BIOCHEMISTRY Organic Molecules Molecules that have a carbon

BIOCHEMISTRY

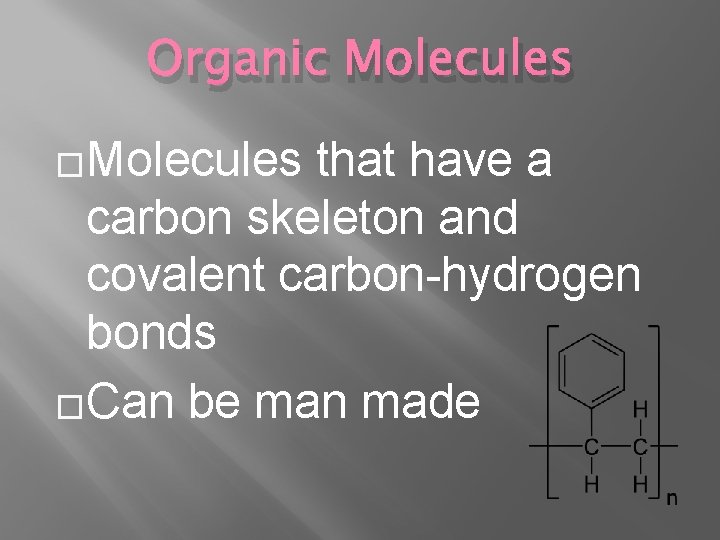
Organic Molecules �Molecules that have a carbon skeleton and covalent carbon-hydrogen bonds �Can be man made
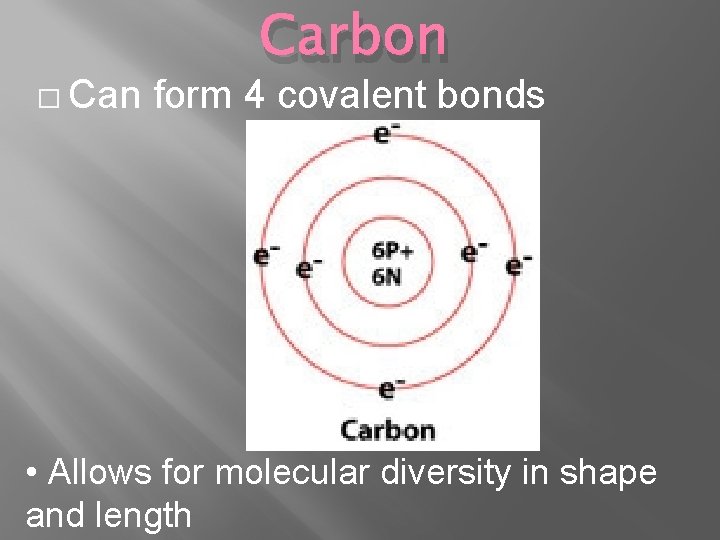
� Can Carbon form 4 covalent bonds • Allows for molecular diversity in shape and length
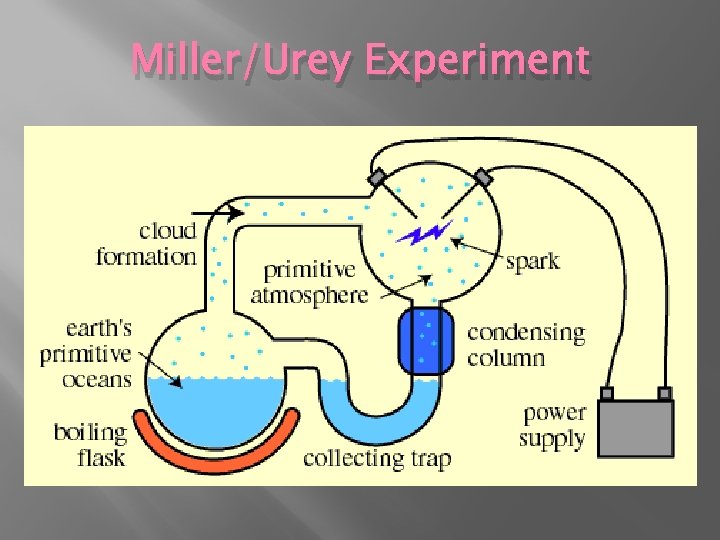
Miller/Urey Experiment

