Biochemistry Lets break this word down Bio Life

Biochemistry Let’s break this word down Bio = Life Chemistry = Chemicals Biochemistry = the chemicals of living things

Matter is made of atoms Atom –The smallest possible unit of an element

An atom is made of three particles 1. Neutrons- have no charge 2. Protons- have a positive charge 3. Electrons- have a negative charge
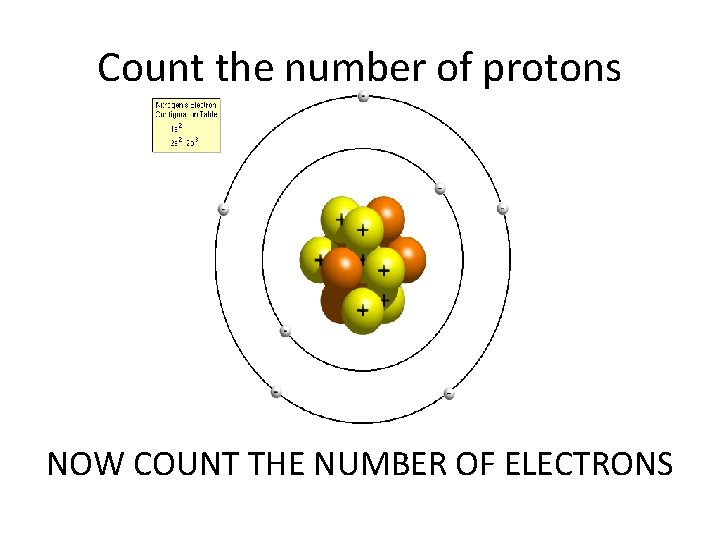
Count the number of protons NOW COUNT THE NUMBER OF ELECTRONS

CAN YOU IDENTIFY SOME OF THESE CHEMICAL SYMBOLS… I BET YOU KNOW MORE THAN YOU THINK!
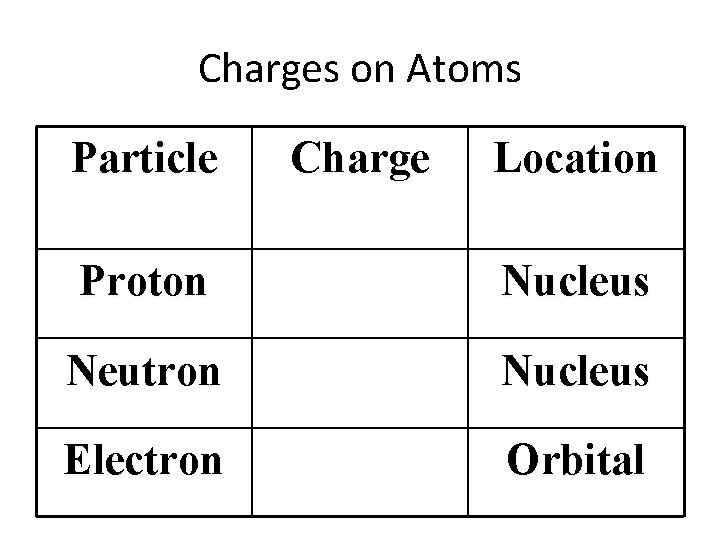
Charges on Atoms Particle Charge Location Proton Nucleus Neutron Nucleus Electron Orbital

Nucleus • Is the core of the atom where protons and neutrons are located. It is extremely small compared to the rest of the atom.

Electrons • Are found outside the nucleus • They travel around the nucleus in orbitals. Let’s Draw a Carbon Atom. . (go back to the Periodic Table of Elements if you need help!)

