Biochemistry of neurodegenerative diseases and prions Alice Skoumalov

Biochemistry of neurodegenerative diseases and prions Alice Skoumalová
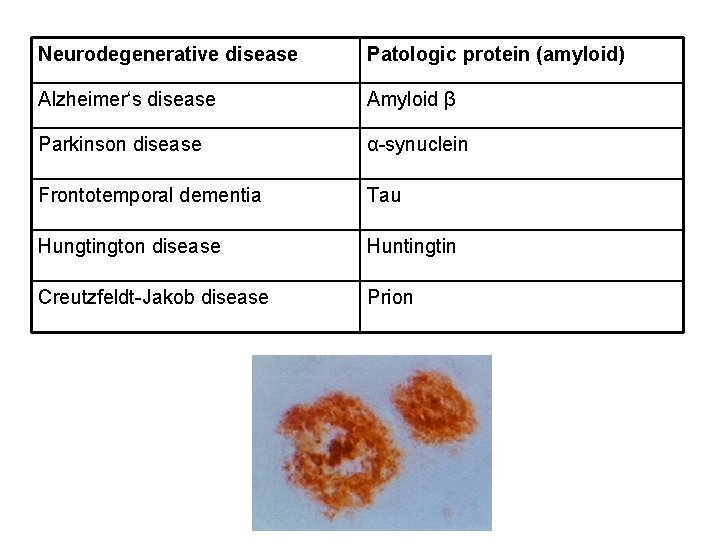
Neurodegenerative disease Patologic protein (amyloid) Alzheimer‘s disease Amyloid β Parkinson disease α-synuclein Frontotemporal dementia Tau Hungtington disease Huntingtin Creutzfeldt-Jakob disease Prion
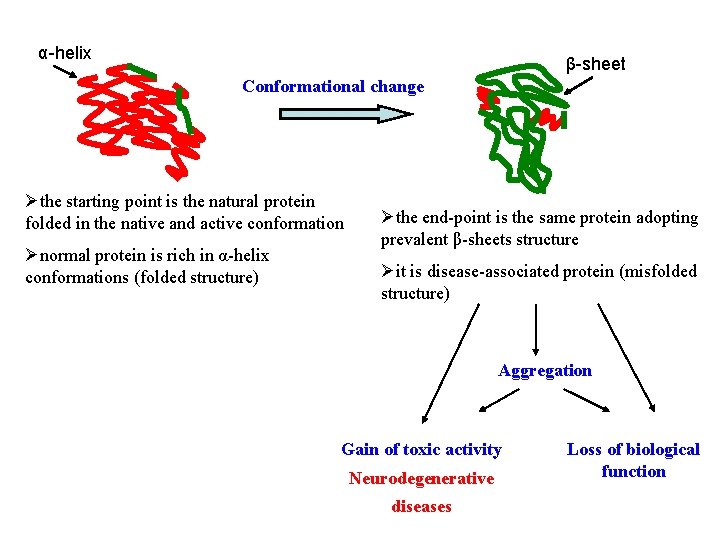
α-helix β-sheet Conformational change Øthe starting point is the natural protein folded in the native and active conformation Ønormal protein is rich in α-helix conformations (folded structure) Øthe end-point is the same protein adopting prevalent β-sheets structure Øit is disease-associated protein (misfolded structure) Aggregation Gain of toxic activity Neurodegenerative diseases Loss of biological function
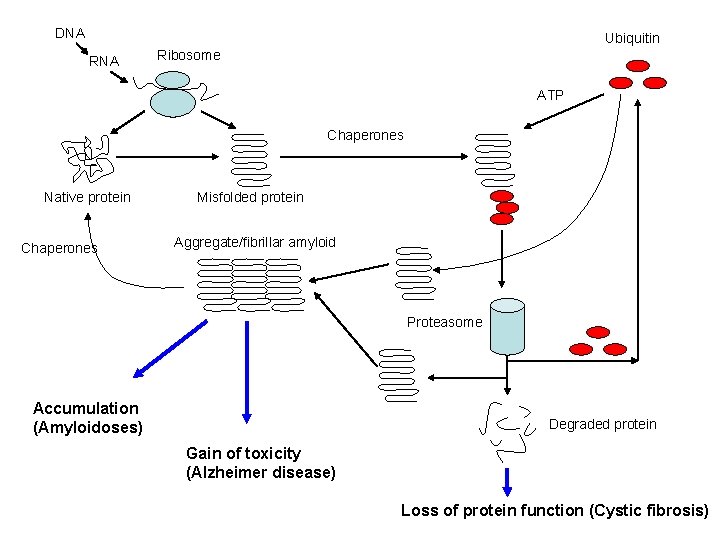
DNA Ubiquitin RNA Ribosome ATP Chaperones Native protein Chaperones Misfolded protein Aggregate/fibrillar amyloid Proteasome Accumulation (Amyloidoses) Degraded protein Gain of toxicity (Alzheimer disease) Loss of protein function (Cystic fibrosis)
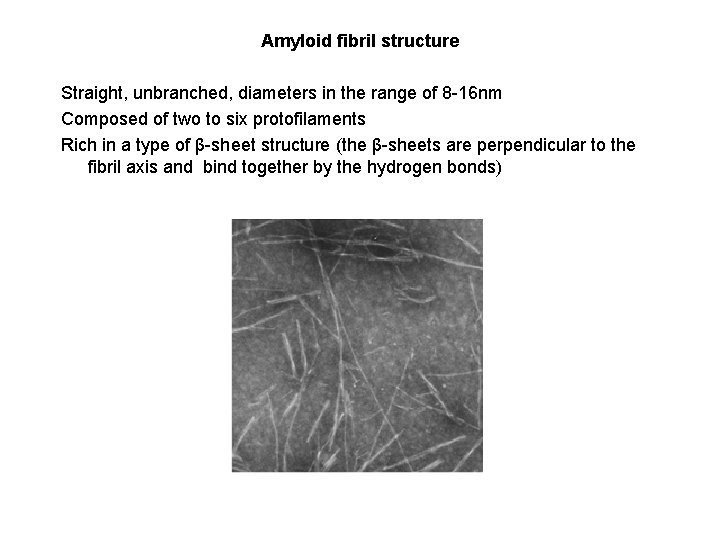
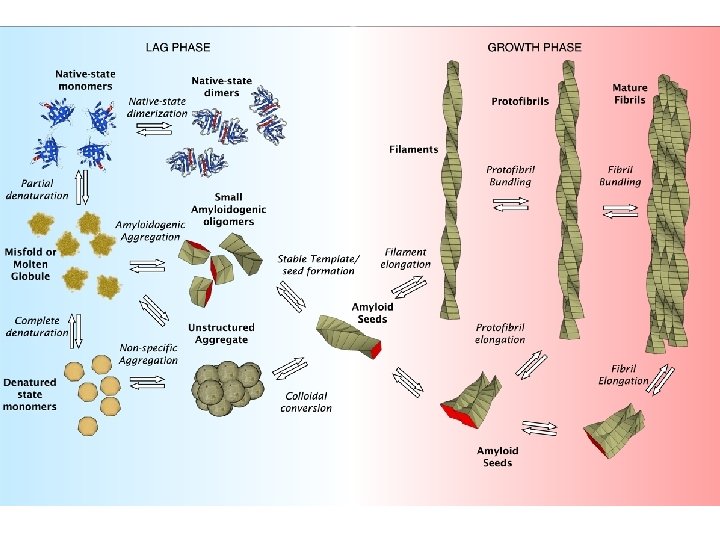
Amyloid fibril structure Straight, unbranched, diameters in the range of 8 -16 nm Composed of two to six protofilaments Rich in a type of β-sheet structure (the β-sheets are perpendicular to the fibril axis and bind together by the hydrogen bonds)

