Biochemistry SOL BIO 3 b c BIO 3

Biochemistry SOL BIO 3 b, c

BIO 3 b, c OBJECTIVE: • TSW investigate and understand the chemical and biochemical principles essential for life. Key concepts include • b) the structure and function of macromolecules • c) the nature of enzymes.

• Most life processes are a series of chemical reactions influenced by environmental and genetic factors.

Metabolism • the sum of all biochemical processes

2 Metabolic Processes • Anabolism- the building up of complex molecules • Catabolism- the breaking down of complex molecules

• Most cells function best within a narrow range of temperature and p. H. • At very low temperatures, reaction rates are too slow. • High temperatures or extremes of p. H can irreversibly change the structure of proteins and alter their function.
–The main components of a living cell are: • Carbon • Hydrogen • Nitrogen • Oxygen -Phosphorus -Sulfur

• Inside every cell is a concentrated mixture of thousands of different macromolecules forming a variety of specialized structures that carry out cell functions, such as: – – – energy production transport waste disposal synthesis (creation) of new molecules storage of genetic material.

Organic Compounds • Compounds that contain CARBON are called organic • Macromolecules are large organic molecules
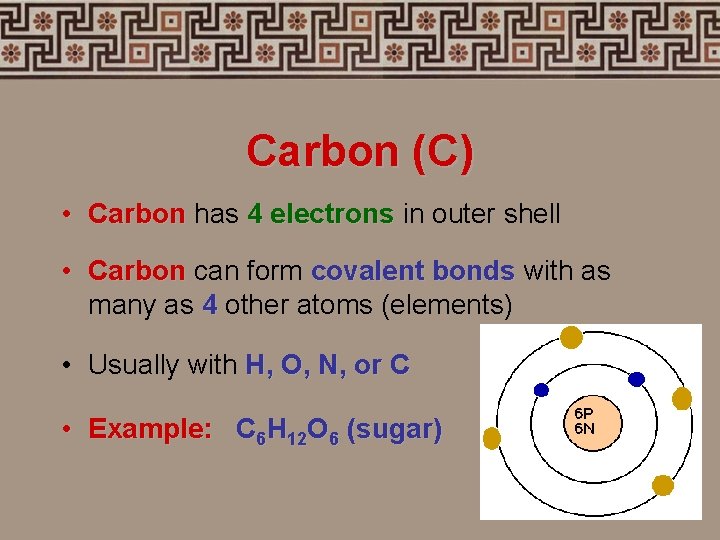
Carbon (C) • Carbon has 4 electrons in outer shell • Carbon can form covalent bonds with as many as 4 other atoms (elements) • Usually with H, O, N, or C • Example: C 6 H 12 O 6 (sugar)

Macromolecules • Large organic molecules. • Also called POLYMERS • Made up of smaller “building blocks” called MONOMERS • Examples: 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic acids (DNA and RNA)

1. Carbohydrates • Small sugar molecules to large sugar molecules • Examples: A. monosaccharide B. disaccharide C. polysaccharide

