BIOCHEMISTRY OF BONE Jana Novotna The lecture content

BIOCHEMISTRY OF BONE Jana Novotna

The lecture content • Bone composition – cells, extracellular matrix, mineral; • Bone formation in growth plate, matrix vesicles, mineral formation, enzymes and other structures involved in process of mineralization; • Bone remodelling – an active and dynamic process, balance between bone resorption by osteoclasts and bone formation; • Hormons regulating bone metabolism; • Bone metabolism markers.

Bone matrix • Bone tissue is a relatively hard and lightweight composite material incorporating the mineral (calcium hydroxyapatite) and collagen, an elastic protein which improves fracture resistance. • Organic matrix - 20% – collagen, glycoproteins, bone specific proteoglycans, growth factors • Inorganic matrix – 65% – – hydroxyapatite – Ca 10(PO 4)6(OH)2, octacalcium phosphate - Ca 8 H 2(PO 4)6. 5 H 2 O brusite – Ca. HPO 4. 2 H 2 O, amorphouse calcium phosphates – Ca 9(PO 4)6 • Water – 10%
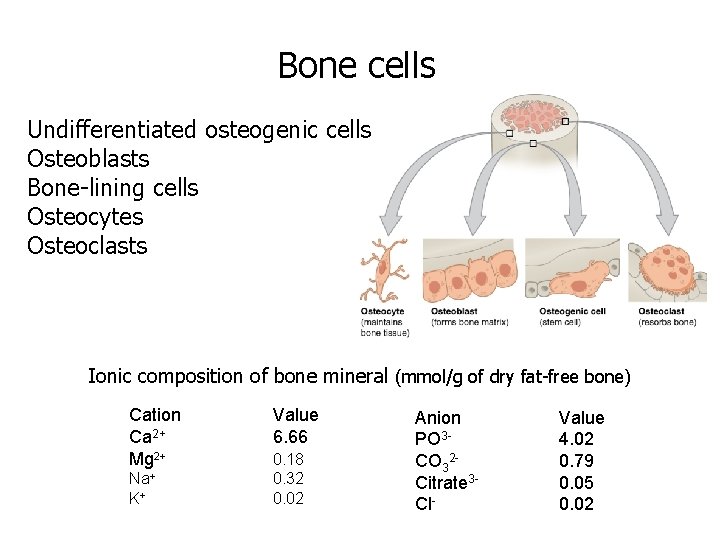
Bone cells Undifferentiated osteogenic cells Osteoblasts Bone-lining cells Osteocytes Osteoclasts Ionic composition of bone mineral (mmol/g of dry fat-free bone) Cation Ca 2+ Mg 2+ Na+ K+ Value 6. 66 0. 18 0. 32 0. 02 Anion PO 3 CO 32 Citrate 3 Cl- Value 4. 02 0. 79 0. 05 0. 02

Bone functions • Mechanical function – protection of internal organs (the skull the brain, the ribs the heart, lungs). – structure — bones provide a frame to keep the body support. – movement – bones provide leverage system for skeletal muscles, tendons, ligaments, joints function together to generate and transfer forces – sound transduction — important in the mechanical aspect of hearing • Synthetic function – the bone marrow produces blood cells in a process called hematopoiesis

Bone functions • Metabolic function – mineral storage - reserves of minerals important for the body (calcium, phosphorus) – growth factors storage (ILF, BMP, TGF) – fat storage (the yellow bone marrow acts as a storage reserve of FA) – acid-base balance (bone buffers the blood against excessive p. H changes by absorbing or releasing alcaline salts – detoxification — bone tissues can also store heavy metals and other foreign elements – endocrine organ – fibroblast growth factor-23 (FGF-23) acts on kidneys to reduce phosphate reabsorption, osteocalcin contributes to the regulation of blood glucose and fat deposition (increasing insulin secretion and sensitivity, reducing stores of fat).

