Biochemistry Macromolecules Carbohydrates Lipids Proteins and Nucleic Acids

Biochemistry Macromolecules Carbohydrates, Lipids, Proteins and Nucleic Acids 2/12/2022 1

Carbohydrates • Body’s most important source of energy • Plants are our source of carbohydrates • Three basics groups of carbohydrates: mono saccharides, disaccharides and polysaccharides 2/12/2022 2
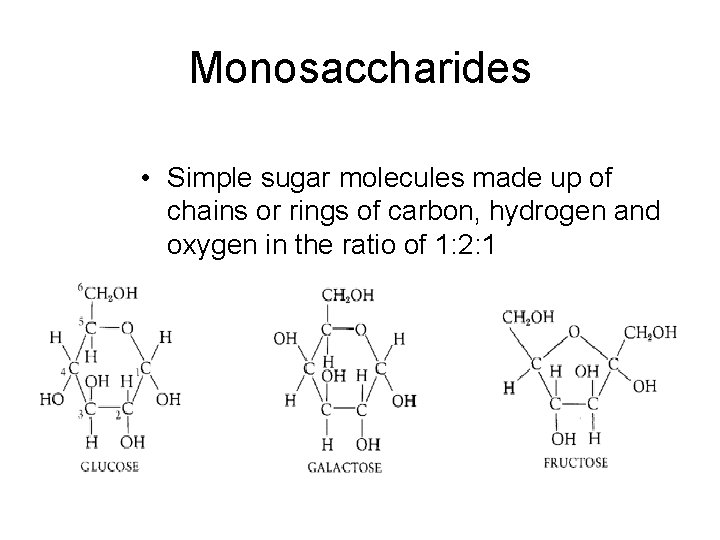
Monosaccharides • Simple sugar molecules made up of chains or rings of carbon, hydrogen and oxygen in the ratio of 1: 2: 1 2/12/2022 3
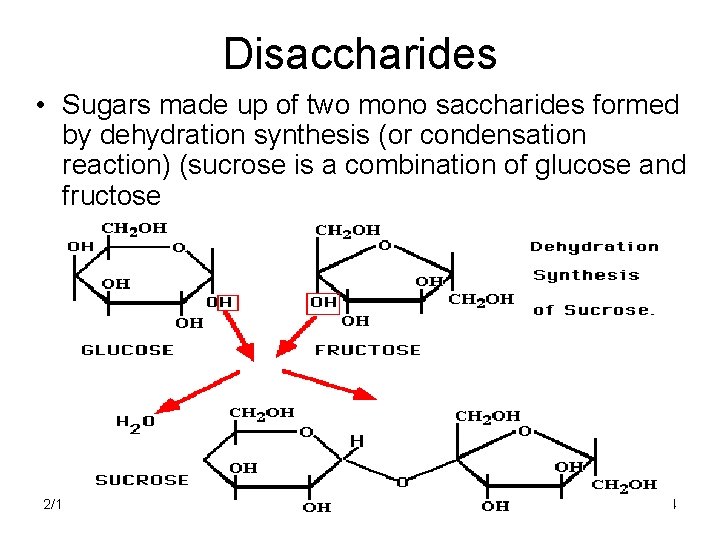
Disaccharides • Sugars made up of two mono saccharides formed by dehydration synthesis (or condensation reaction) (sucrose is a combination of glucose and fructose 2/12/2022 4
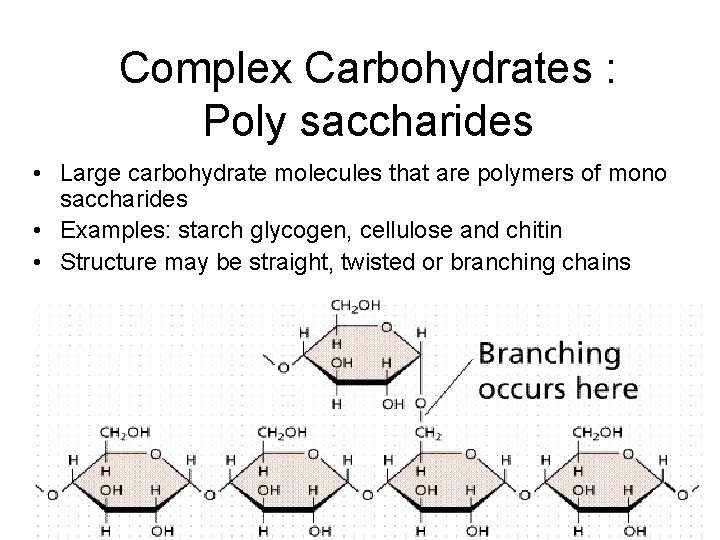