Review of Functional Groups � ID the various functional groups in the following molecules…
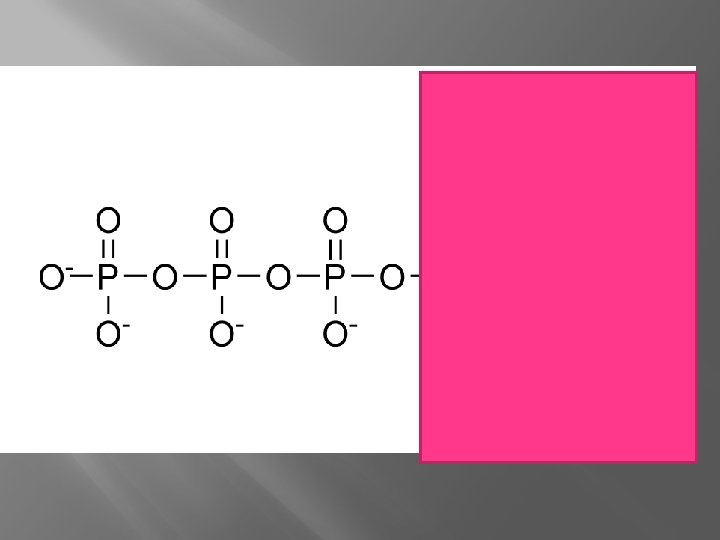
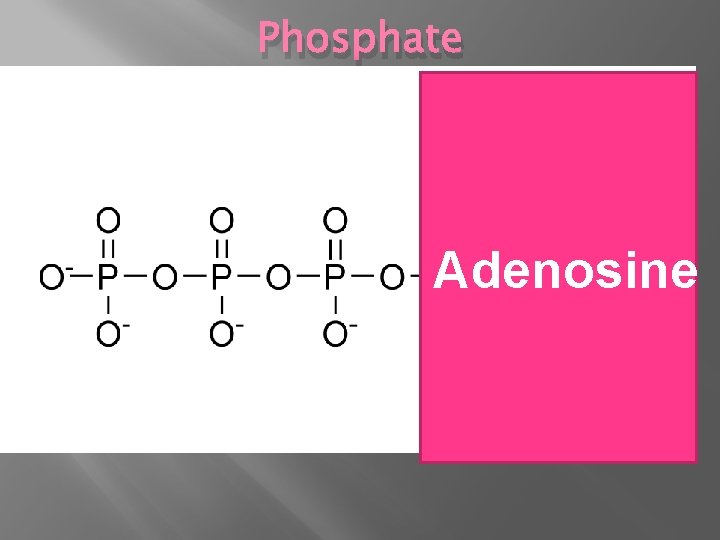
Phosphate Adenosine
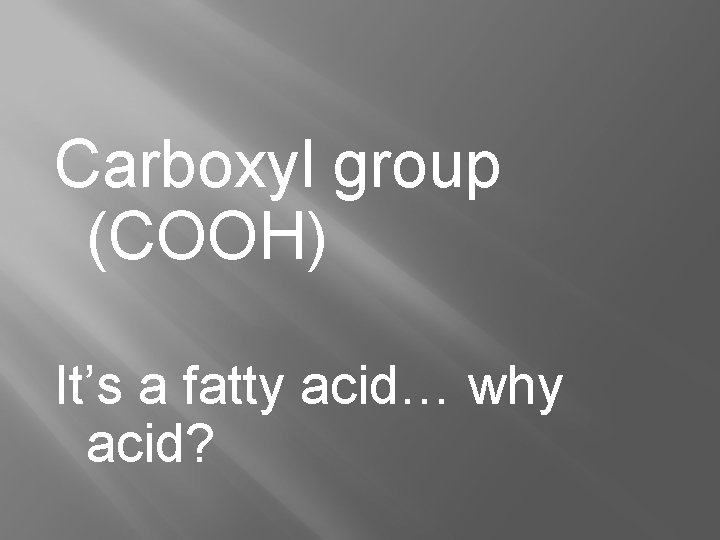
Carboxyl group (COOH) It’s a fatty acid… why acid?
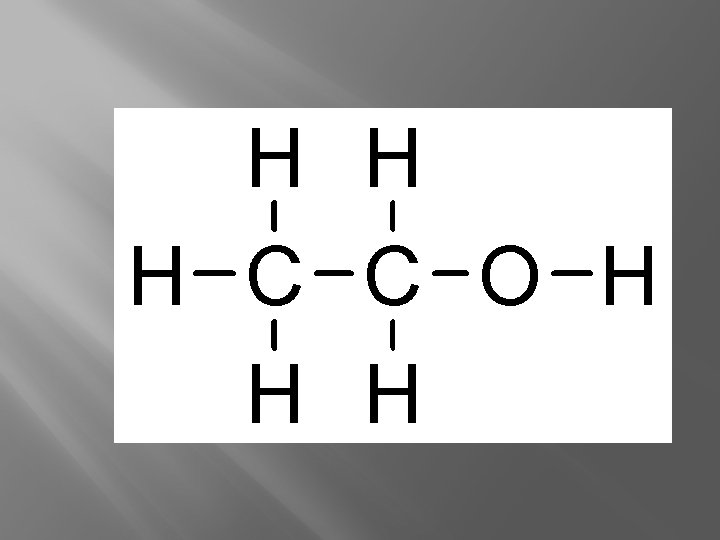

Ethanol… alcohol in adult beverages OH… looks like ____. . . Alcohol can make you do these types of things…
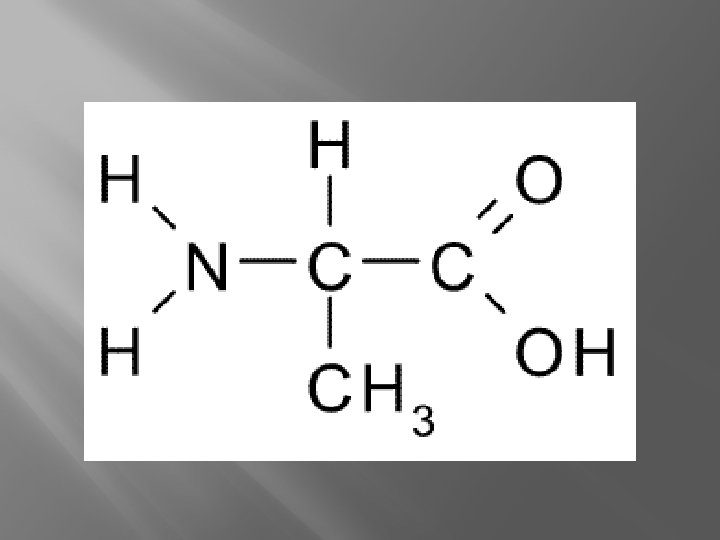

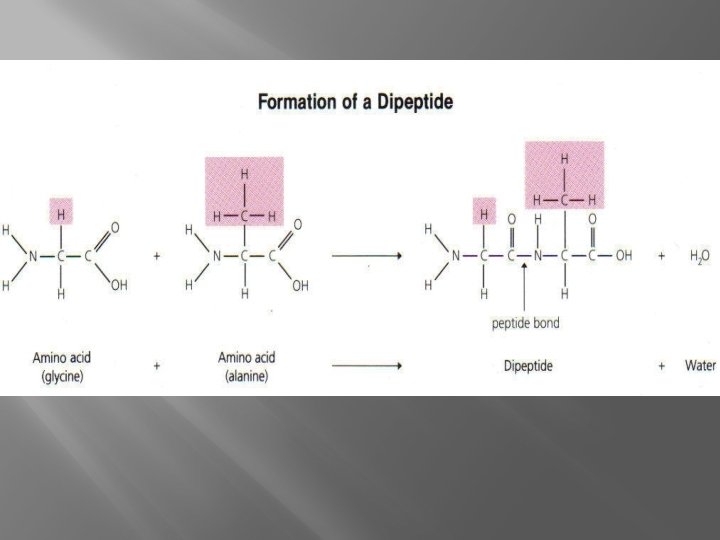
Alanine Amino NH 2 Carboxyl COOH (think about what this is going to do when in a solution) What is alanine an example of? There are 20 of them.
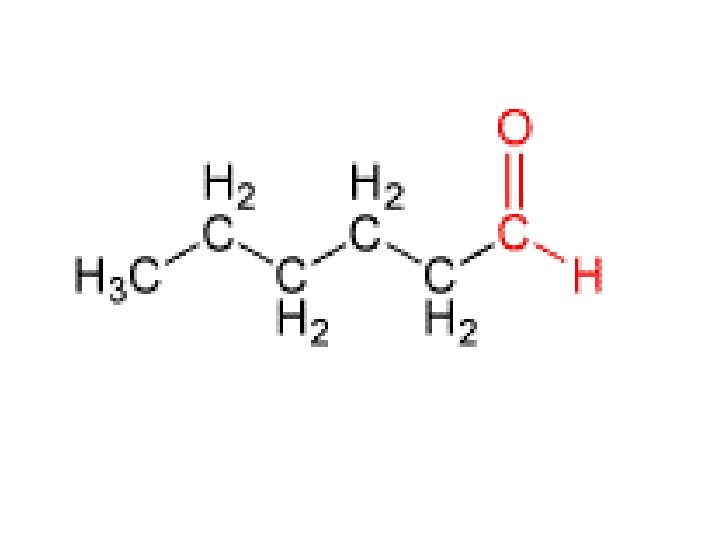