Molecular factors in amyloid formation Protein misfolding is central to amyloid formation Protein stability- the resistance of the folded conformation to misfolding- is an important factor in determining susceptibility to amyloid formation Destabilizing factors: 1. Extreme environments in the body, such as acidic cell compartments 2. Proteolytic removal of a portion of a protein by an endogenous protease 3. Mutations that alter the primary structure (many of the amyloid diseases involve amino acid substitutions in an amyloid precursor protein) 4. Interactions with lipid bilayers (Aβ)
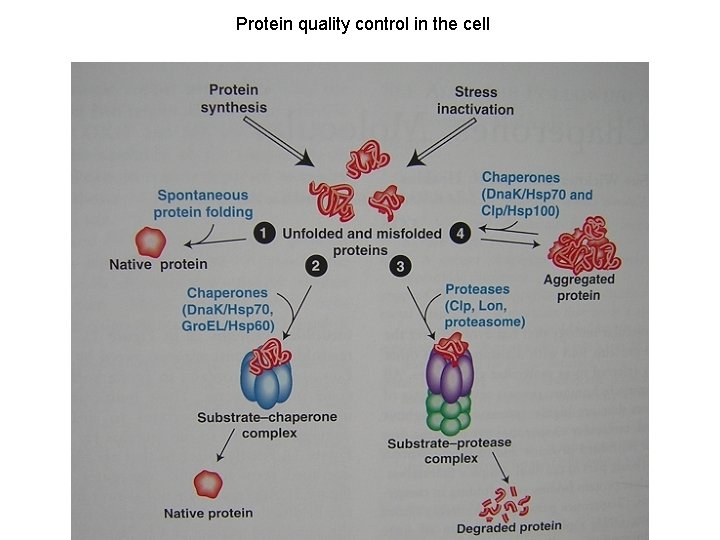
Protein quality control in the cell
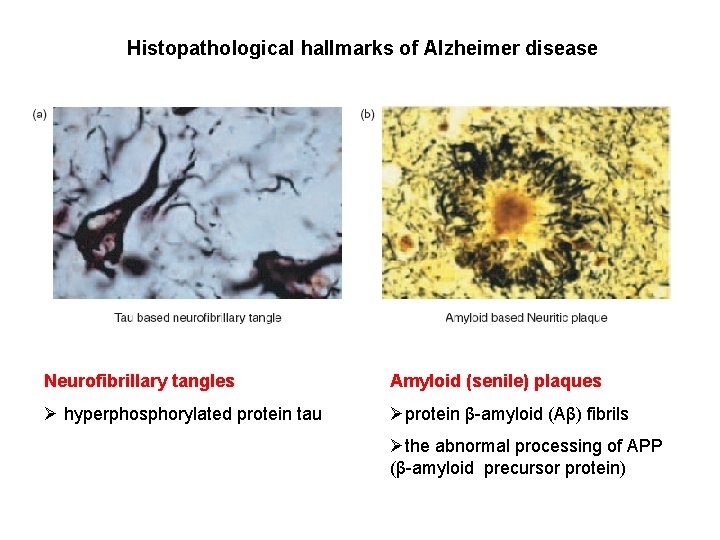
Histopathological hallmarks of Alzheimer disease Neurofibrillary tangles Amyloid (senile) plaques Ø hyperphosphorylated protein tau Øprotein β-amyloid (Aβ) fibrils Øthe abnormal processing of APP (β-amyloid precursor protein)
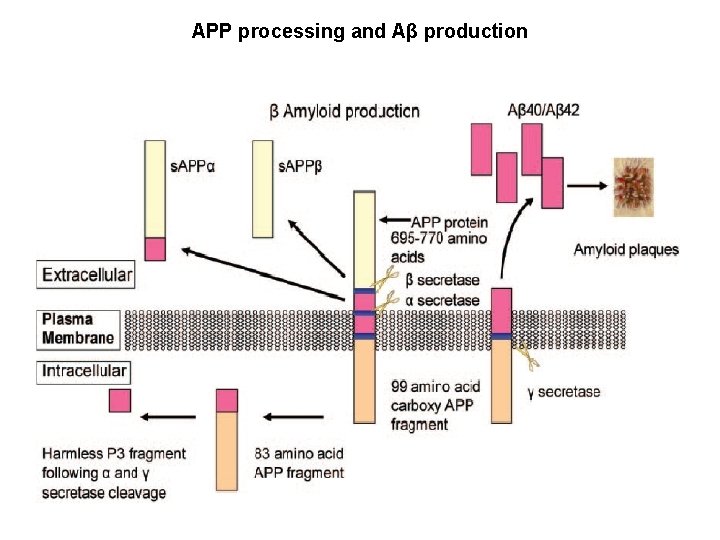
APP processing and Aβ production
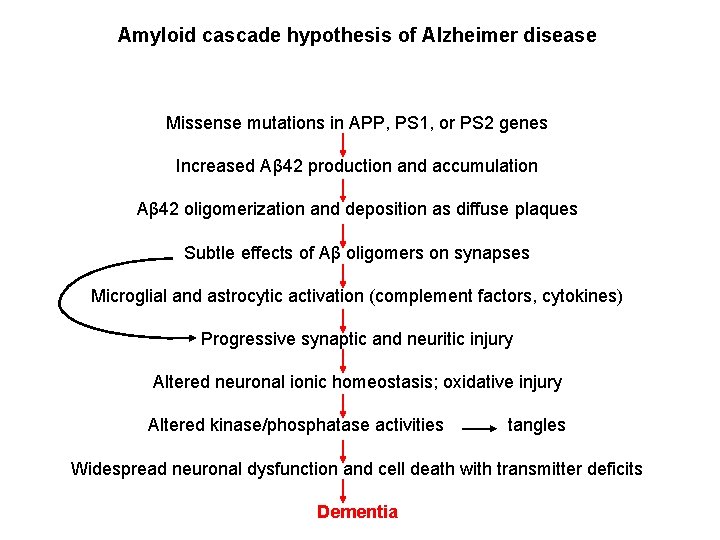
Amyloid cascade hypothesis of Alzheimer disease Missense mutations in APP, PS 1, or PS 2 genes Increased Aβ 42 production and accumulation Aβ 42 oligomerization and deposition as diffuse plaques Subtle effects of Aβ oligomers on synapses Microglial and astrocytic activation (complement factors, cytokines) Progressive synaptic and neuritic injury Altered neuronal ionic homeostasis; oxidative injury Altered kinase/phosphatase activities tangles Widespread neuronal dysfunction and cell death with transmitter deficits Dementia