The physiological unit of bone tissue A = vascular space B = osteocyte C = bone canaliculi D = gap junction between adjacent osteoblasts E = connective tissue
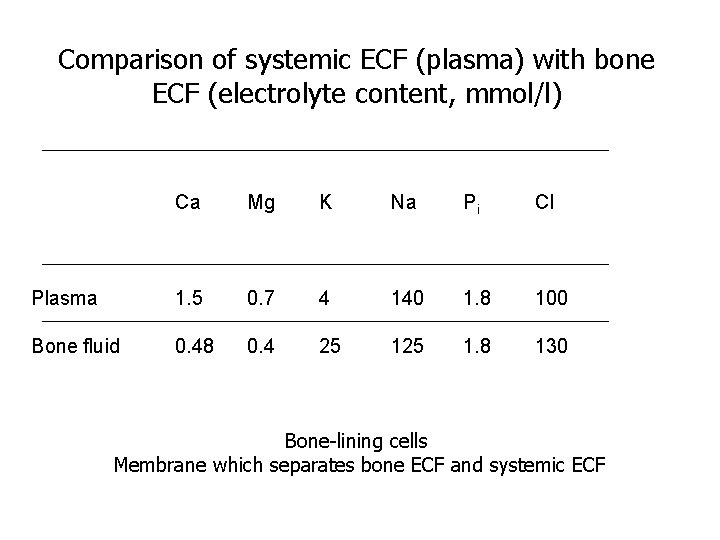
Comparison of systemic ECF (plasma) with bone ECF (electrolyte content, mmol/l) Ca Mg K Na Pi Cl Plasma 1. 5 0. 7 4 140 1. 8 100 Bone fluid 0. 48 0. 4 25 1. 8 130 Bone-lining cells Membrane which separates bone ECF and systemic ECF

Phases of bone formation § § bone formation (embryonic development) bone growth maintenance of the skeleton – modeling and remodeling healing of the skeleton (soft-tissue trauma, fracture, response to neoplasma, infection) There is only one mechanism of bone formation and the formation of bone follows the same sequence in development, growth, and maintenance of skeleton: § § § within cartilage – enchondral ossification within an organic matrix membrane – intramembranous ossification deposition of a new bone on existing bone – appositional ossification