Aldehyde �Think… formaldehyde stinks… put it on the end! �Ketone trick?

Proteins �More than 50% of the dry weight of an organism �Made of smaller subunits (monomers) amino acids
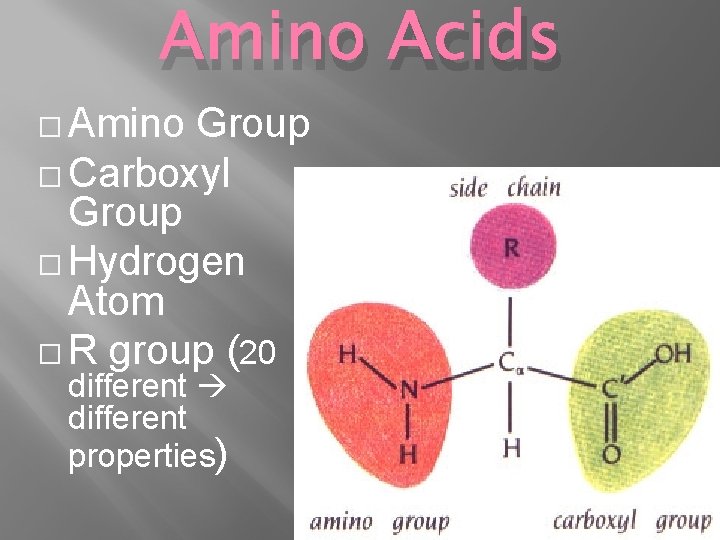
Amino Acids � Amino Group � Carboxyl Group � Hydrogen Atom � R group (20 different properties)
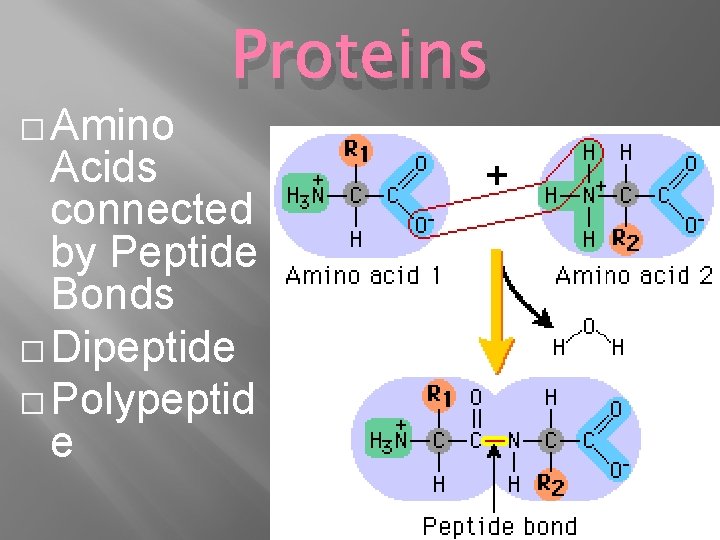
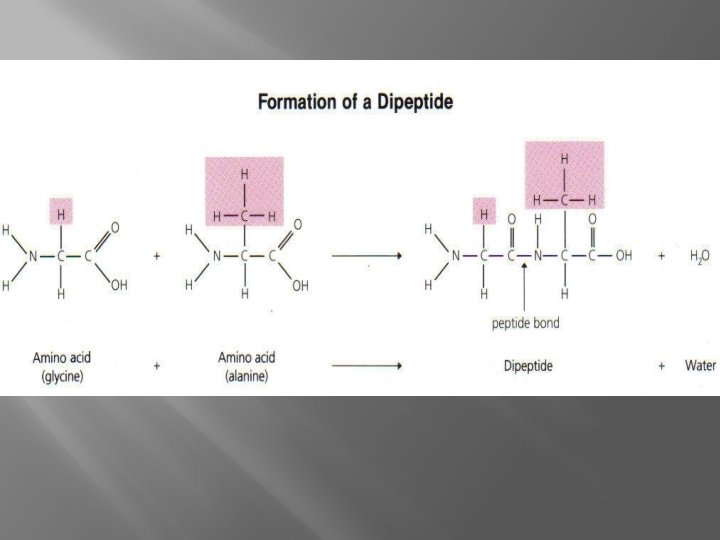
� Amino Proteins Acids connected by Peptide Bonds � Dipeptide � Polypeptid e
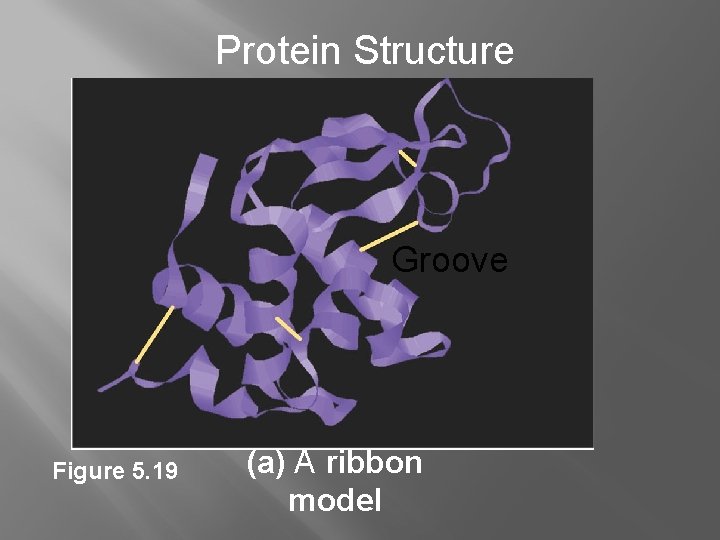
Protein Structure Groove Figure 5. 19 (a) A ribbon model