Elements • A pure substance that cannot be broken down into simpler substances by PHYSICAL or CHEMICAL means • Own set of unique properties –EXAMPLES: Cobalt, Hydrogen, Carbon, Iron

Compounds • A pure substance composed of two or more elements that are CHEMICALLY bonded: EXAMPLE: H 2 O (water) contains 2 atoms of hydrogen and 1 atom of oxygen
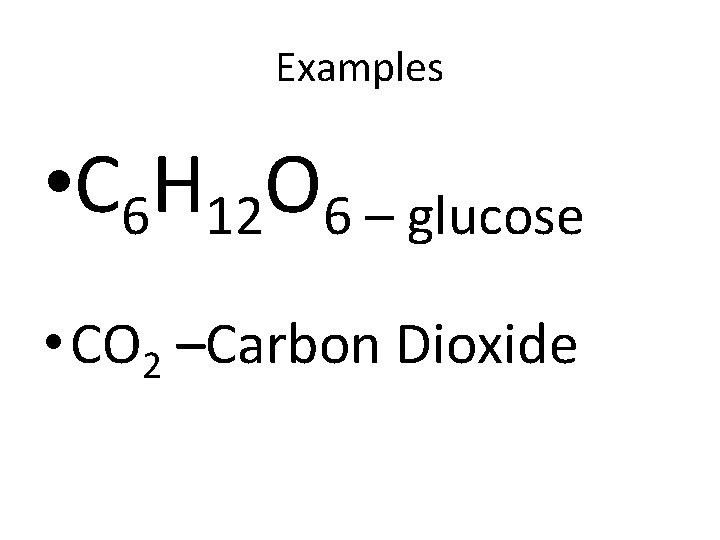
Examples • C 6 H 12 O 6 – glucose • CO 2 –Carbon Dioxide
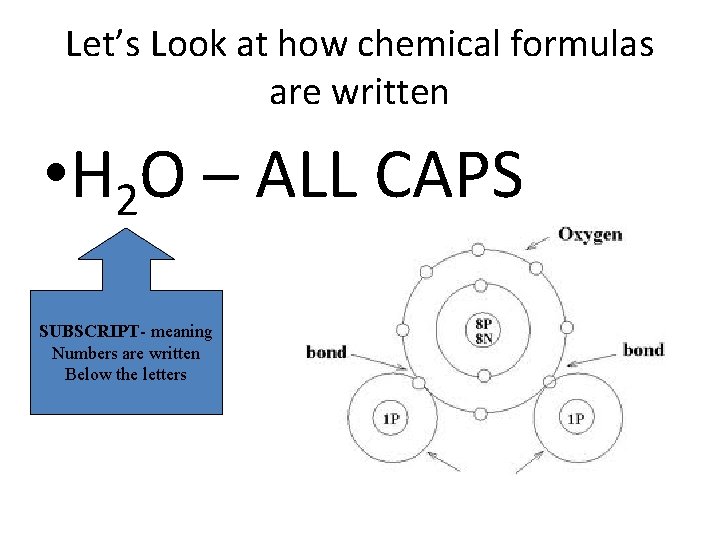
Let’s Look at how chemical formulas are written • H 2 O – ALL CAPS SUBSCRIPT- meaning Numbers are written Below the letters
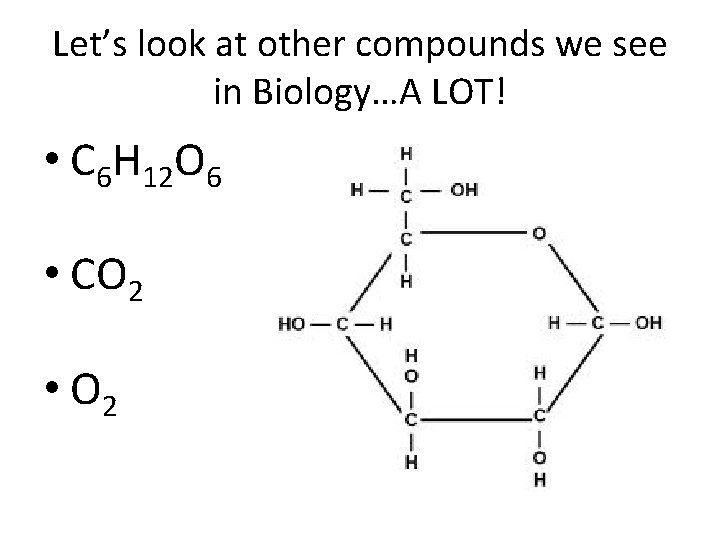
Let’s look at other compounds we see in Biology…A LOT! • C 6 H 12 O 6 • CO 2 • O 2

Chemical Bonds • Energy is required to put together compounds and energy is released when bonds are broken

ATP – Adenosine Triphosphate When we break the bond in between the phosphates. . we get our energy !

