Biochemistry Notes Carbon Organic molecules contain carbon Carbon

Biochemistry Notes

Carbon • Organic molecules contain carbon. • Carbon has 4 electrons available for bonding. •
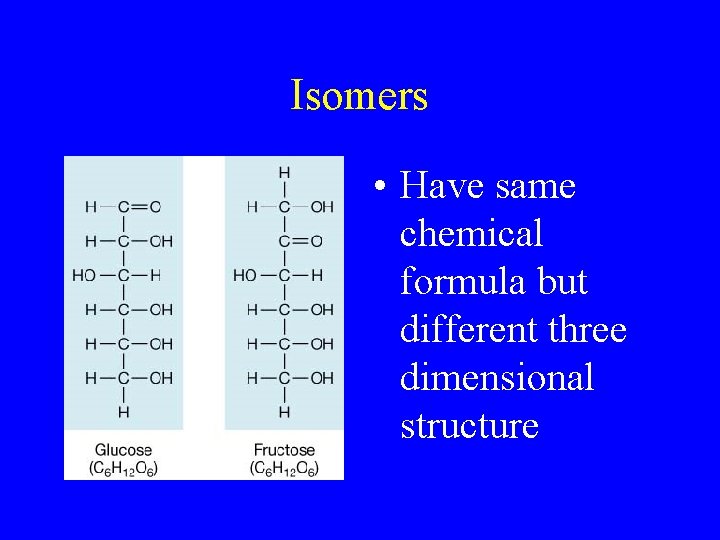
Isomers • Have same chemical formula but different three dimensional structure
Polymer • Polymer - a molecular chain made up of smaller molecules bonded together (poly = many)
Monomer • Monomer - 1 subunit of a polymer (mono = one) • Glucose is an example of a monomer

Macromolecules • Macromolecule - large organic molecule made up of polymers

Carbohydrates • Organic compound made up of C, H & O • Store and release energy • Monosaccharide (glucose: C 6 H 12 O 6, fructose) • Disaccharide - 2 monosaccharides (glucose + fructose = sucrose) • Polysaccharides (starch, cellulose, glycogen) - food storage and cell structures – Glycogen - animal cells – Starch - plant energy – Cellulose-plant cell struct.
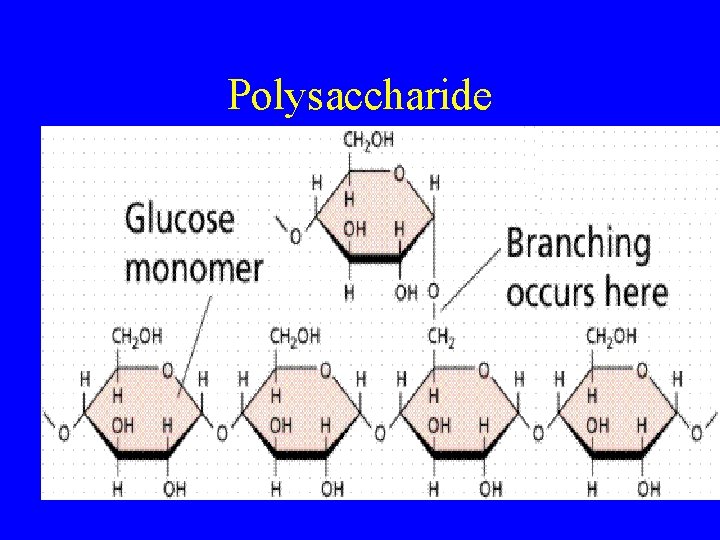
Polysaccharide
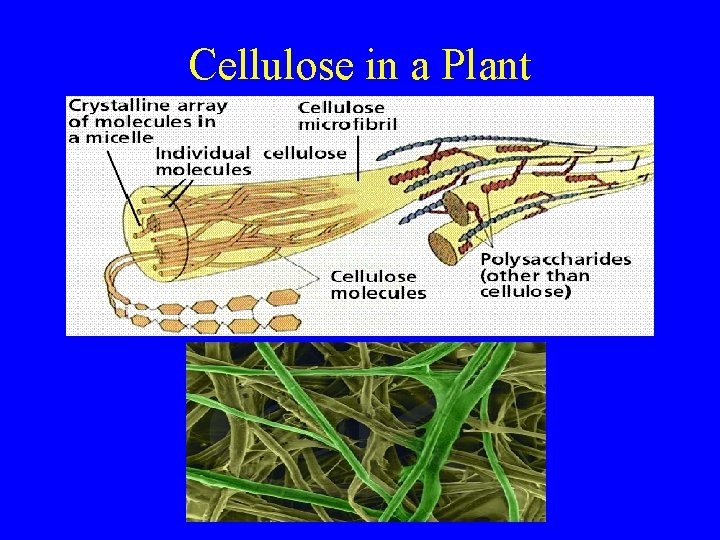
Cellulose in a Plant
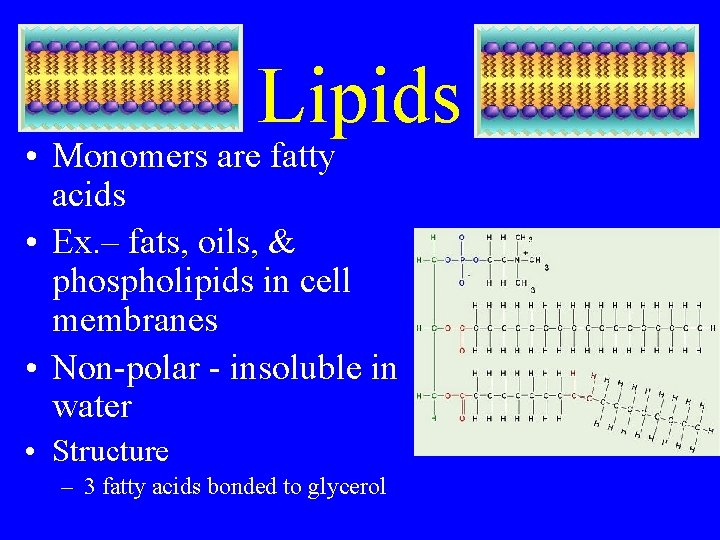
Lipids • Monomers are fatty acids • Ex. – fats, oils, & phospholipids in cell membranes • Non-polar - insoluble in water • Structure – 3 fatty acids bonded to glycerol
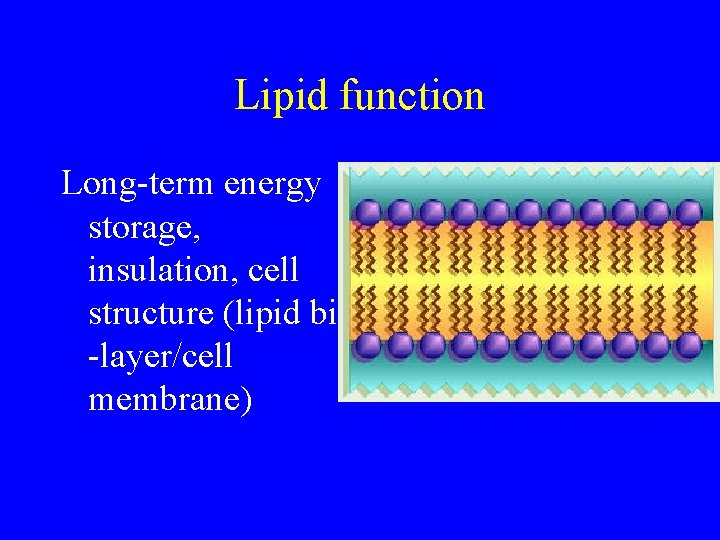
Lipid function Long-term energy storage, insulation, cell structure (lipid bi -layer/cell membrane)