1. Carbohydrates A. monosaccharide: one sugar unit • Examples: glucose (C ( 6 H 12 O 6 blood sugar) deoxyribose galactose (milk sugar) glucose fructose (honey)
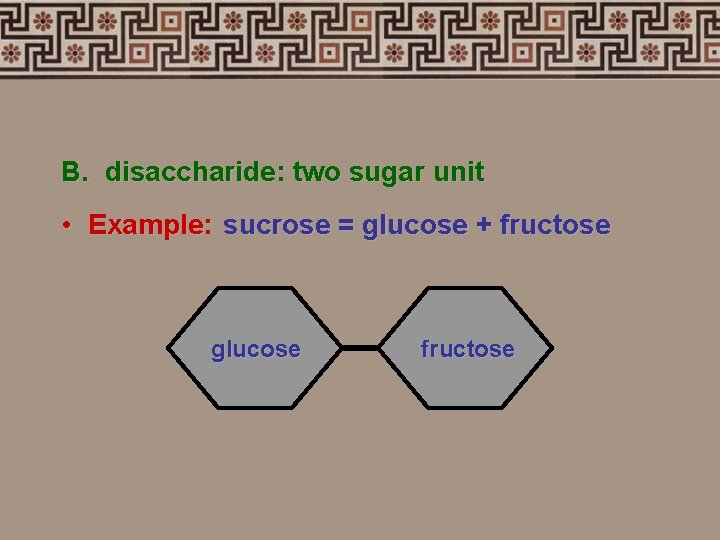
B. disaccharide: two sugar unit • Example: sucrose = glucose + fructose glucose fructose
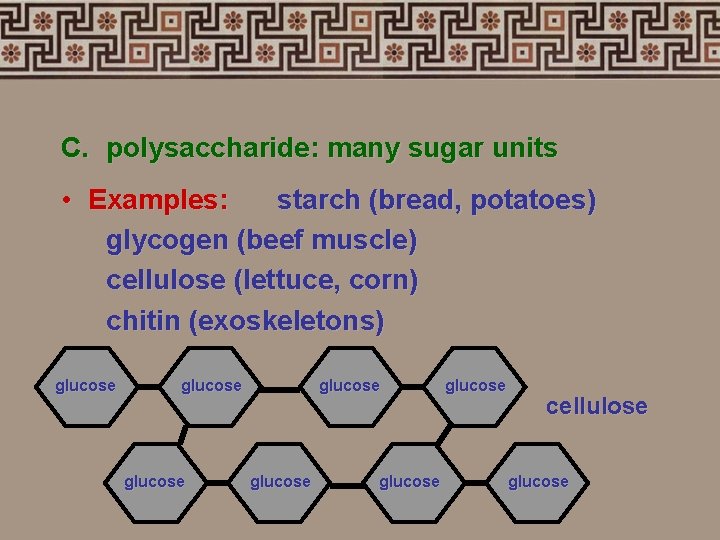
C. polysaccharide: many sugar units • Examples: starch (bread, potatoes) glycogen (beef muscle) cellulose (lettuce, corn) chitin (exoskeletons) glucose glucose cellulose glucose

• The primary functions of carbohydrate macromolecules are to: – provide and store energy.

2. Lipids • General term for compounds which are not soluble in water • Lipids are soluble in hydrophobic solvents • Remember: “stores the most energy”

2. Lipids • Examples: 1. 2. 3. 4. 5. 6. Fats Phospholipids Oils Waxes Steroid hormones Triglycerides

• 5 functions of lipids: 1. Long term energy storage (fat) 2. Protection against heat loss (insulation) 3. Protection against water loss & germs (oils & waxes) 4. Chemical messengers (hormones & steroids) 5. Major component of membranes (phospholipids)
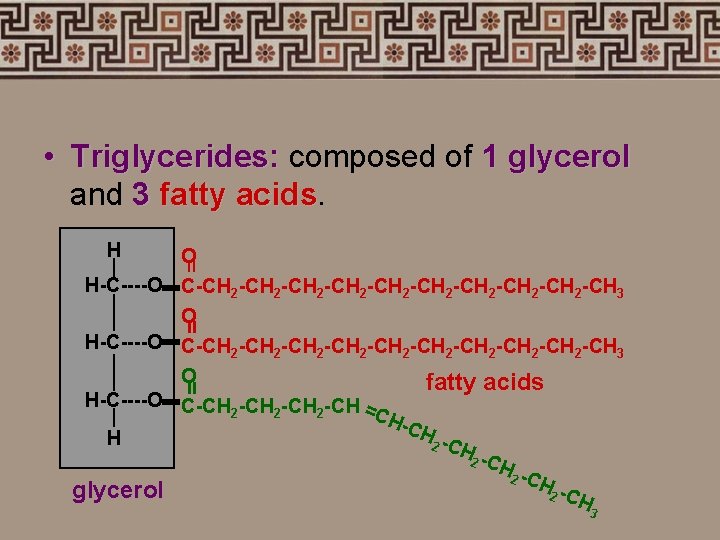
• Triglycerides: composed of 1 glycerol and 3 fatty acids H = O H-C----O C-CH 2 -CH 2 -CH 2 -CH 2 -CH 2 -CH 3 O fatty acids H-C----O C-CH -CH = 2 2 2 CH -CH H 2 -CH glycerol 2 -CH = = 3

• There are two kinds of fatty acids you may see on food labels: 1. Saturated fatty acids: no double bonds (bad) 2. Unsaturated fatty acids: double bonds (good) = O C-CH 2 -CH 2 -CH 2 -CH 3 saturated = unsaturated O C-CH 2 -CH =C H-C H 2 CH 2 -CH 3