Complex Carbohydrates : Poly saccharides • Large carbohydrate molecules that are polymers of mono saccharides • Examples: starch glycogen, cellulose and chitin • Structure may be straight, twisted or branching chains 2/12/2022 5

Lipids • include fats, phospholipids, waxes and steroids • fats serve as long-term energy storage molecules • phospholipids forms cell membranes • steroids include cholesterol and certain sex hormones 2/12/2022 6

FATS • Fats consist of a wide group of compounds that are generally soluble in organic solvents and largely insoluble in water. • Chemically, fats are generally triesters of glycerol and fatty acids • Fats may be either solid or liquid at normal room temperature, depending on their structure and composition. 2/12/2022 7
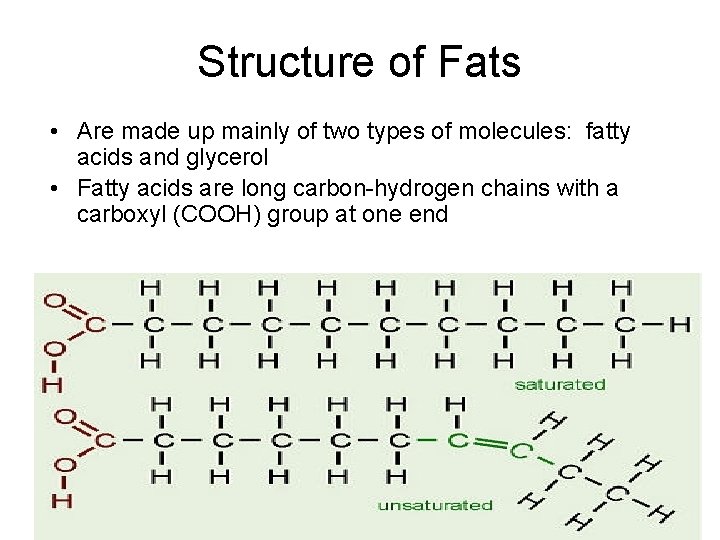
Structure of Fats • Are made up mainly of two types of molecules: fatty acids and glycerol • Fatty acids are long carbon-hydrogen chains with a carboxyl (COOH) group at one end 2/12/2022 8