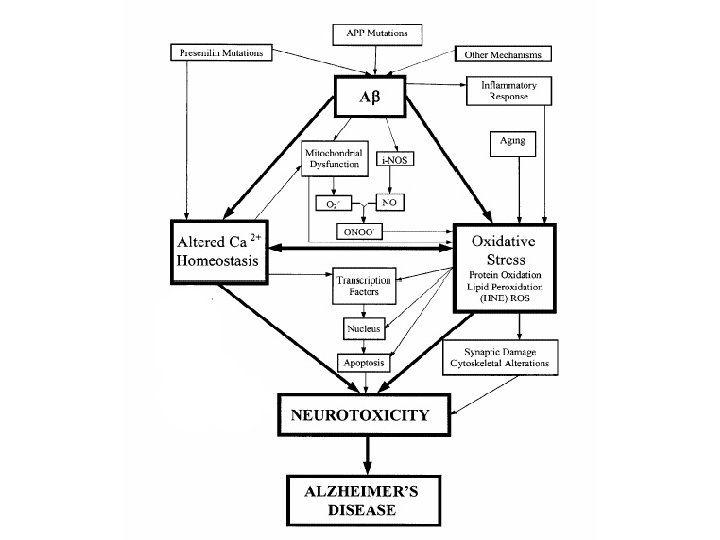

Prions are proteinaceous transmissible pathogens responsible for a series of fatal neurodegenerative diseases (in humans, Creutzfeld-Jakob disease and kuru, in animals, bovine spongioform encephalopathy) A prion (proteinaceous infectious particle, analogy for virion) is a type of infectious agent that does not carry the genetic information in nucleid acid! Prions are proteins with the pathological conformation that are believed to infect and propagate the conformational changes of the native proteins into the abnormally srtructured form
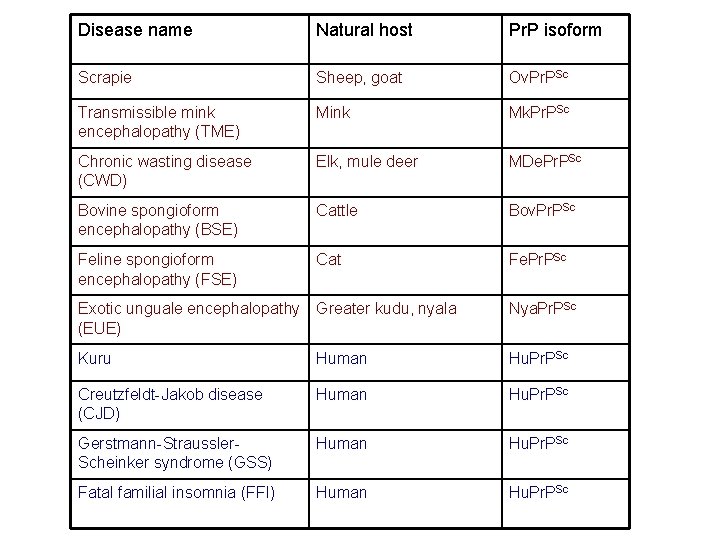
Disease name Natural host Pr. P isoform Scrapie Sheep, goat Ov. Pr. PSc Transmissible mink encephalopathy (TME) Mink Mk. Pr. PSc Chronic wasting disease (CWD) Elk, mule deer MDe. Pr. PSc Bovine spongioform encephalopathy (BSE) Cattle Bov. Pr. PSc Feline spongioform encephalopathy (FSE) Cat Fe. Pr. PSc Exotic unguale encephalopathy Greater kudu, nyala (EUE) Nya. Pr. PSc Kuru Human Hu. Pr. PSc Creutzfeldt-Jakob disease (CJD) Human Hu. Pr. PSc Gerstmann-Straussler. Scheinker syndrome (GSS) Human Hu. Pr. PSc Fatal familial insomnia (FFI) Human Hu. Pr. PSc
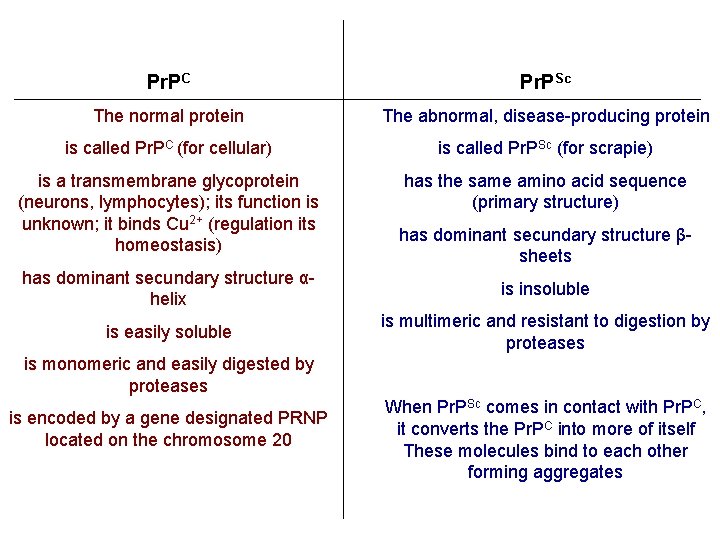