The growth plate • Reverse zone - vertical column of chondrocytes in different phases of the cell cycle (flat and inactive chondrocytes, give rise to the daughter chondrocytes in the proliferative zone). • Proliferative zone - chondrocytes start to proliferate. • In the zone of maturation chondrocytes start to mature and give rise matrix vesicles, sites of calcification. • In the upper hyperthrophic zone chondrocytes start to enlarge and synthesize collagen type X, and produce alkaline phosphatase. Matrix vesicles are sites of the first mineral crystals. • Lower hyperthrofic zone contains degenerating apoptotic condrocytes. Proliferating mineral crystals spread in the extracellular matrix, small vessels penetrate into the matrix, osteoblasts from bone marrow deposit new bone and synthesize bone extracellular matrix.
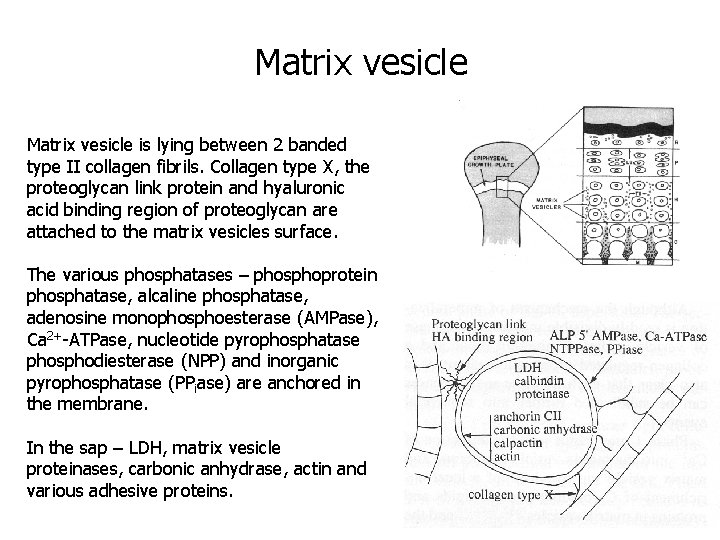
Matrix vesicle is lying between 2 banded type II collagen fibrils. Collagen type X, the proteoglycan link protein and hyaluronic acid binding region of proteoglycan are attached to the matrix vesicles surface. The various phosphatases – phosphoprotein phosphatase, alcaline phosphatase, adenosine monophosphoesterase (AMPase), Ca 2+-ATPase, nucleotide pyrophosphatase phosphodiesterase (NPP) and inorganic pyrophosphatase (PPiase) are anchored in the membrane. In the sap – LDH, matrix vesicle proteinases, carbonic anhydrase, actin and various adhesive proteins.
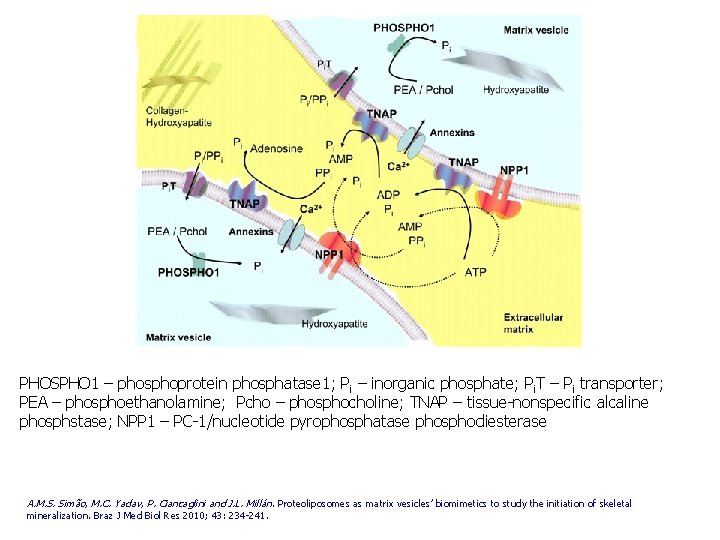
PHOSPHO 1 – phosphoprotein phosphatase 1; Pi – inorganic phosphate; Pi. T – Pi transporter; PEA – phosphoethanolamine; Pcho – phosphocholine; TNAP – tissue-nonspecific alcaline phosphstase; NPP 1 – PC-1/nucleotide pyrophosphatase phosphodiesterase A. M. S. Simão, M. C. Yadav, P. Ciancaglini and J. L. Millán. Proteoliposomes as matrix vesicles’ biomimetics to study the initiation of skeletal mineralization. Braz J Med Biol Res 2010; 43: 234 -241.

Matrix vesicle enzymes, channels, transporters • Tissue-nonspecific alcaline phosphatase (TNAP) – generation of inorganic phosphate, hydrolysis of local PPi to two phosphates (Pi) – Deficiency hypophosphatasia (perinatal, infantile, childhood, adult hypophosphatasia, odontohypophosphatasia). Clinical severity varies widely - range from complete absence of bone mineralization to spontaneous fractures, loss of decidual teeth. • Nucleotide pyrophosphatase/phospho-diesterase (NPP 1, alcaline phosphatase superfamily) – hydrolyzis of diesters of phosphoric acid into monoesters (ATP AMP + PPi, ADP adensine + PPi). Role of NPP 1 – modulation of mineral growth inhibiting hydroxyapatite precipitation by PPi – Deficiency – development of soft tissue calcification, including vascular calcification.