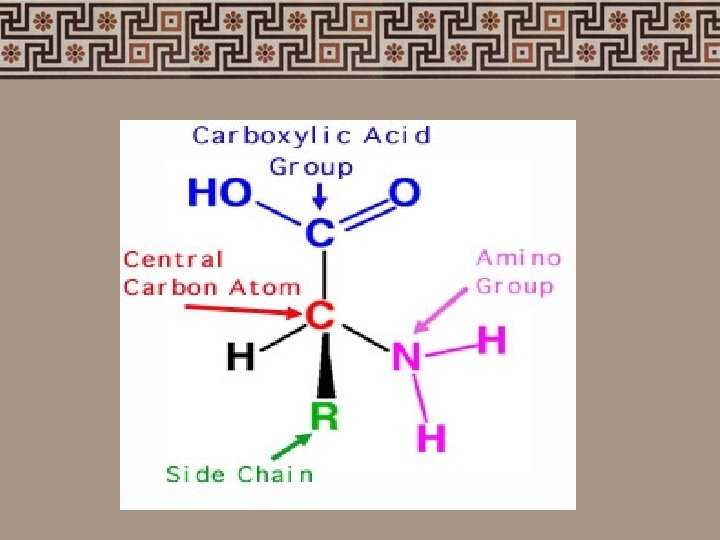
3. Proteins (Polypeptides) • Amino acids (the building blocks of protein) • 2 kinds of amino acids - essential & non-essential amino acids - Essential amino acids cannot be synthesized by our body & need to be obtained through our diet

• 7 functions of proteins: 1. 2. 3. 4. 5. 6. 7. Storage: Transport: Regulatory: Movement: Structural: Enzymes: Defense: albumin (egg white) hemoglobin hormones muscles membranes, hair, nails cellular reactions antibodies

• A protein’s structure depends on its specific job • The sequence of amino acids and the shape of the chain are a consequence of attractions between the chain’s parts.

• Four levels of protein structure: A. Primary Structure (1°) B. Secondary Structure (2°) C. Tertiary Structure (3°) D. Quaternary Structure (4°)
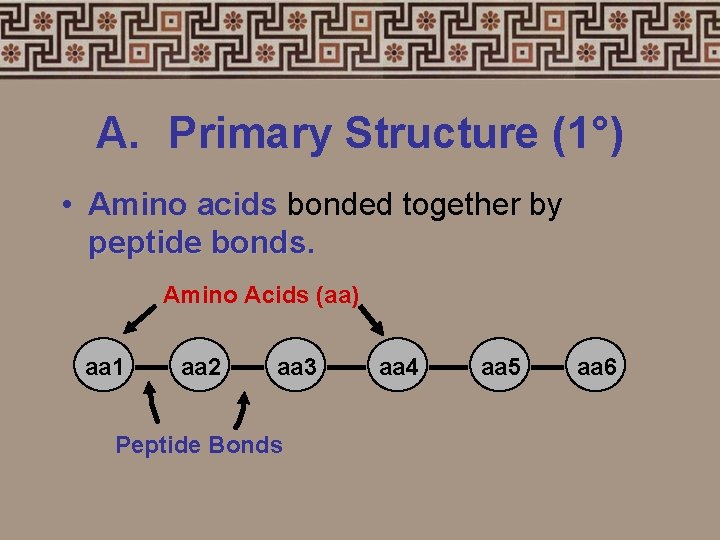
A. Primary Structure (1°) • Amino acids bonded together by peptide bonds. Amino Acids (aa) aa 1 aa 2 aa 3 Peptide Bonds aa 4 aa 5 aa 6