Saturated Fatty Acid • • Solid at room temperature • Bad for heart • Meat, butter • Contain lots of hydrogen atoms and carbons are linked with single bonds in fatty acid

Unsaturated Fatty Acid • Liquid at room temperature • Good for your heart • Olive oil, nuts, fish. • Contain few hydrogen atoms because carbon atoms have double bonds in fatty acid chain.

Proteins provide structure (skin, hair, cartilage, muscle) and are carry out all cell metabolism (enzymes) The shape determines function Monomer—amino acids Linked together by peptide bonds (type of covalent bond)
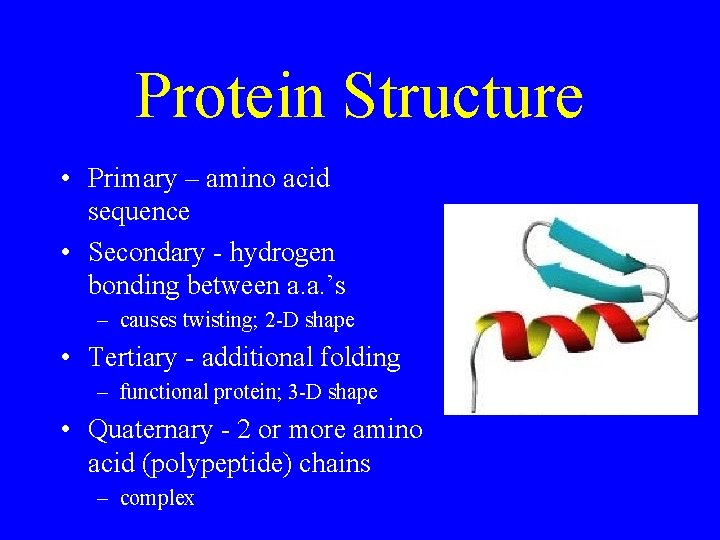
Protein Structure • Primary – amino acid sequence • Secondary - hydrogen bonding between a. a. ’s – causes twisting; 2 -D shape • Tertiary - additional folding – functional protein; 3 -D shape • Quaternary - 2 or more amino acid (polypeptide) chains – complex
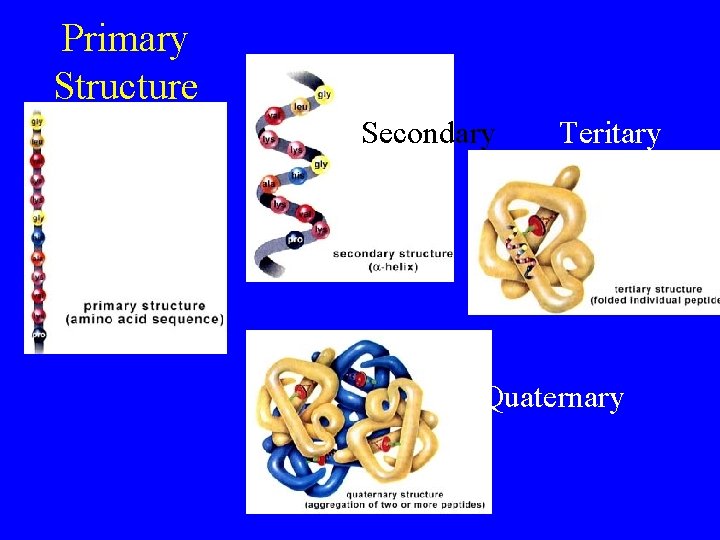
Primary Structure Secondary Teritary Quaternary