Pr. PC Pr. PSc The normal protein The abnormal, disease-producing protein is called Pr. PC (for cellular) is called Pr. PSc (for scrapie) is a transmembrane glycoprotein (neurons, lymphocytes); its function is unknown; it binds Cu 2+ (regulation its homeostasis) has the same amino acid sequence (primary structure) has dominant secundary structure βsheets has dominant secundary structure αhelix is insoluble is easily soluble is multimeric and resistant to digestion by proteases is monomeric and easily digested by proteases is encoded by a gene designated PRNP located on the chromosome 20 When Pr. PSc comes in contact with Pr. PC, it converts the Pr. PC into more of itself These molecules bind to each other forming aggregates
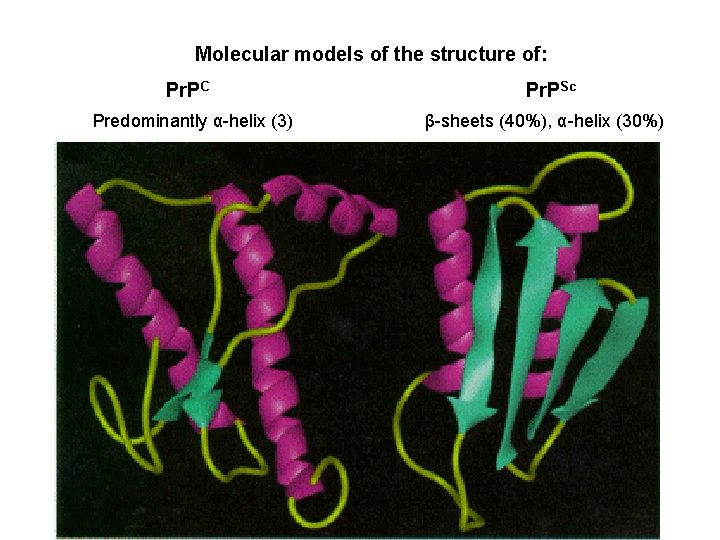
Molecular models of the structure of: Pr. PC Predominantly α-helix (3) Pr. PSc β-sheets (40%), α-helix (30%)
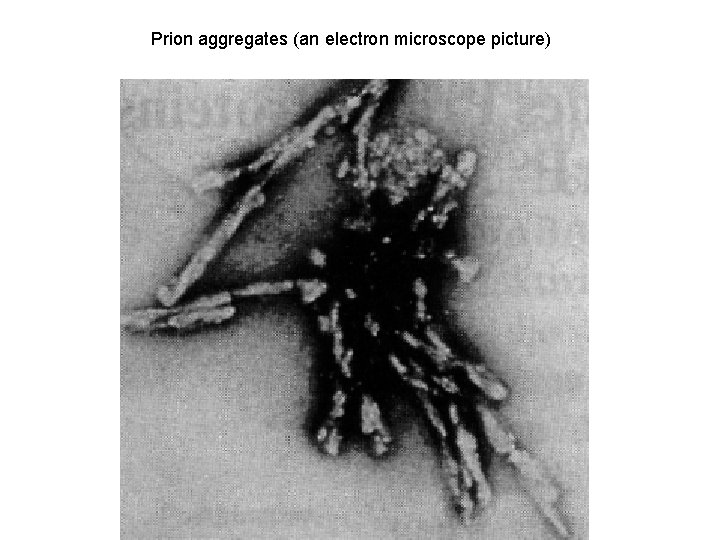
Prion aggregates (an electron microscope picture)
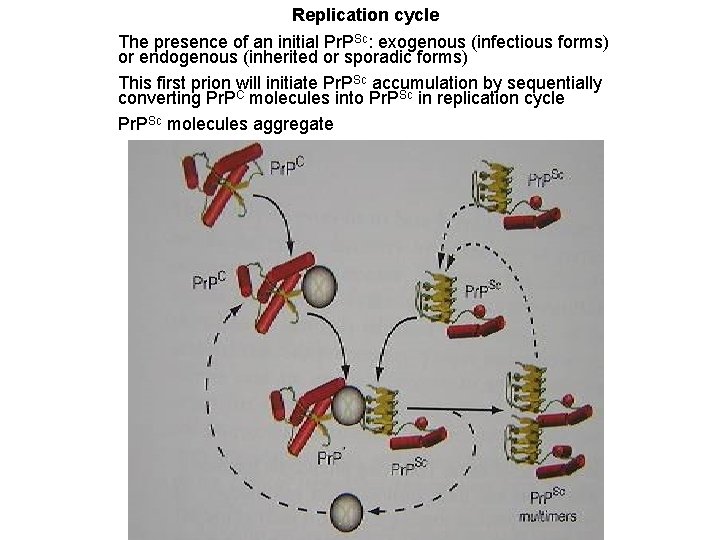
Replication cycle The presence of an initial Pr. PSc: exogenous (infectious forms) or endogenous (inherited or sporadic forms) This first prion will initiate Pr. PSc accumulation by sequentially converting Pr. PC molecules into Pr. PSc in replication cycle Pr. PSc molecules aggregate
Mechanism of prion progression