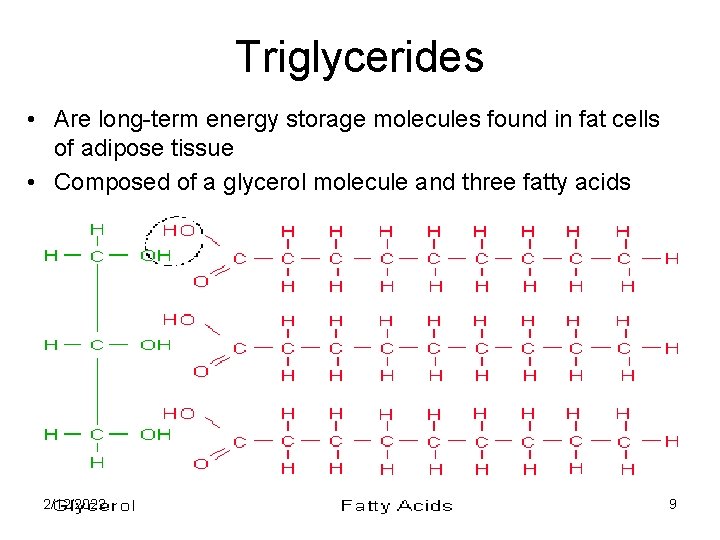
Triglycerides • Are long-term energy storage molecules found in fat cells of adipose tissue • Composed of a glycerol molecule and three fatty acids 2/12/2022 9
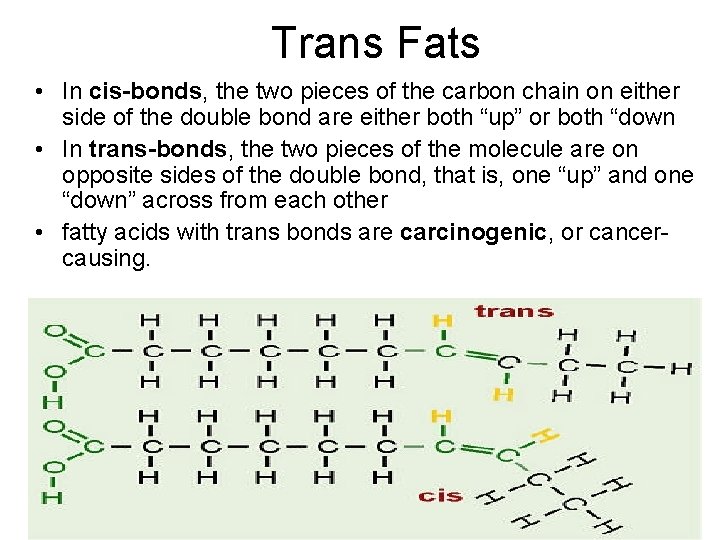
Trans Fats • In cis-bonds, the two pieces of the carbon chain on either side of the double bond are either both “up” or both “down • In trans-bonds, the two pieces of the molecule are on opposite sides of the double bond, that is, one “up” and one “down” across from each other • fatty acids with trans bonds are carcinogenic, or cancercausing. 2/12/2022 10
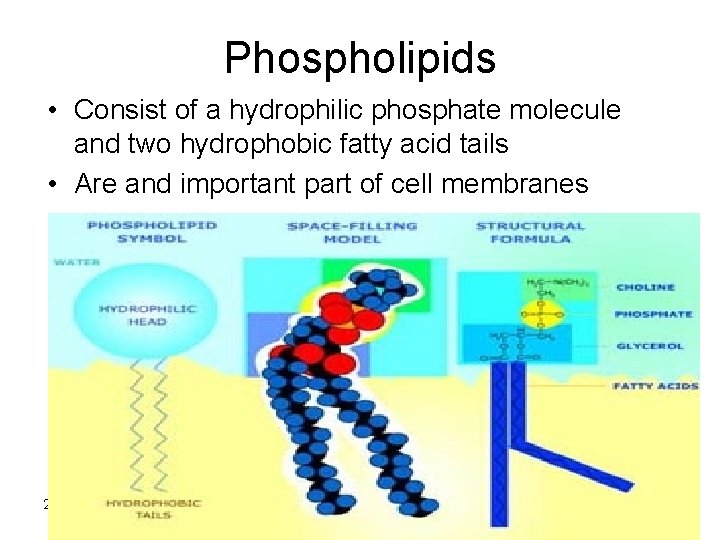
Phospholipids • Consist of a hydrophilic phosphate molecule and two hydrophobic fatty acid tails • Are and important part of cell membranes 2/12/2022 11
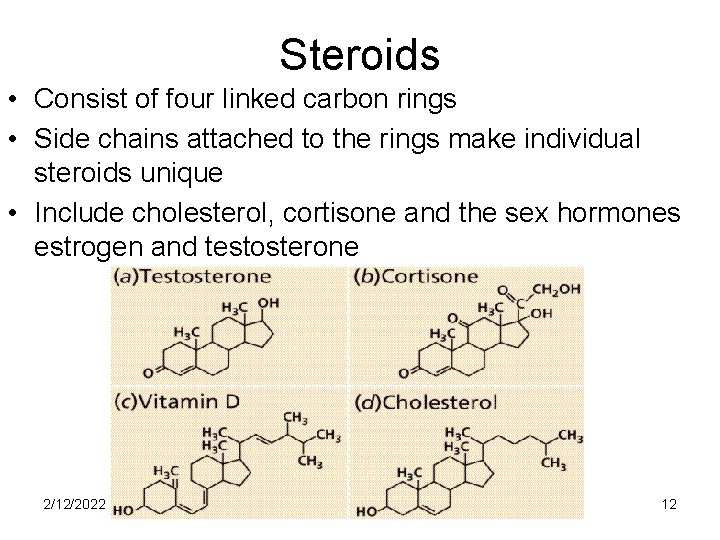