Nucleic Acids • Macromolecule that stores and transfers genetic material • Made up of nucleotides – made up of a sugar, a phosphate group, and a nitrogen base • DNA: deoxyribonucleic acid • RNA: ribonucleic acid
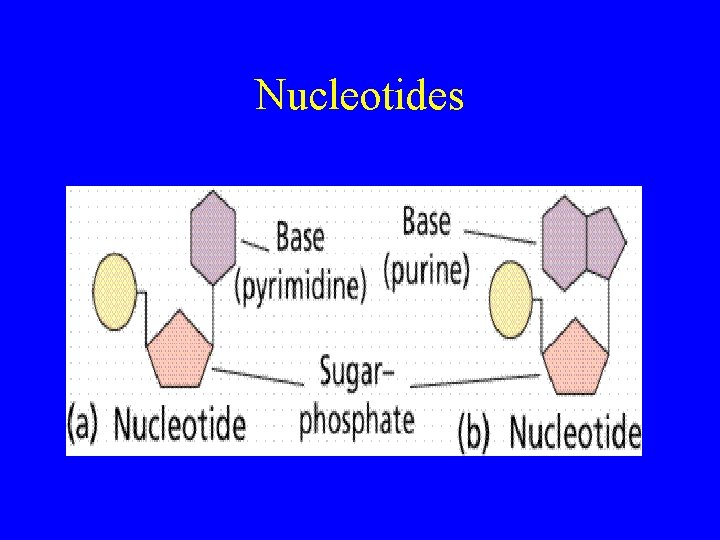
Nucleotides

Reactions of Polymers • Dehydration • Hydrolysis – large Synthesis – small molecules are broken molecules are linked down into smaller together to form molecules. large molecules • Water molecule is • Lose one molecule of used to break up the water when this polymer happens
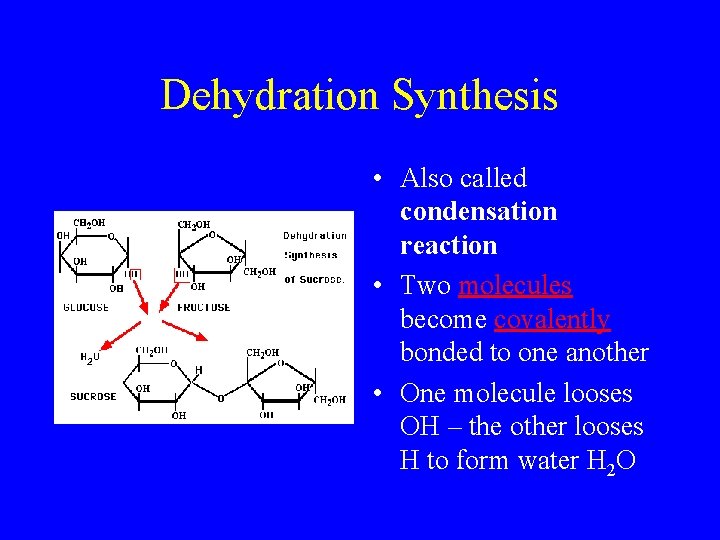
Dehydration Synthesis • Also called condensation reaction • Two molecules become covalently bonded to one another • One molecule looses OH – the other looses H to form water H 2 O

Hydrolysis Molecule is split into two parts by reacting with water. One of the parts gets an OH- from the water molecule and the other part gets an H+ from the water.
- Slides: 21