Matrix vesicle enzymes, channels, transporters • Phosphoprotein phosphatase 1 (PHOSPHO 1) – generating of Pi from phosphocholine and phosphoethanolamine • Annexins – calcium channels, annexin V binds type II and X collagen, interactions stimulate channel activities ( influx of Ca 2+) • Inorganic phosphate transporter (Pi. T) – Na+-dependent Pi symporters • Carbonic anhydrase - regulation of p. H inside of matrix vesicules, removing of protons as biproduct of hydroxyapatite formation
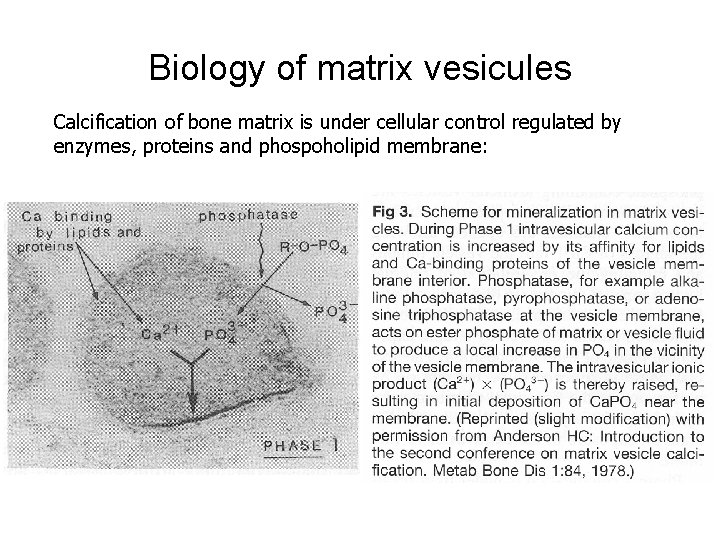
Biology of matrix vesicules Calcification of bone matrix is under cellular control regulated by enzymes, proteins and phospoholipid membrane:
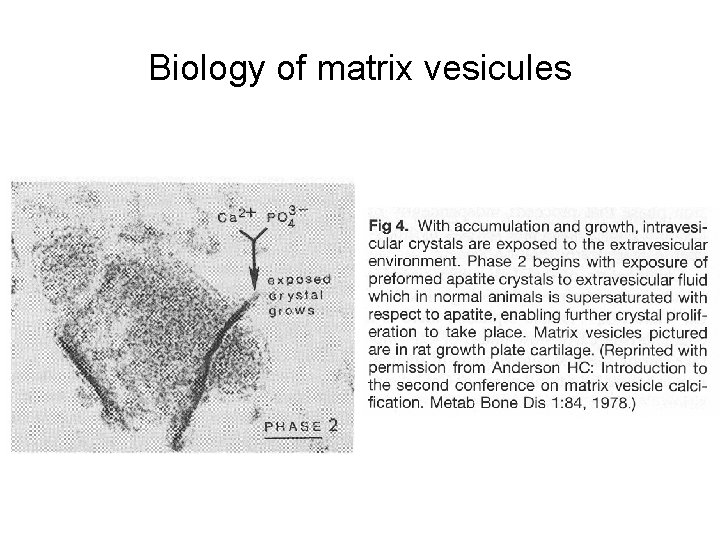
Biology of matrix vesicules

Matrix of hyperthrofic cartilage contains typical cartilage proteoglycans and collagen type II. During nucleation phase matrix get to shrink, proteoglycans aggregate. Ca 2+ from adjacent matrix withdrawn into hyperthrophic cartilage matrix. Chondrocytes and matrix vesicule produce alkaline phosphatase (production of free phosphates for calcification). Mineral crystals of hydroxyapatite are formed and mineralization continues.
Phases of enchondral ossification • • • hypertrophy of chondroblasts calcification of cartilage extracellular matrix penetration of blood capillaries differentiation of osteoblasts and osteoclasts osteoid formation osteoid calcification Remodeling of new bone • bone resorption • synthesis and secretion of the bone matrix • calcification of newly formed bone lamellae