Steroids • Consist of four linked carbon rings • Side chains attached to the rings make individual steroids unique • Include cholesterol, cortisone and the sex hormones estrogen and testosterone 2/12/2022 12

Proteins • Direct and control the chemical reactions in life processes • Make up part of the cell membrane • Provide support and shape to cells • Function as hormones (chemical messengers) • Function as enzymes to speed up chemical reactions • Make up about ½ the body’s dry weight 2/12/2022 13

Amino Acids • Proteins are polymers of amino acids • AA contain C, H and O like carbohydrates and lipids but also contain N • All AA’s have same basic structure • Each AA varies only in the structure of the R group • There are 20 different AA’s, eight of which are essential 2/12/2022 14
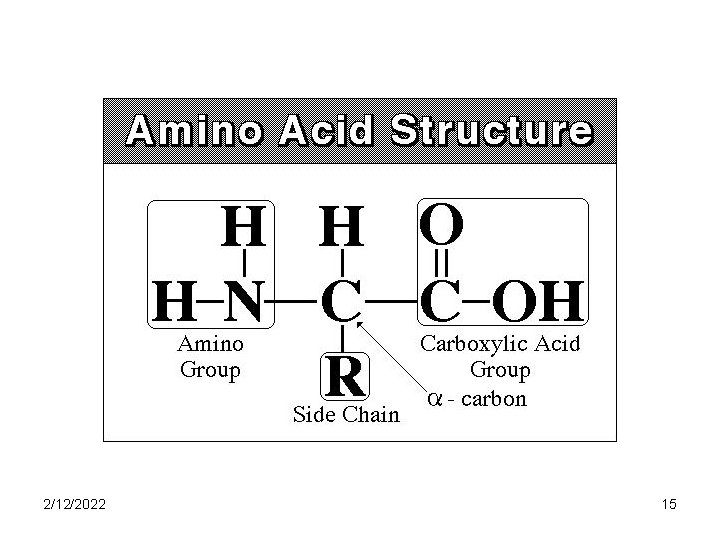
2/12/2022 15
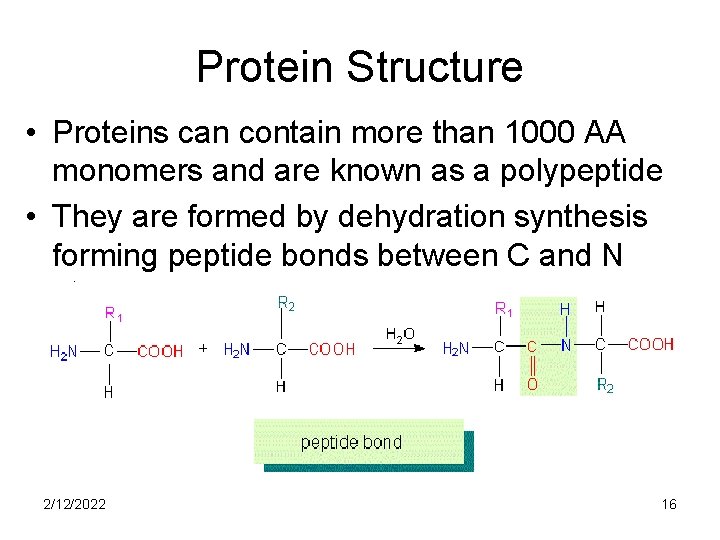