B. Secondary Structure (2°) • 3 -dimensional folding arrangement of a primary structure into coils and pleats held together by hydrogen bonds
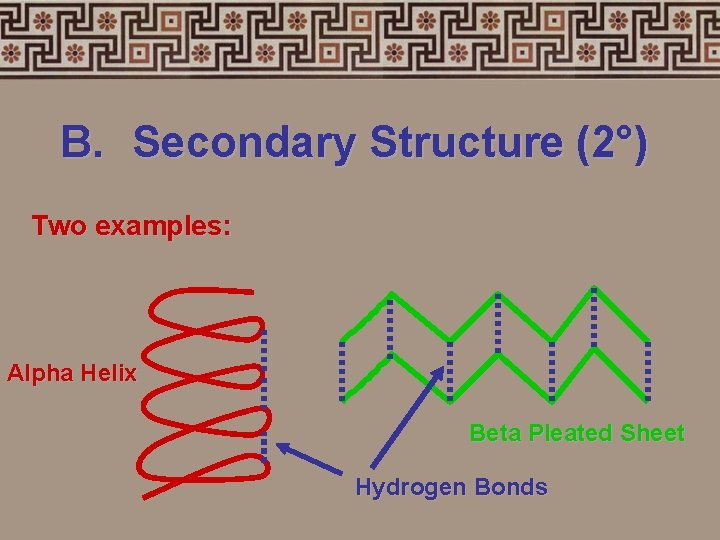
B. Secondary Structure (2°) Two examples: Alpha Helix Beta Pleated Sheet Hydrogen Bonds
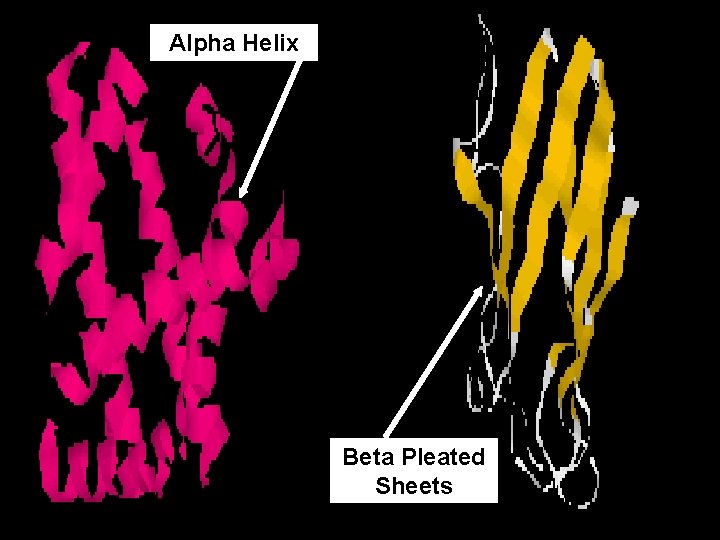
Alpha Helix Beta Pleated Sheets

