Biochemistry Molecules of Life Organic Chemistry Organic Compounds
Biochemistry Molecules of Life.

Organic Chemistry • Organic Compounds - compounds synthesized by cells and contain carbon • • • ---Four types of organic molecules (biomolecules) exist in organisms: carbohydrates lipids proteins nucleic acids • Organic compounds are made from a carbon skeleton which can vary in length, be branched or unbranched, have double bonds which vary in location, or may be arranged in rings.

Carbon Bonding • Carbon has 4 e- on outer shell therefore it will for 4 covalent bonds to become stable. • Three types of bonds are generally formed: – Straight: – Branched: – Ring:

Carbon Cont. • Carbon can also pair with another carbon molecule form: – Single Bonds – Double Bonds – Triple Bonds

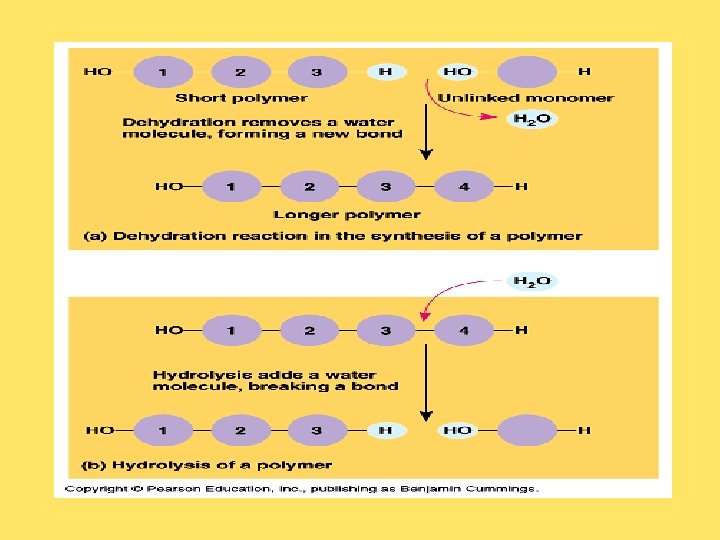
MACROMOLECULES • 1. Carbohydrates, lipids, proteins, and nucleic acids are called macromolecules because of their large size. • 2. The largest macromolecules are called polymers, constructed by linking many of the same type of small subunits, called monomers. *****Examples: amino acids (monomers) are linked to form a protein (polymer); many nucleotides (monomers) are linked to form a nucleic acid (polymer). • 3. Dehydration reactions to synthesize macromolecules. In a dehydration reaction, a water molecule is removed and a covalent bond is made between two atoms of the monomers. • Ex. Glucose + Fructose = Sucrose • 4. Hydrolysis ("water breaking") reactions break down polymers in reverse of dehydration; • 5. Enzymes are molecules that speed up chemical reactions by bringing reactants together; an enzyme may even participate in the reaction but is not changed by the reaction.
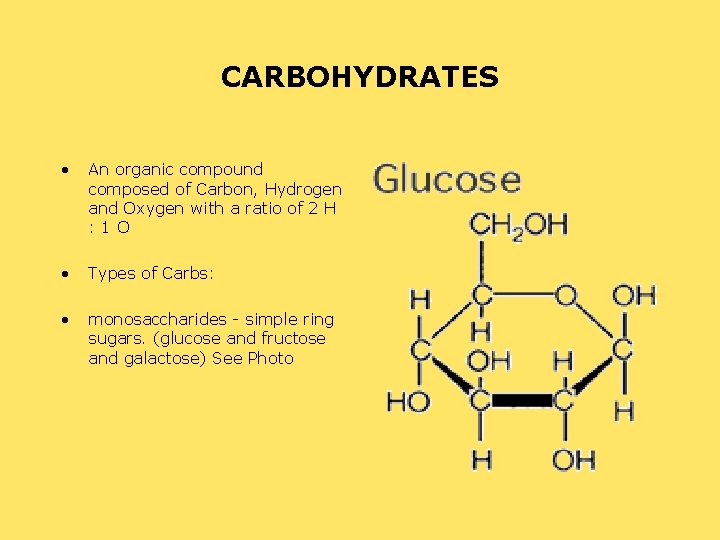
CARBOHYDRATES • An organic compound composed of Carbon, Hydrogen and Oxygen with a ratio of 2 H : 1 O • Types of Carbs: • monosaccharides - simple ring sugars. (glucose and fructose and galactose) See Photo
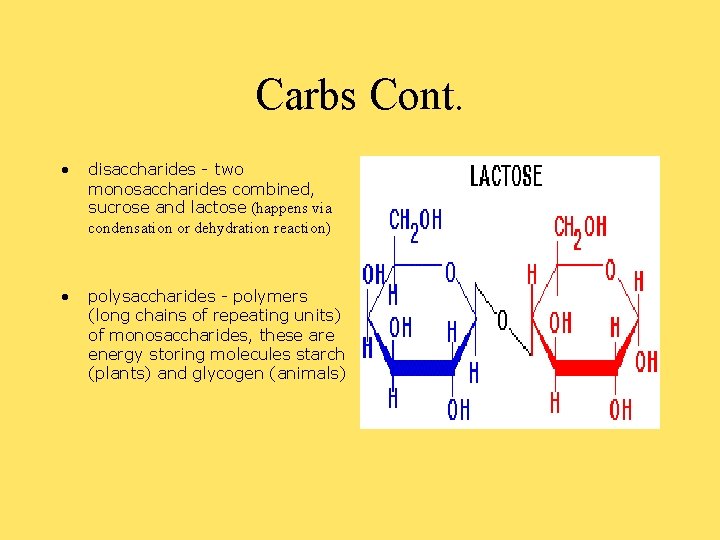
Carbs Cont. • disaccharides - two monosaccharides combined, sucrose and lactose (happens via condensation or dehydration reaction) • polysaccharides - polymers (long chains of repeating units) of monosaccharides, these are energy storing molecules starch (plants) and glycogen (animals)
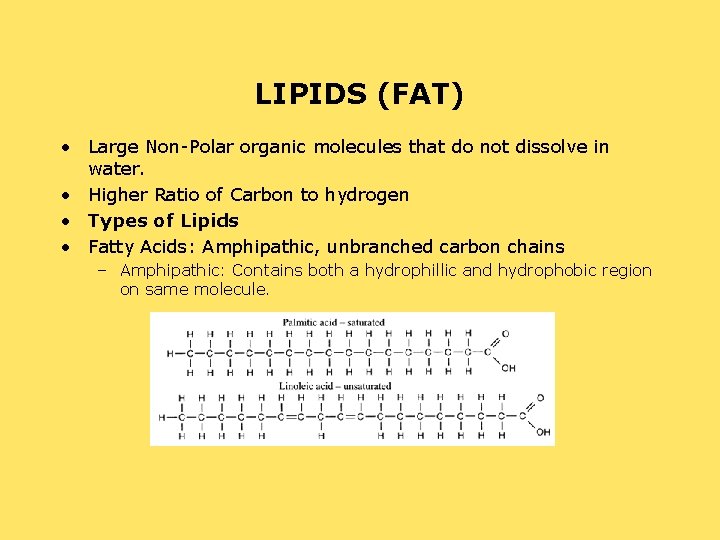
LIPIDS (FAT) • Large Non-Polar organic molecules that do not dissolve in water. • Higher Ratio of Carbon to hydrogen • Types of Lipids • Fatty Acids: Amphipathic, unbranched carbon chains – Amphipathic: Contains both a hydrophillic and hydrophobic region on same molecule.