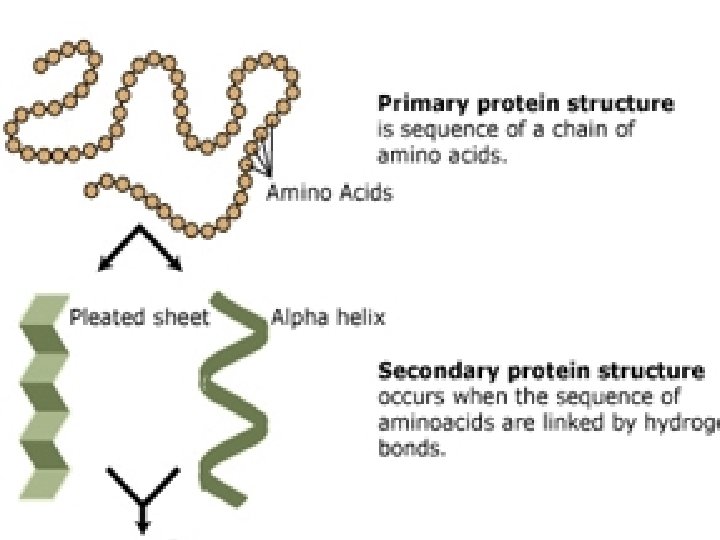
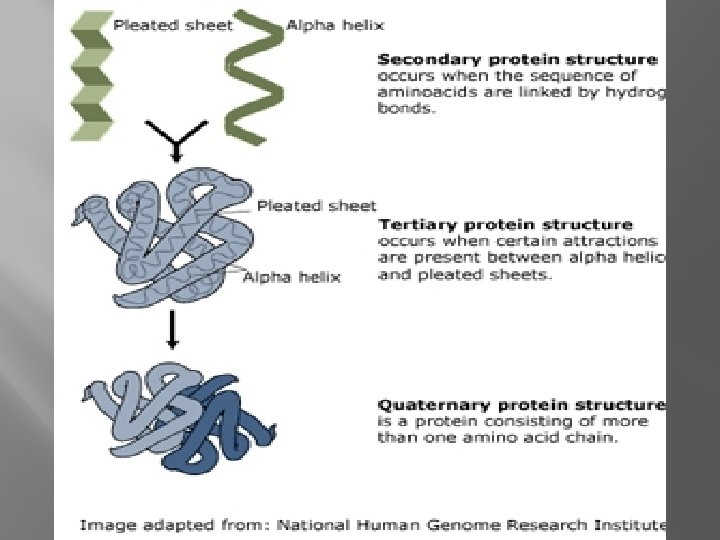
Protein Structure � Primary- Chain of amino acids � Secondary- Due to H-Bonds between amino acids

Protein Structure � Tertiary- “Active” Shape �H-Bonds between R-groups �Ionic Bonds between R- groups �Nonpolarity (pushed together by water) �Sulfur bridges (covalent bond between 2 cysteines) �Van der waals attractions

Protein Structure �Quarternary �Joining Structure of two or more polypeptide chains
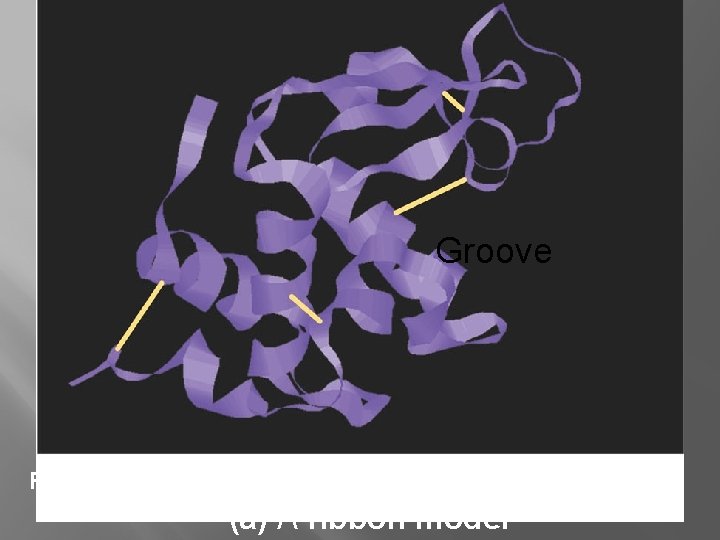
Groove Figure 5. 19 (a) A ribbon model

1. 2. 3. 4. Directions Pick up an amino acid sequence, scissors, and tape Grad JUST the amino acids you will need from the back and 9 water molecules (they’re in sheets of 13 so cut them apart) Assemble your amino acids showing what must be removed to put your amino acids together ANSWER THE FOLLOWING 1/GROUP (put both names on)

Questions to turn in at the end of the period 1. 2. 3. 4. 5. What is the polymer that is created when amino acids are bonded together? What 2 functional groups are present on EVERY amino acid (name & draw) If all amino acids share 2 functional groups then why are they different? EXPLAIN. What reaction must occur in order to break a chain of amino acids into individual amino acids? What must happen to a chain of amino acids to make it a protein?

Fig. 5. 1 Shows all the functions proteins can have:

Groups � Catalysts- Enzymes speed up rates of chem. rxns � Structural- for support; hair, silk, connective tissue � Storage- source of amino acids for young

Groups �Transport- of other substances �Hormonal- coordinates and directs regulation of organisms �Receptors- decide what comes in/out of cells

Groups �Transcription factors- tells cells what to make �Motor- make up muscles, cilia, flagella, etc �Defensive- antibodies

Enzymes �Organic Catalysts �Lower Activation Energy �May need a coenzyme in order to function
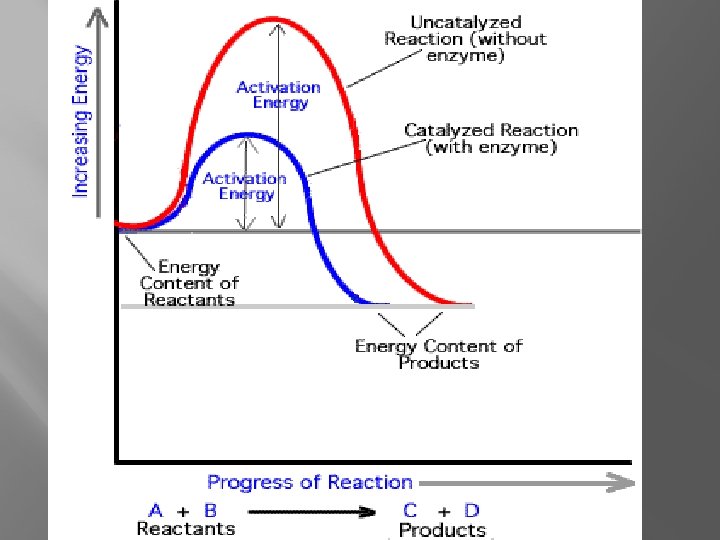

Factors affecting enzymes �Do the Lab!!!