Protein Structure • Proteins can contain more than 1000 AA monomers and are known as a polypeptide • They are formed by dehydration synthesis forming peptide bonds between C and N atoms 2/12/2022 16

• Proper function of proteins is determined by shape • There are four different levels of protein structure: primary, secondary, tertiary and quaternary 1. 2. 3. 4. 2/12/2022 17
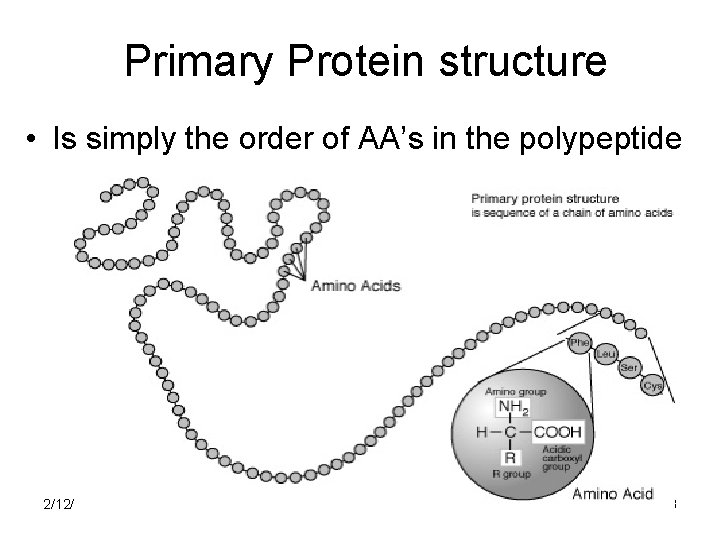
Primary Protein structure • Is simply the order of AA’s in the polypeptide 2/12/2022 18
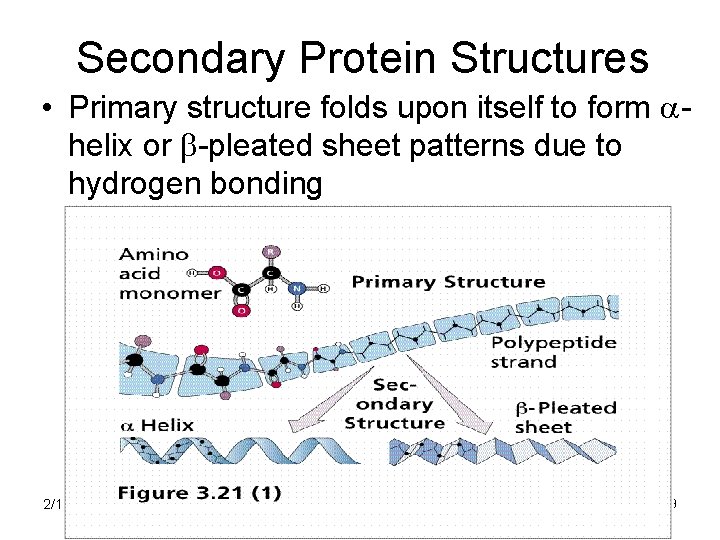
Secondary Protein Structures • Primary structure folds upon itself to form ahelix or b-pleated sheet patterns due to hydrogen bonding 2/12/2022 19

Tertiary Protein Structures • Are formed when the secondary structures fold over upon themselves and are held by covalent, ionic, hydrogen and non-polar bonds • The covalent bonds are disulfide bridges between Sulphur-containing AA’s 2/12/2022 20
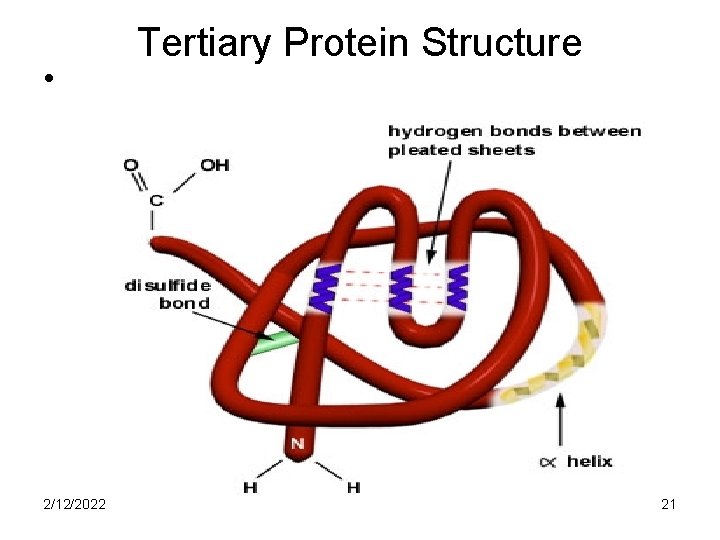