Bone resorption and bone formation are not separated, independently regulated process. Osteoblast and osteoclasts belong to structure, known as a basic multicellular unit (BMU). Osteoclasts are responsible to bone resorption Proton pupm, citric acid, lactic acid p. H 7 p. H 4 Lysosomal enzymes Acid phosphatase Collagenase Glycosidase Sulphatase Catepsins Osteoblasts are responsible to organic extracellular matrix formation.
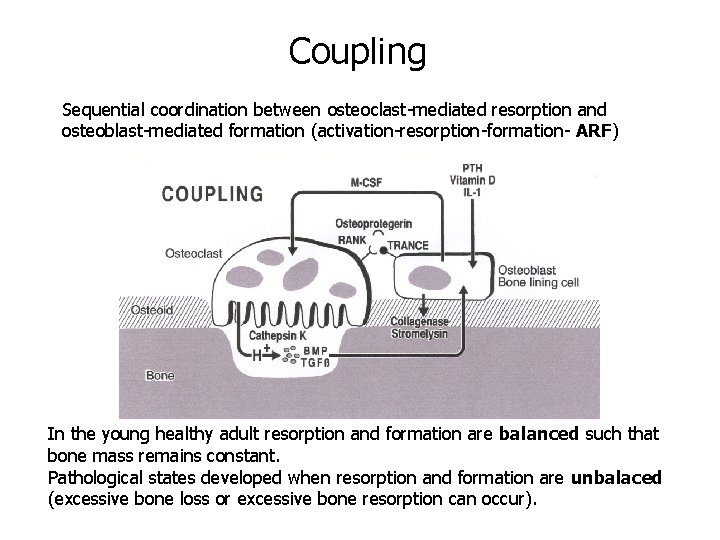
Coupling Sequential coordination between osteoclast-mediated resorption and osteoblast-mediated formation (activation-resorption-formation- ARF) In the young healthy adult resorption and formation are balanced such that bone mass remains constant. Pathological states developed when resorption and formation are unbalaced (excessive bone loss or excessive bone resorption can occur).
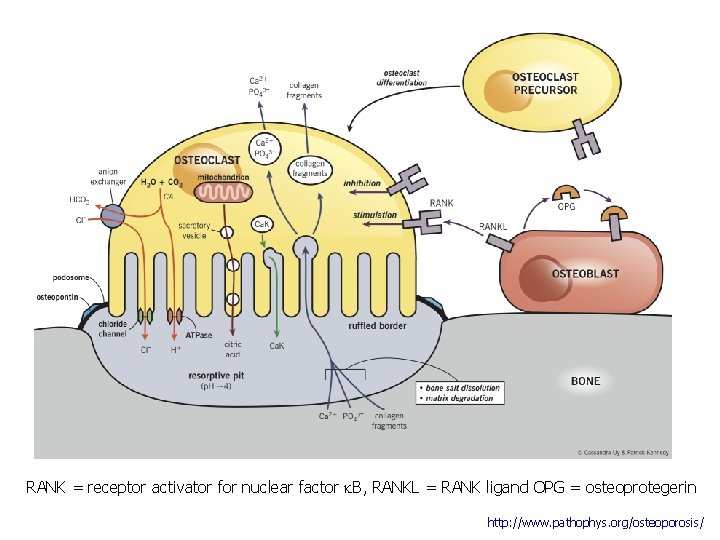
RANK = receptor activator for nuclear factor k. B, RANKL = RANK ligand OPG = osteoprotegerin http: //www. pathophys. org/osteoporosis/
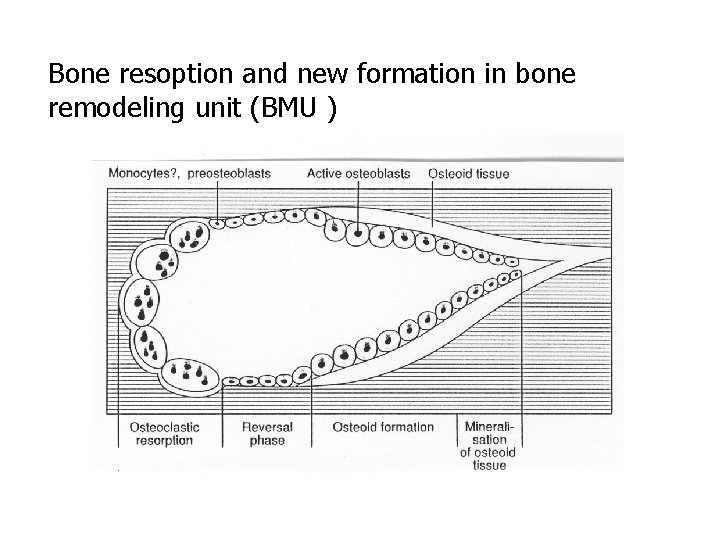
Bone resoption and new formation in bone remodeling unit (BMU )
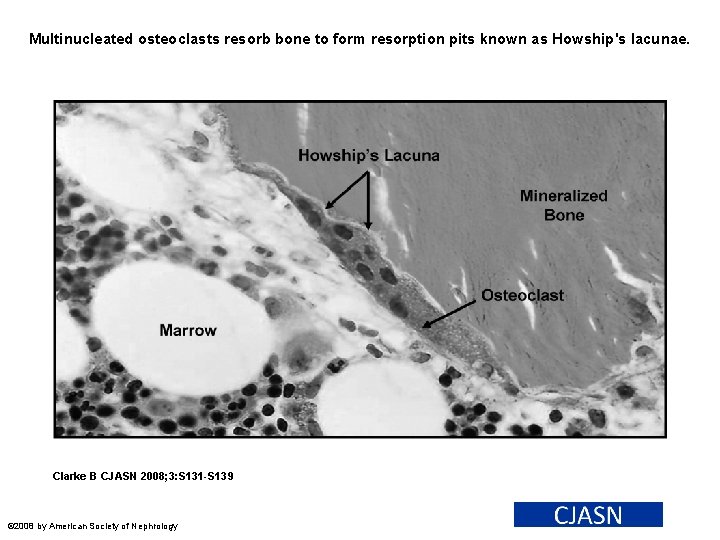
Multinucleated osteoclasts resorb bone to form resorption pits known as Howship's lacunae. Clarke B CJASN 2008; 3: S 131 -S 139 © 2008 by American Society of Nephrology