Isomers �Isomers- Molecules with same atoms but different arrangement and properties
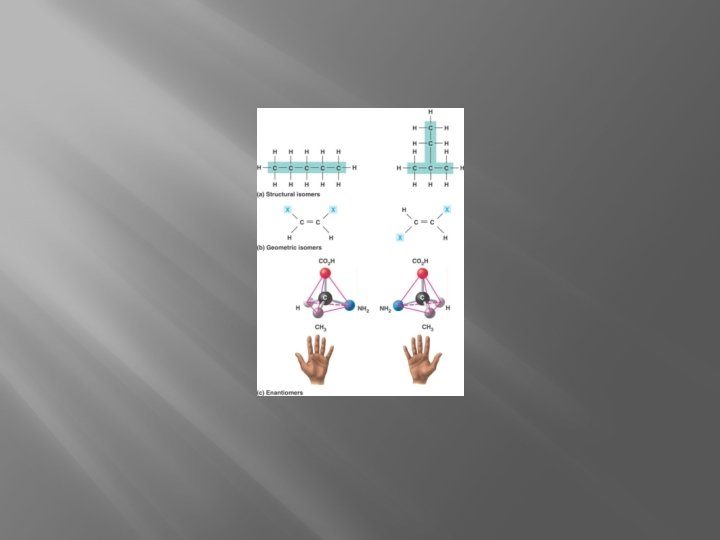
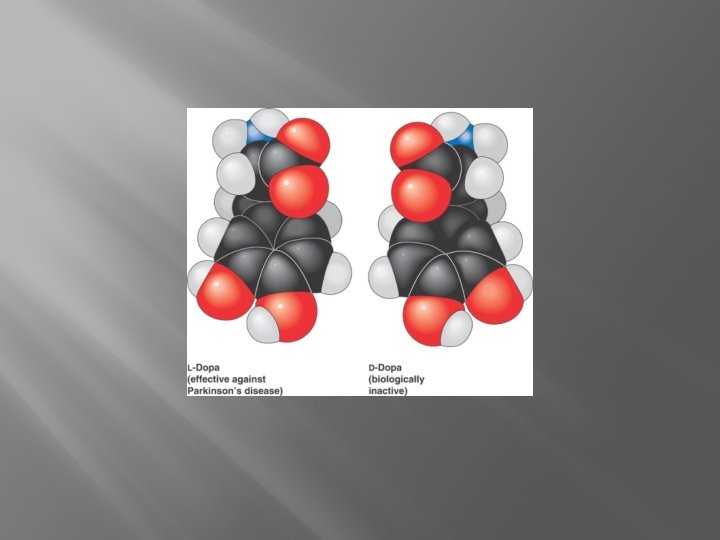

Functional Groups �Groups of atoms that give a molecule specific properties

Hydroxyl � Alcohols
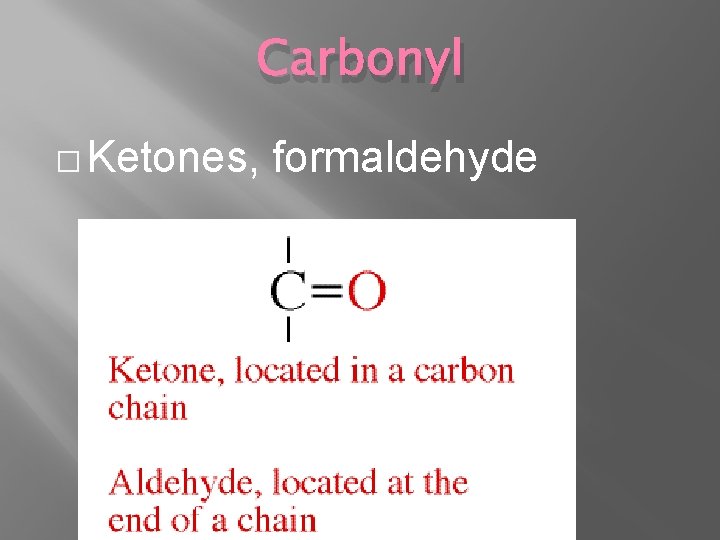
Carbonyl � Ketones, formaldehyde

Carboxyl � “Organic” Acids

Amino � Amino Acids

Sulfhydryl � Found in proteins
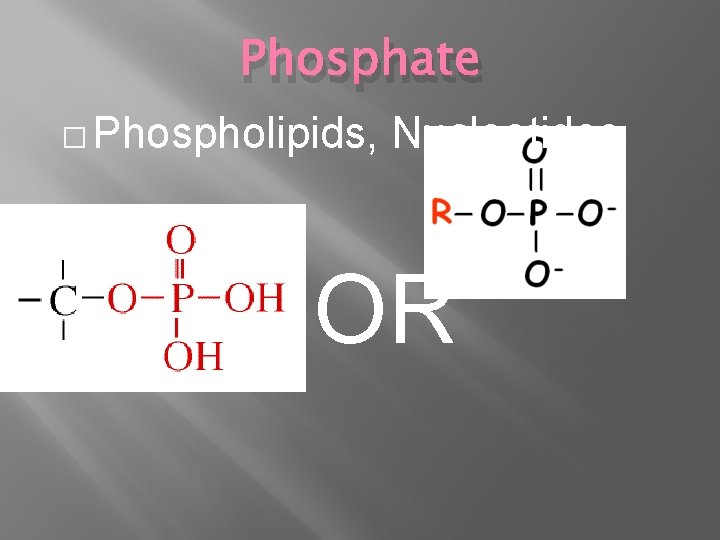
Phosphate � Phospholipids, Nucleotides OR

