Substrate Complex 34

Do enzymes always work? n NO!!! n They by: can be inactivated (denatured) n p. H changes n High temperatures (above normal body temperature. ) n Salty environments 35

So…why are high fevers so dangerous? n The high temperatures will break down the proteins in your body, including the enzymes. 36

Review 37

#1 n The is: n A. ) reactant in a chemical equation what you finish with. n B. ) what is produced. n C. ) what you start with n D. ) Both A and C.

#2 n The n A. ) product in a chemical reaction is what you finish with. n B. ) what you start with. n C. ) also called an ingredient n D. ) Both A and C.

#3 n Identify the reactant in the following equation: n Fe. Cl 2 n A. ) + Mg. O Fe. O + Mg. Cl 2 Fe. Cl 2 + Fe. O n B. ) Mg. O + Mg. Cl 2 n C. ) Fe. O + Mg. Cl 2 n D. ) Fe. Cl 2 + Mg. O

#4 n Anything that can speed up a reaction is known as: n A. ) an enzyme n B. ) a catalyst n C. ) a protein n D. ) activation energy

#5 n. A protein that functions as a catalyst is called a/an: n A. ) amino acid n B. ) enzyme n C. ) accelerator n D. ) decelerator

#6 n The energy needed to start a chemical reaction is called: n A. ) Start-up cost n B. ) Activation energy n C. ) Reactivation energy n D. ) ATP

#7 n Enzymes n A. ) work by: increasing activation energy. n B. ) decreasing activation energy n C. ) making the cell work harder n D. ) breaking down monomers into polymers.

#8 n Which of the following will interfere with an enzyme? n A. ) increased activation energy n B. ) p. H changes n C. ) other enzymes n D. ) A and C

#9 n The part of an enzyme where the substrate binds is called the: n A. ) active site n B. ) passive site n C. ) enzyme site n D. ) reactivation site
- Slides: 46