Osteoblasts synthesize proteinaceous matrix, composed mostly of type I collagen, to fill in resorption pits. Clarke B CJASN 2008; 3: S 131 -S 139 © 2008 by American Society of Nephrology
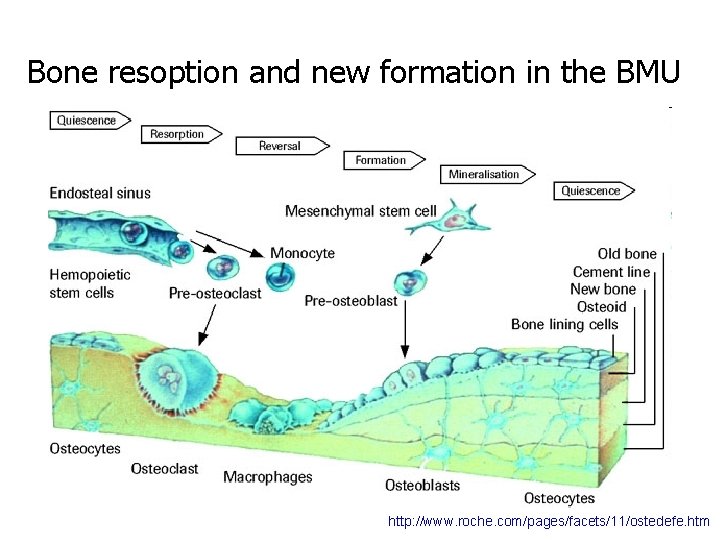
Bone resoption and new formation in the BMU http: //www. roche. com/pages/facets/11/ostedefe. htm

Components of bone extracellular matrix Collagen type I, trace amount of type III and V (regulation of collagen fibrils) Small proteoglycans – aggrecan, decorin, biglycan, hyaluronic acid Bone glycoproteins : bone sialoprotein (initiation of mineralization), osteonectin (regulation of collagen fibril diameter), osteopontin (inhibition of mineralization and remodeling) Addhesive glycoproteins – fibronectin, vitronectin, g-carboxy glutamic acid-containing proteins (martix Gla proteins – inhibits mineralization), fibrilin (regulation of elastic fiber formation), osteocalcin (regulation of osteoclasts, inhibition of mineralization) Growth factors