Prion diseases: rare neurodegenerative disorders (one person per million) 1. Sporadic (85 %) In the sixth or seventh decade, rapidly progressive (death in less than a year) Creutzfeldt-Jakob disease (CJD) 2. Familial (inherited-15%) Mutations in the Pr. P gene that favour the transition from the cellular form to the pathological form of Pr. P Gerstmann-Straussler-Scheinker disease (GSS), fatal familial insomnia (FFI) 3. Transmissible (rare; a source of great concern) In the laboratory, disease can be readily transmitted to mice by intracranial injection of brain homogenate taken from prion-infected animals Propagation of kuru disease in New Guinea natives (ritualistic cannibalism) Recently, it has been discovered that BSE had been transmitted to humans in Europe after consumption of infected beef, producing a variant of the CJD called v. CJD

Transmissible spongioform encephalopathy (TSE)=prion disease is a group of progressive conditions that affect the brain and nervous system of humans and animals and are transmitted by prions The pathology: vacuolar degeneration, neuronal loss, astrocytosis and amyloid plaque formation The clinical signs: loss of motoric functions (lack of coordination, ataxia, involuntary jerking movements), personality changes, depression, insomnia, confusion, memory problems, dementia, progressive tonic paralysis, death Definitive diagnostic test: biopsy of brain tissue There is no cure