Building Macromolecules �Monomers- Small organic molecules that can be linked together �Polymers- Big things (monomers linked together)

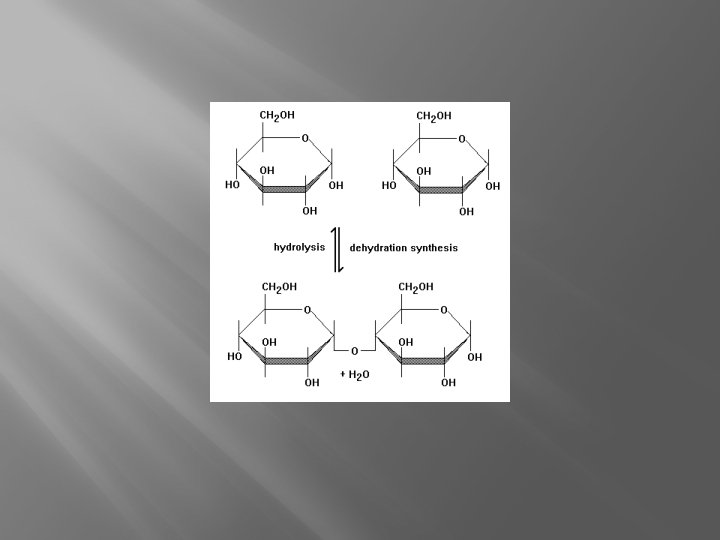
Building � Condensation (dehydration synthesis): connects 2 molecules. � One molecule loses an H, the other loses an OH � One water molecule is formed

Taking Apart �Hydrolysis- Breaking down a polymer �Requires a water molecule

Four Main Classes �Carbohydrates �Lipids �Proteins �Nucleic Acids

Carbohydrates �Sugars and their related compounds �Oxygen and Hydrogen in a 1: 2 ratio �Energy stores or structure �Most end in -ose

Monosaccharide � Simple Sugars � Contain 3, 4, 5, or 6 carbon atoms
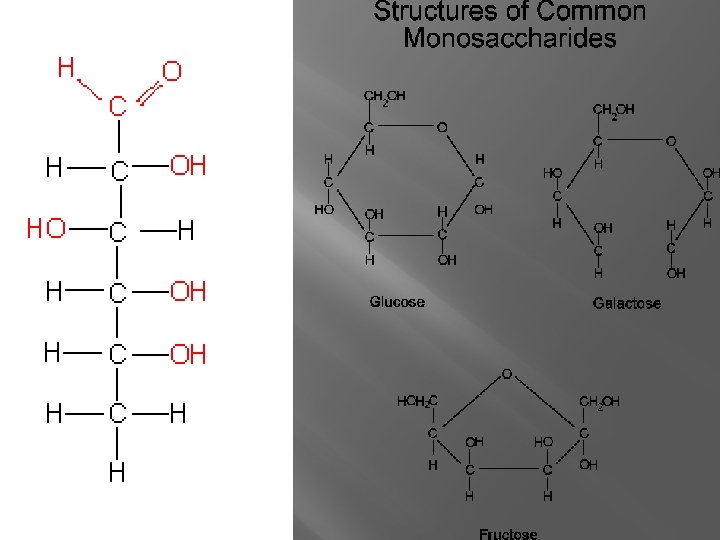

Monosaccharide �Deoxyribose Five carbons In Nucleic Acids �Glucose Six carbons Energy

Disaccharides � Two monosaccharides � Ex. lactose and sucrose

Polysaccharide � Glycogen- Energy storage in animals � Starch- Energy storage in plants � Cellulose- Cell walls in plants � Chitin- Amino Sugars used in exoskeletons
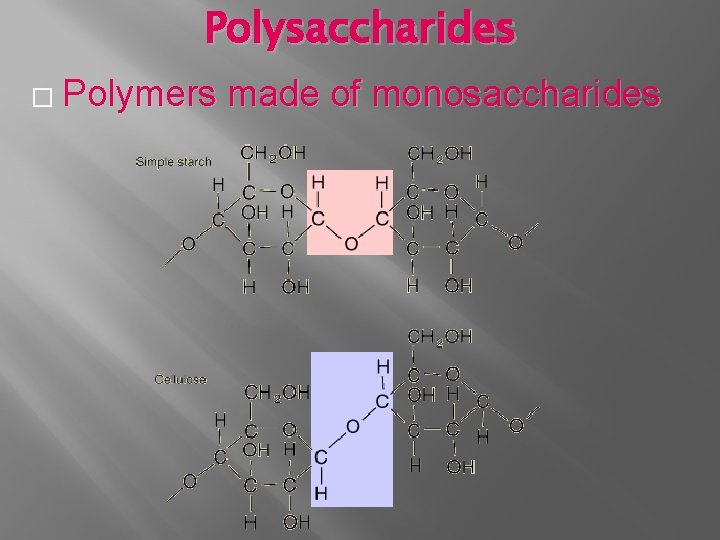
Polysaccharides � Polymers made of monosaccharides

Lipids �Nonpolar �Mostly carbon and hydrogen �Excellent in storing energy �Also used in membranes and waterproof surfaces
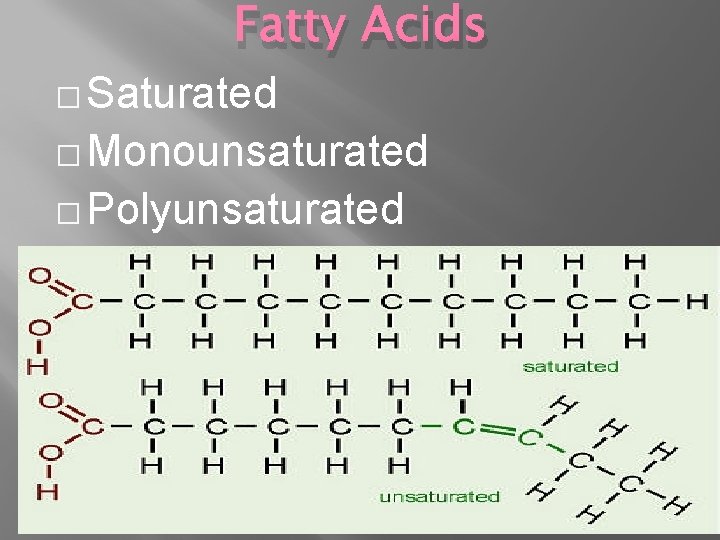
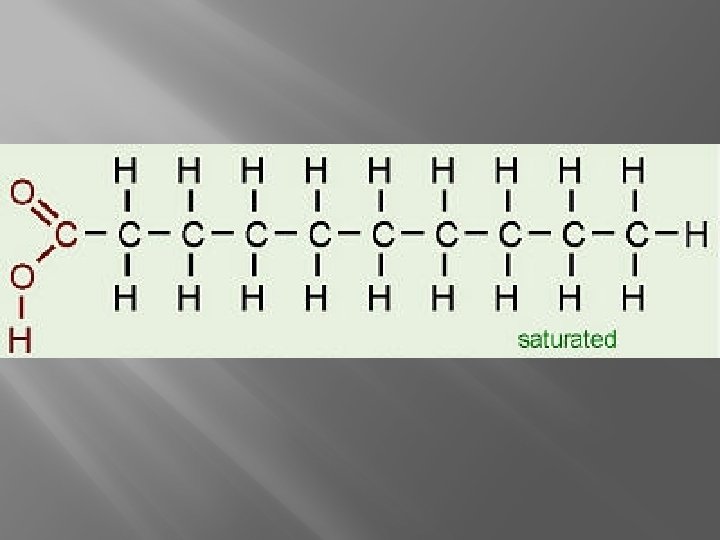
Fatty Acids � Saturated � Monounsaturated � Polyunsaturated