C. Tertiary Structure (3°) • Secondary structures bend and fold into a more complex 3 -D arrangement • Called a “subunit”.
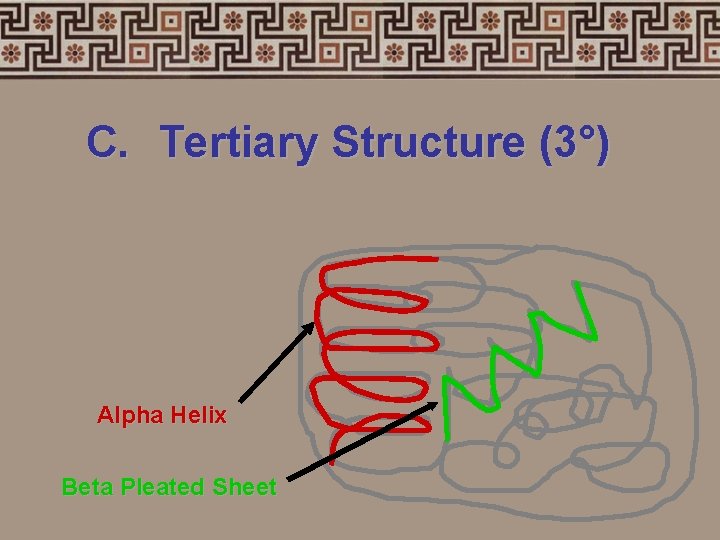
C. Tertiary Structure (3°) Alpha Helix Beta Pleated Sheet
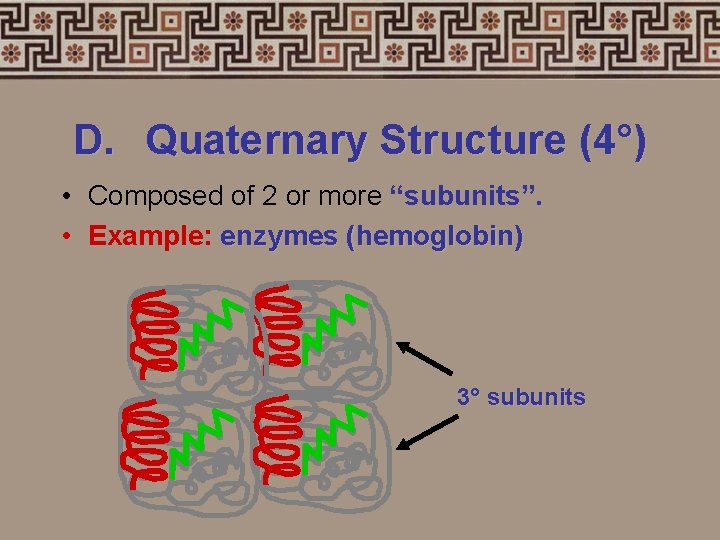
D. Quaternary Structure (4°) • Composed of 2 or more “subunits”. • Example: enzymes (hemoglobin) 3° subunits
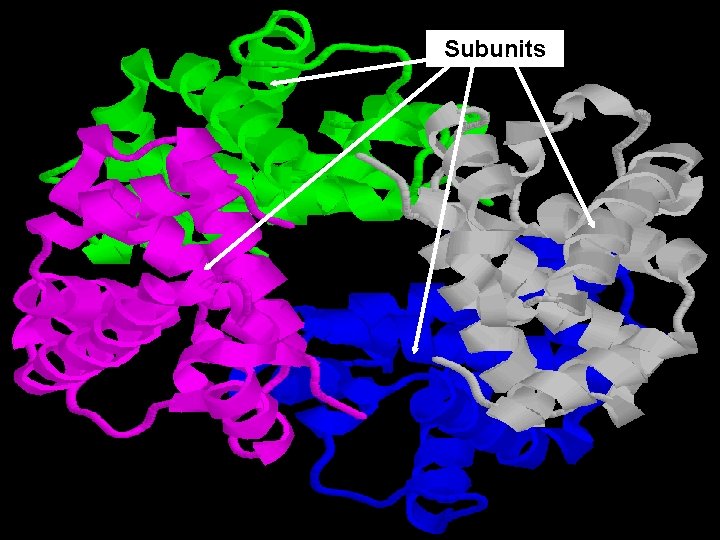
Subunits
Enzymes: Special Proteins
4. Nucleic acids • Nucleic acids (DNA and RNA) control cell activities by controlling protein synthesis

4. Nucleic acids • Two types: 1. deoxyribonucleic acid (DNA-double helix) 2. ribonucleic acid (RNA-single strand) • Nucleic acids are composed of long chains of nucleotides