• 2/12/2022 Tertiary Protein Structure 21

Quaternary Protein Structure • Occurs when two or more polypeptide chains combine to form the protein 2/12/2022 22

Protein Denaturation • Exposing a protein to heat, radiation or change in p. H will alter its shape • Physical or chemical factors that disrupt the bonds between the amino acids changing the shape of the protein is called denaturing • Sometimes removing the factor allows the protein to assume is original shape • When the change is permanent coagulation has occurred 2/12/2022 23
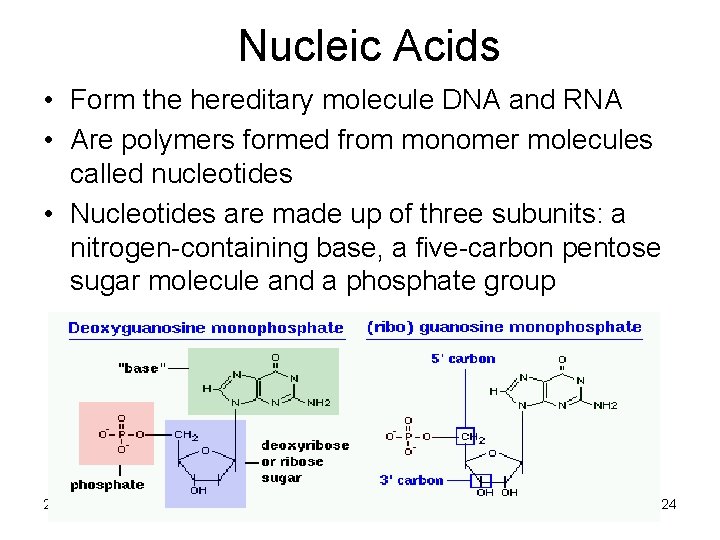
Nucleic Acids • Form the hereditary molecule DNA and RNA • Are polymers formed from monomer molecules called nucleotides • Nucleotides are made up of three subunits: a nitrogen-containing base, a five-carbon pentose sugar molecule and a phosphate group 2/12/2022 24