Two Types of Compounds ORGANIC - Must contain both Carbon and Hydrogen examples: ___C 6 H 12 O 6_(glucose) INORGANIC – everything else examples: CO 2 (carbon dioxide) H 2 O (water)

Circle the Inorganic Compounds • • NH 4 C 6 H 12 O 6 H 2 O CH 4 CO 2 HCl Na. Cl

Let’s 1 st look at Inorganic Compounds • Produced by non-living natural processes or in a laboratory • Important Compounds include: CO 2 , H 2 O, Na. Cl, O 2 • These compounds are needed in Plants and Animals in order to perform all important reactions such as Respiration, Photosynthesis, Digestion

Why is Water so important? • Water covers 3/4 ths of the Earth’s surface

Why is Water so important? • Most abundant compound in most living things

Why is Water so important? • Is the best solvent (things dissolve in it)

Why is Water so important? • Water expands as it freezes

Drawing a Water Molecule

Polarity of Water • Uneven distribution of charges

Cohesion • H 2 O sticks to H 2 O
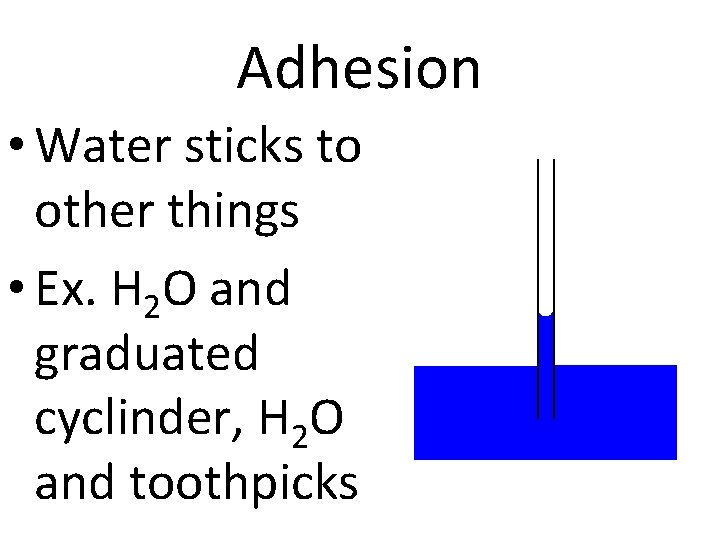
Adhesion • Water sticks to other things • Ex. H 2 O and graduated cyclinder, H 2 O and toothpicks

Surface Tension • Surface tension is an effect within the surface layer of a liquid that causes that layer to behave as an elastic sheet. • Ex. Insects to walk on water, water on a penny


Capillary Action • Tendency of water to rise in a thin tube

Solids, Liquids, Gases • Solid Ice • Liquid Water • Gas Water Vapor

p. H Scale • Measures the amount, or concentration of Hydrogen ions in a solution • Hydrogen ions = H+ • Hydroxide or Hydroxyl group = OH-