Hormonal Control of Calcium Homeostasis • The extracellular fluid (or plasma) calcium concentration is tightly controlled by a complex homeostatic mechanism involving fluxes of calcium between the ECF and the kidney, bone, and gut. • These fluxes are carefully regulated by three major hormones: parathyroid hormone (PTH), calcitonin, and 1, 25 -dihydroxyvitamin D [1, 25(OH)2 D 3]. • Total corrected serum of biologically active ionized calcium is >2. 9 mmol/L (11. 5 mg/d. L) or <2. 1 mmol/L (8. 5 mg/d. L).
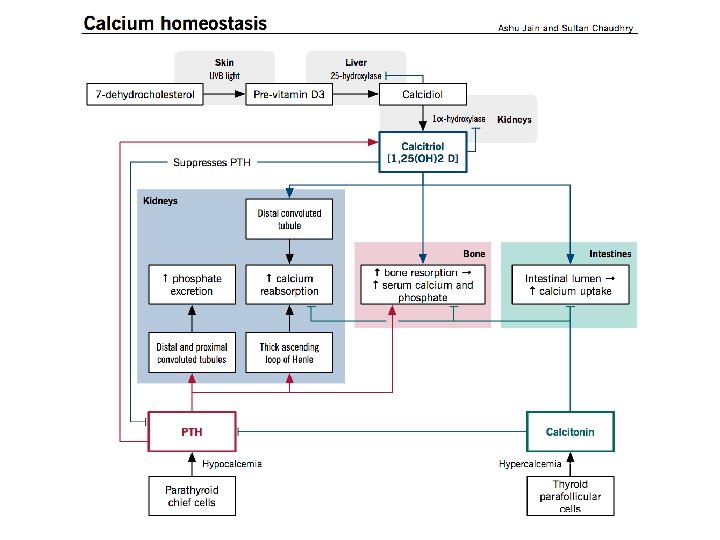

Parathyroid hormone • • PTH is an 84 -amino acid peptide synthesized by the chief cells of the parathyroid gland. Secretion of PTH is highly dependent on the ionized calcium concentration and represents a simple negative feedback loop serum Ca 2+ PTH (calcium sensing receptor - Ca. SR in parathyroid cells). High phosphate levels also induce PTH secretion The biological actions of PTH: a) stimulation of osteoclastic bone resorption and release of calcium and phosphate from bone, b) stimulation of calcium reabsorption and inhibition of phosphate reabsorption from the renal tubules, c) stimulation of renal production of 1, 25(OH)2 D 3, which increases intestinal absorption of calcium and phosphate.

Calcitonin • Calcitonin is a 32 -amino acid peptide synthesized and secreted by the parafollicular cells of the thyroid gland. • Increases in ionized Ca 2+ produce an increase in calcitonin secretion, and conversely, a fall in the ambient Ca 2+ concentration inhibits calcitonin secretion. • Calcitonin lowers blood Ca 2+ levels in three ways: – directly inhibits osteoclastic bone resorption (inhibition is accompanied by the production of c. AMP, increase in cytosolic calcium, leads to contraction of the osteoclast cell membrane. This effect is transient). – Inhibits Ca 2+ absorption by the intestines – Inhibits renal tubular cells reabsorption of Ca 2+

Vitamin D • • • The vitamin D precursor (previtamin D 3) is either ingested in the diet or synthesized in the skin from 7 -dehydrocholesterol Ergocalciferol (vitamin D 2) and cholecalciferol (vitamin D 3) are found in plant and animal sources, absorbed via the intestines. Vitamin D effects: a) 1, 25(OH)2 D 3 increases plasma calcium and phosphate concentrations by increasing the absorption of calcium and phosphate from the intestinal lumen b) It also increases bone resorption and enhances the effects of PTH in the nephron to promote renal tubular calcium reabsorption. c) It stimulates osteoblasts to secrete RANKL, thereby activating osteoclasts to resorb bone to raise serum calcium levels. • Vitamin D also suppresses PTH production