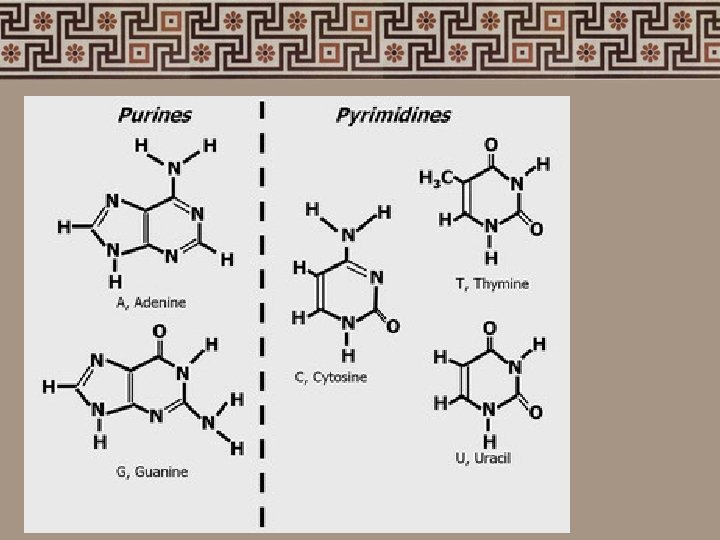
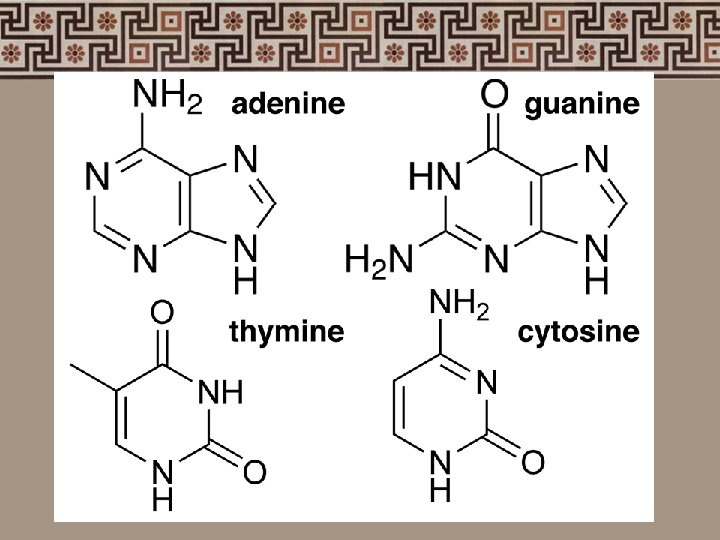
4. Nucleic acids • Nucleotides: phosphate group sugar (5 -carbon) nitrogenous bases adenine (A) thymine (T) - uracil (U) RNA cytosine (C) guanine (G)
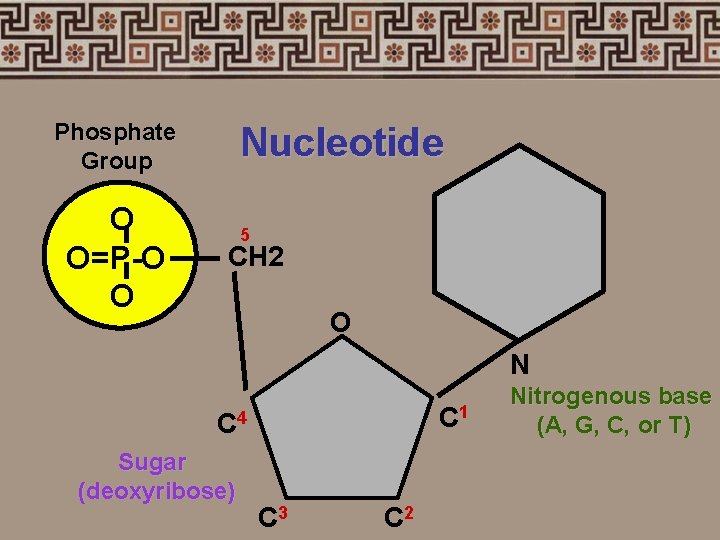
Phosphate Group O O=P-O O Nucleotide 5 CH 2 O N C 1 C 4 Sugar (deoxyribose) C 3 C 2 Nitrogenous base (A, G, C, or T)
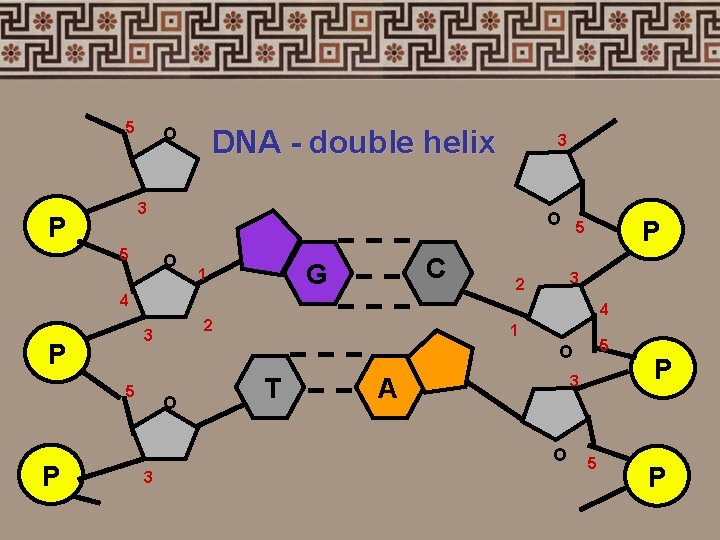
5 O DNA - double helix 3 P 5 O C G 1 4 5 3 2 1 5 O O T 3 A O 3 P 4 2 3 P P 3 5 P P

2017
- Slides: 43