Nitrogenous bases • There are five nitrogenous bases, adenine, guanine (the purines), cytosine, thymine and uracil (the pyrimidines) • Thymine is present only in DNA and uracil is present only in RNA 2/12/2022 25
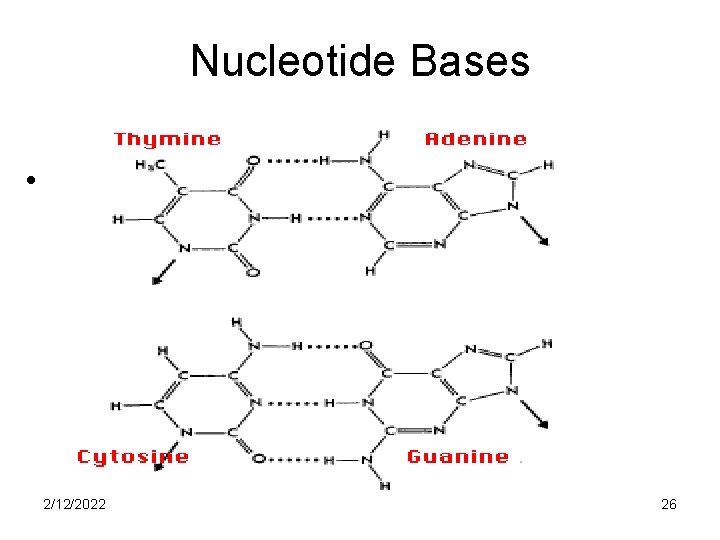
Nucleotide Bases • 2/12/2022 26
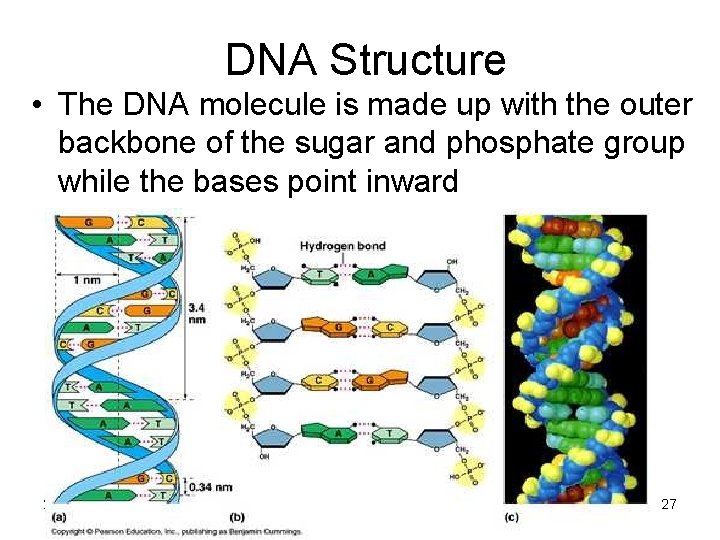
DNA Structure • The DNA molecule is made up with the outer backbone of the sugar and phosphate group while the bases point inward 2/12/2022 27
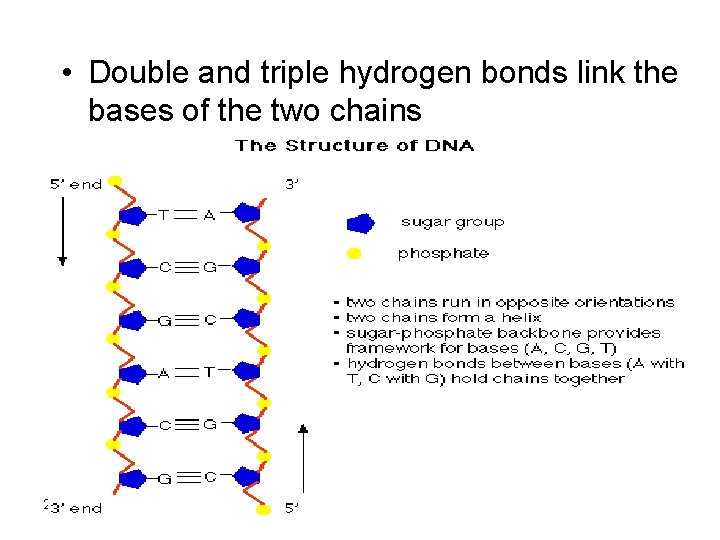
• Double and triple hydrogen bonds link the bases of the two chains 2/12/2022 28

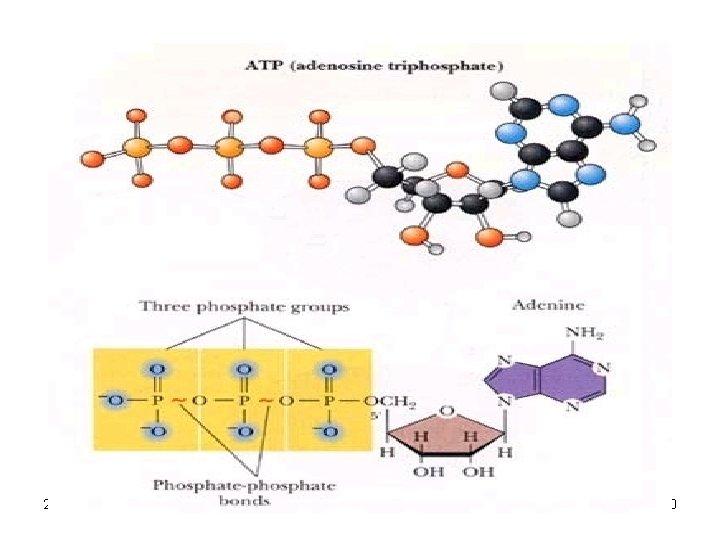
Adenosine Triphosphate (ATP) • Is an important nucleotide found in the cell • Is a monomer Consisting of three phosphate groups attached to the ribose • ATP is the energy providing molecule of the cell and when the third phosphate group is split off by hydrolysis a net release of energy results 2/12/2022 29
2/12/2022 30
- Slides: 30