Triglycerides �Three Fatty Acids PLUS One Glycerol �Fats- have mostly saturated fatty acids (can be packed closer, so they are solid) �Oils- Unsaturated fatty acids (kinks make them liquid)

Triglycerides

Wax � One long fatty acid connected to one long alcohol � Waterproof

Steroids ? � Some Hormones � In Cell Membranes

Phospholipids � Two Fatty Acid Tails AND One Phosphat e Group

Phospolipids � Tails- Hydrophobic (water hating) � Head. Hydrophilic (water loving)

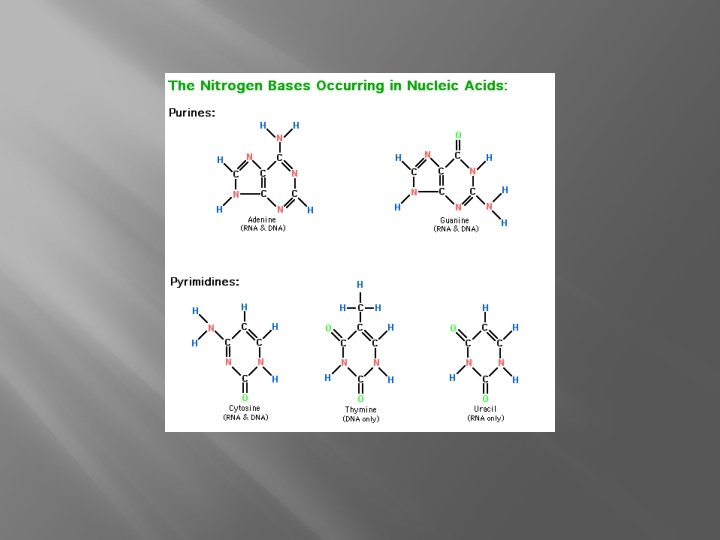
Nucleic Acids � DNA and RNA � Made of nucleotides �Pentose sugar �Phosphate group �Nitrogen base (A, T, G, C, U)

Nitrogen Bases �Purines (2 rings): Adenine and Guanine �Pyrimidines (1 ring): Cytosine, Thymine, Uracil
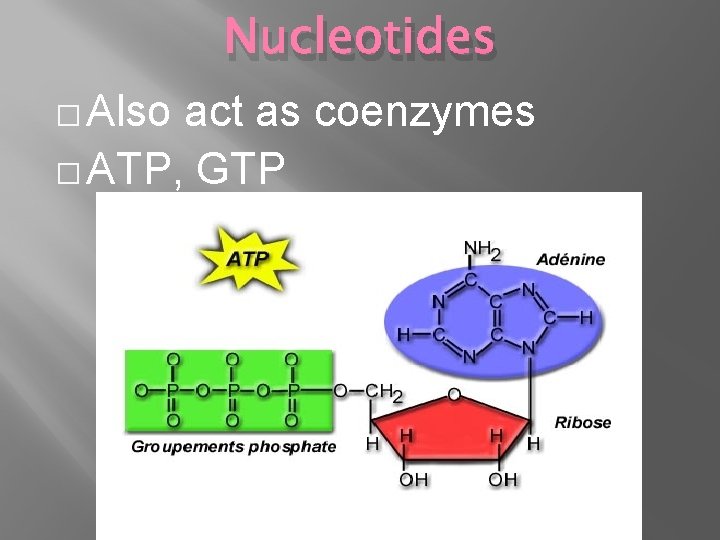
Nucleotides � Also act as coenzymes � ATP, GTP

Proteins �More than 50% of the dry weight of an organism �Made of amino acids
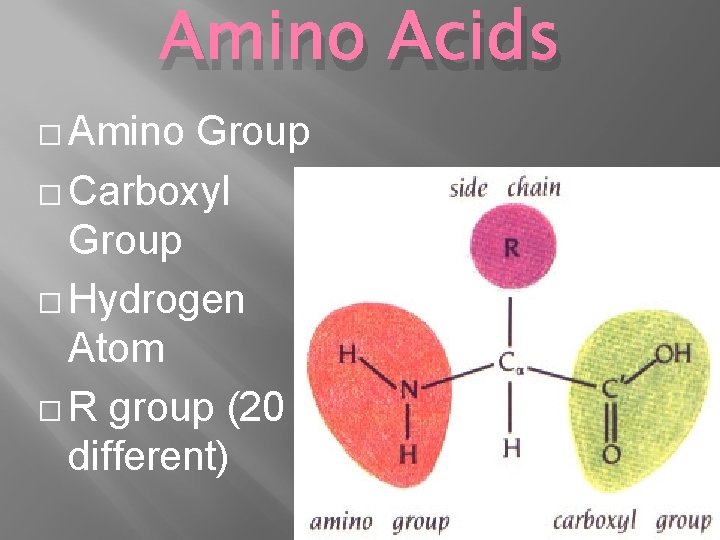
Amino Acids � Amino Group � Carboxyl Group � Hydrogen Atom � R group (20 different)
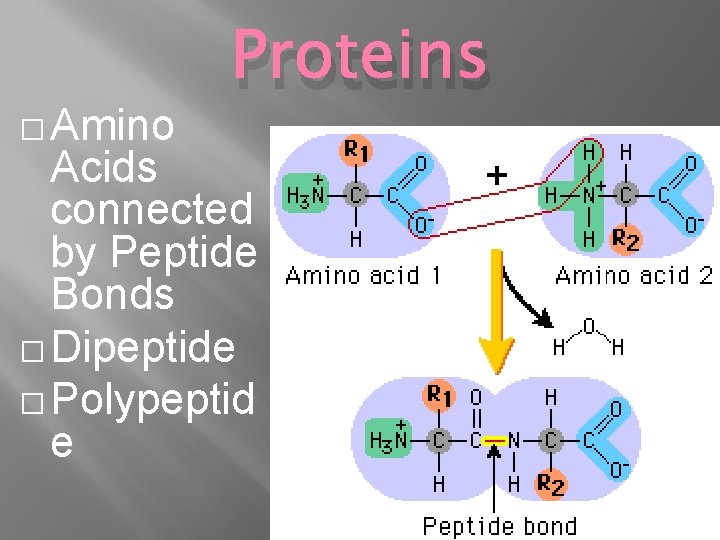
� Amino Proteins Acids connected by Peptide Bonds � Dipeptide � Polypeptid e