Lipids Cont • Phospholipids - form membranes of cells (See Drawing) • Steroids (cell messengers) - composed of 4 rings of carbon, also includes Chlolesterol, examples: testosterone, estrogen • Waxes (protection & waterproofing) • Triglycerides (fats and oils) - fats are made of chains of fatty acids • Saturated fats contain no double bonds Saturated fats solid at room temperature Unsaturated fats have double bonds are liquid at room temperature
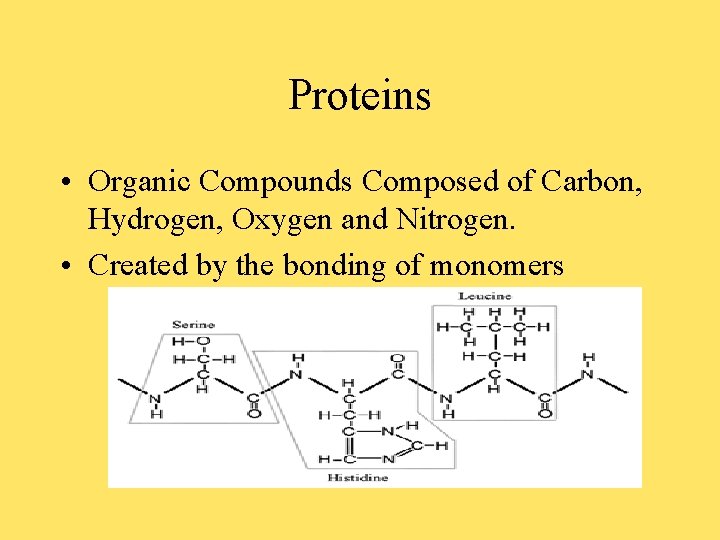
Proteins • Organic Compounds Composed of Carbon, Hydrogen, Oxygen and Nitrogen. • Created by the bonding of monomers

Protein Cont. • There are 20 different Amino Acids and they are the Building Blocks (monomers) of Proteins. • Amino Acids are composed of Carbon bonded to 4 other Atoms. (see prior picture) • The “R” group (Functional group) determines the differences in each Amino Acid. • The bond between each Amino Acid is called a dipeptide and is the result of a Condensation reaction.

Nucleic Acids • NUCLEIC ACIDS: Very Large Complex organic molecules that store important information in the cell. • Informational polymers (Contain Genetic Information) • DNA(deoxyribonucleic acid) & RNA (ribonucleic acid) Structure of DNA • Composed of Nucleotides – 5 carbon sugar (deoxyribose) – Nitrogen base (adenine, thymine, guanine, cytosine) – Phosphate group
- Slides: 13