Prion transmission 1. Direct contact with infected tissues CJD has been transmitted: -to patients taking injections of growth hormone harvested from human pituitary glands -from instruments used for brain surgery (prions can survive the autoclave sterilization process) -in corneal grafts -in electrode implants 2. Consumption of affected tissues Kuru was transmitted through cannibalism in Papua New Guinea Humans can contract the disease by consuming material from animals infected with the BSE (v. CJD) How can prions make their way through the gut and into the brain? Proteins normally are digested down to amino acids in the gut Hypothesis: They circumvent the normal process of intestinal absorption by passing into the Gut-Associated Lymphoid Tissue (GALT)

Prion strains =subclassification of prions following the structural characteristics that define their pathological profile The various prion diseases differ in incubation times and neuropathologic profiles, although in all cases the same misfolded protein (Pr. PSc) is responsible The existence of various prion strains: although they are chemically identical, they differ in their exact conformations The structural characteristics of individual prion strains are propagated via the replication cycle These characteristics appear to direct the precise tissue targeting, incubation time, and pathogenesis of the strain Consumption of BSE-infected meat BSE prion Variant CJD: younger cohort, distinct clinical characteristics v. CJD prion is identical to BSE prion Classical CJD: older cohort CJD prion is different
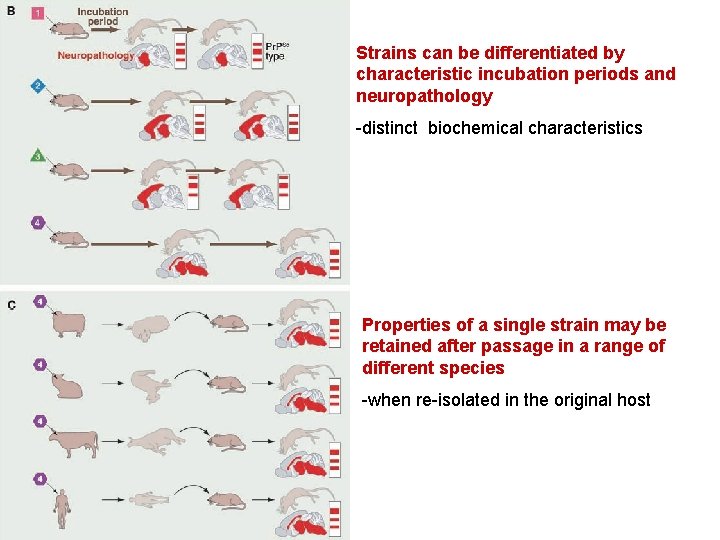
Strains can be differentiated by characteristic incubation periods and neuropathology -distinct biochemical characteristics Properties of a single strain may be retained after passage in a range of different species -when re-isolated in the original host
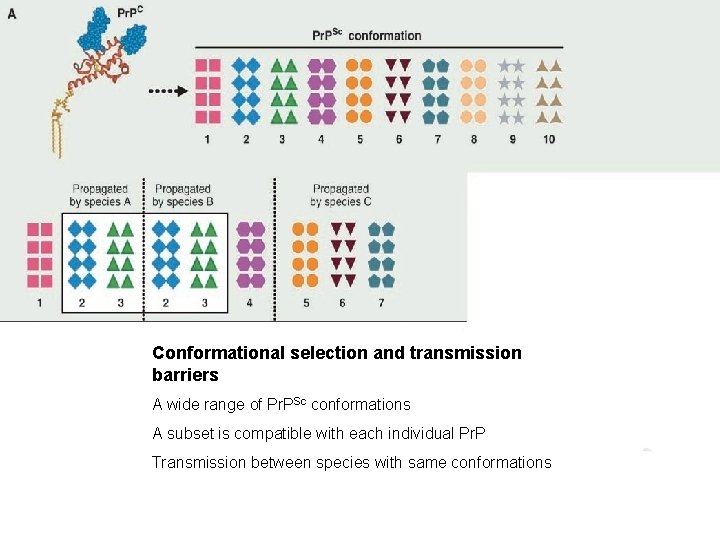
Conformational selection and transmission barriers A wide range of Pr. PSc conformations A subset is compatible with each individual Pr. P Transmission between species with same conformations

Therapeutic strategies 1. Compounds can be designed to specifically disrupt the replication cycle of the Pr. PSc Design of such compounds had proven successful in cell-based models but must now be extended to animal models and human clinical trials 2. Vaccine design: The abnormally folded proteins expose a side chain of amino acids which the properly folded protein does not expose. Antibodies specifically coded to this side chain amino acid sequence stimulate an immune response to the abnormal prions 3. Design of peptides that break the β-sheet structures 4. Gene therapy: modification of the prion gene Genetic engineering research: cattle lacking a necessary gene for prion production - thus theoretically making them immune to BSE (December 2006)
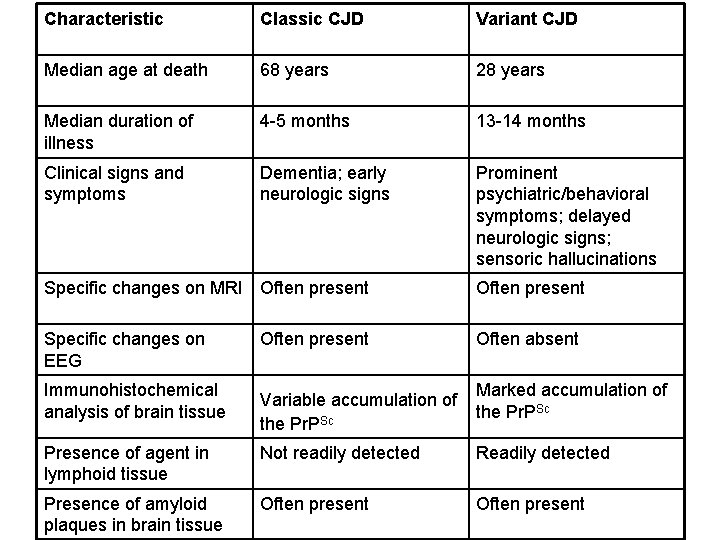