Other systemic hormones : Glucocortocuides – inhibition of bone formation. Growth hormone (GH) – stimulation of bone formation through somatomedins (growth factors IGF-1 and IGF-2). Insulin – stimulation of synthetic activity of osteoblasts. Thyroid hormones – stimulation of osteoclasts, activation of bone remodelation. Estrogens – inhibition of bone resorption (inhibition of osteoclastic activity through specific local factors). Catecholamines – antagonists of calcitonin. Prostaglandins – different classes of prostaglandins have different effect, which is dependent on concentration (10 -9 – 10 -7 mol/l) stimulates synthesis of collagen, 10 -6 mol/l inhibits collagen synthesis.

Local factors which regulate bone metabolism • Factors which stimulate osteoblasts differentiation : Bone morphogenic factor (BMF) Platelet-derived growth factor (PDGF) Fibroblast growth factor (FGF) Insulin-like growth factor 1 (IGF-1) Transforming growth factor-b (TGF-b). • Factors which regulate osteoclasts: Colony-stimulating factor (CSF) – suppression of osteoclasts development. Interferons (g-interferon) – inhibition of osteoclasts differentiation. Interleukins (IL-1, IL-3, IL-6, IL-11) – stimulates osteoclasts differentiation.

Biochemical markers of bone formation • Alkaline phosphatase – isoenzyme of bone alkaline phosphatase activity in serum reflects osteoblastic activity. • Osteocalcin - also called bone gamma-carboxyglutamate (Gla) protein – small, noncollagenous protein that is specific for bone tissue and dentin – its precise function remains unknown – is synthesized predominantly by the osteoblast and is mostly incorporated into the extracellular matrix of the bone. • Procollagen I extension propeptides

Biochemical markers of bone resorption • Urinary calcium – fasting urinary calcium test performed on a morning sample and corrected for creatinine excretion (urinary calcium tests lack sensitivity - reflects not only skeletal resorption, but also intestinal absorption as well as renal tubular filtration and reabsorption of calcium) • Hydroxyproline – is found mainly in collagen and represents about 13% of the amino acid content of the molecule. – Methods for measuring hydroxyproline - photometry, fluorometry and high pressure liquid chromatography (HPLC). • Urinary pyridinium crosslinks (pyridinoline and hydroxypyridinoline).
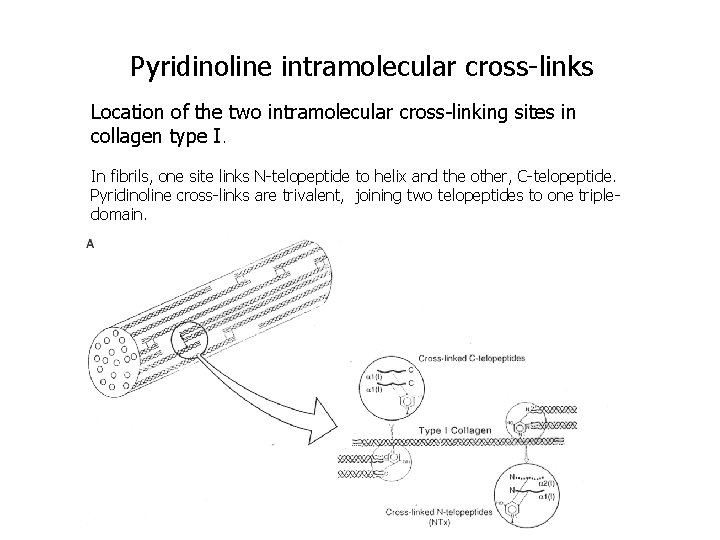
Pyridinoline intramolecular cross-links Location of the two intramolecular cross-linking sites in collagen type I. In fibrils, one site links N-telopeptide to helix and the other, C-telopeptide. Pyridinoline cross-links are trivalent, joining two telopeptides to one tripledomain.

- Slides: 37