Protein Structure � Primary- Chain of amino acids � Secondary- Due to H-Bonds between amino acids


Protein Structure � Tertiary- “Active” Shape �H-Bonds between R-groups �Ionic Bonds between R- groups �Nonpolarity (pushed together by water) �Sulfur bridges (covalent bond between 2 cysteines) �Van der waals attractions

Protein Structure �Quarternary �Joining Structure of two or more polypeptide chains


Groups �Catalysts- Enzymes �Structural- for support; hair, silk, connective tissue �Storage- source of amino acids for young

Groups �Transport- of other substances �Hormonal- coordinates and directs regulation of organisms �Receptors- decide what comes in/out of cells

Groups �Transcription factors- tells cells what to make �Motor- make up muscles, cilia, flagella, etc �Defensive- antibodies

Enzymes �Organic Catalysts �Lower Activation Energy �May need a coenzyme in order to function
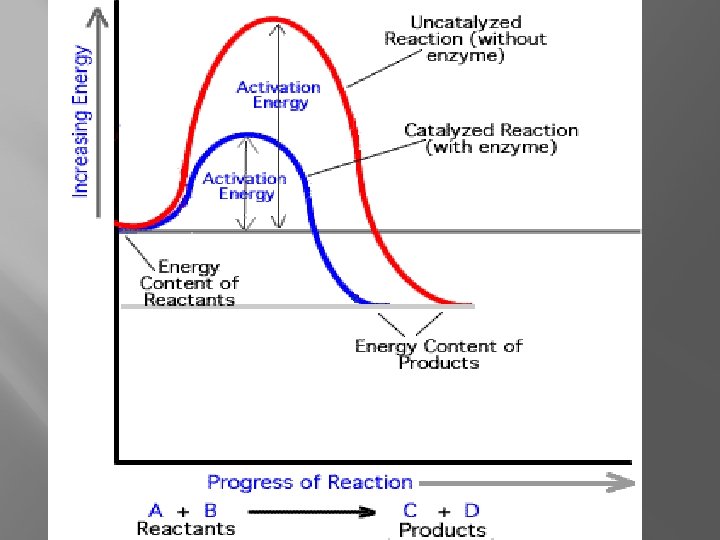


Factors affecting enzymes �Do the Lab!!!
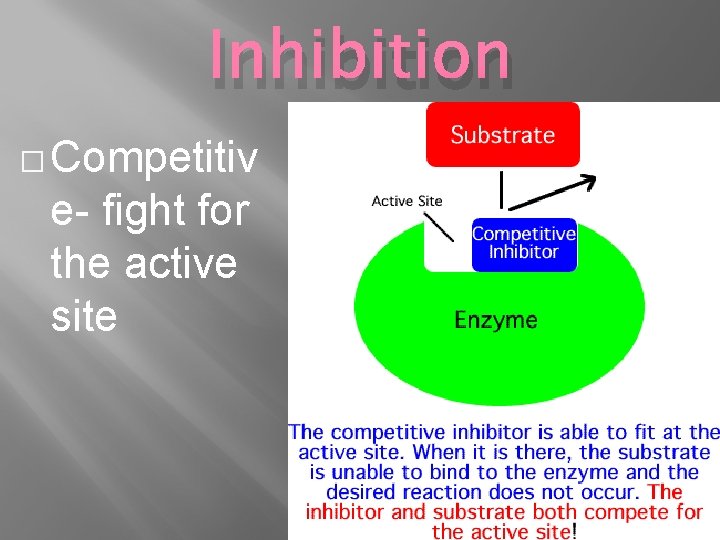
Inhibition � Competitiv e- fight for the active site

Inhibition �Noncompetitive- inhibitor binds somewhere else (allosteric site) �Allosteric Enzyme- active site changes
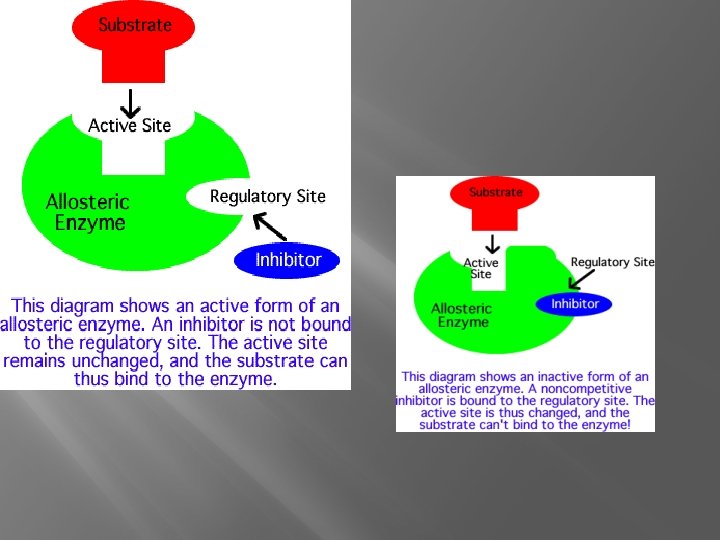

Why? �Control Metabolism �Feedback �On/Off Switches
- Slides: 90