Characteristic Classic CJD Variant CJD Median age at death 68 years 28 years Median duration of illness 4 -5 months 13 -14 months Clinical signs and symptoms Dementia; early neurologic signs Prominent psychiatric/behavioral symptoms; delayed neurologic signs; sensoric hallucinations Specific changes on MRI Often present Specific changes on EEG Often absent Immunohistochemical analysis of brain tissue Often present Variable accumulation of the Pr. PSc Marked accumulation of the Pr. PSc Presence of agent in lymphoid tissue Not readily detected Readily detected Presence of amyloid plaques in brain tissue Often present

Summary Conformational defect of proteins - protein misfolding diseases neurodegenerative diseases Oligomeric aggregates - neurotoxic The mechanism: oxidative stress, disruption homeostasis The prions are proteins that carry information for self-reproduction (contradict the central dogma of modern biology) The prions are expressed in cells of healthy humans and animals; their abnormal conformations (Pr. PSc) are insoluble, resistent to digestion and aggregate The Pr. PSc attacks the native prion Pr. PC, changes its conformation into an abnormal form and causes an exponential production of insoluble proteins; they aggregate and form the fibrillar structure Prion disease are rare fatal degenerative disorders; a portion of them can be transmitted; this mechanism is not clear (e. g. transmision of BSE to human)
- Slides: 28