Acids and Bases • Acid contain higher concentration of H+ ions than H 2 O and have a p. H below 7 • Base contain lower concentration of H+ ions than H 2 O and have a p. H above 7
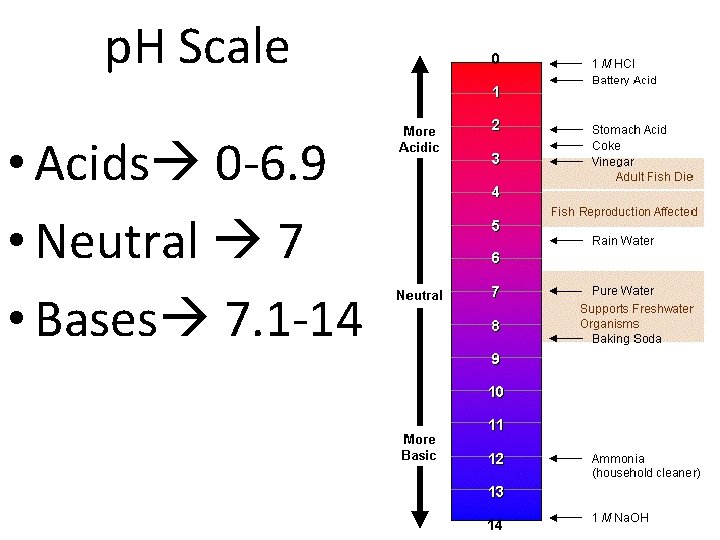
p. H Scale • Acids 0 -6. 9 • Neutral 7 • Bases 7. 1 -14
Let’s draw in H+ and OH- in the p. H scale
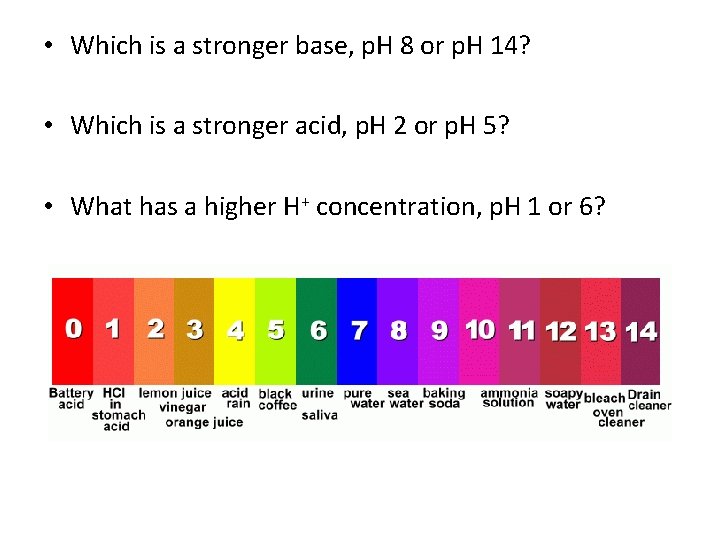
• Which is a stronger base, p. H 8 or p. H 14? • Which is a stronger acid, p. H 2 or p. H 5? • What has a higher H+ concentration, p. H 1 or 6?

CONCEPT CHECK! In many investigations, both in the laboratory and in natural environments, the p. H of substances is measured. Explain why p. H is important to living things. In your explanation be sure to: • identify one example of a life process of an organism that could be affected by a p. H change [1] • state one environmental problem that is directly related to p. H [1] * identify one possible cause of this environmental problem [1]
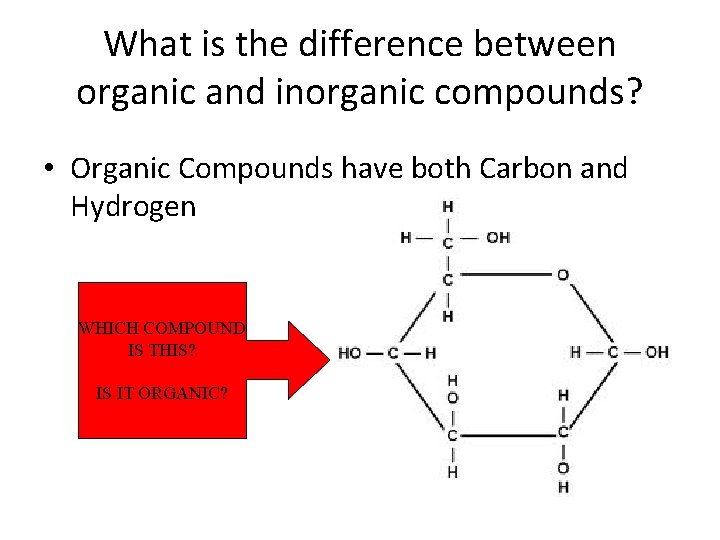
What is the difference between organic and inorganic compounds? • Organic Compounds have both Carbon and Hydrogen WHICH COMPOUND IS THIS? IS IT ORGANIC?

Four types of ORGANIC COMPOUNDS • • CARBOHYDRATES LIPIDS NUCLEIC ACIDS PROTEINS What comes to mind when you see some of these words?

Carbohydrates • Sugars that provide organisms with immediate energy • Glucose is used in cells to make ATP during cellular respiration

CONCEPT CHECK! In the transfer of energy from the Sun to ecosystems, which molecule is one of the first to store this energy? (1) protein (3) DNA (2) fat (4) glucose
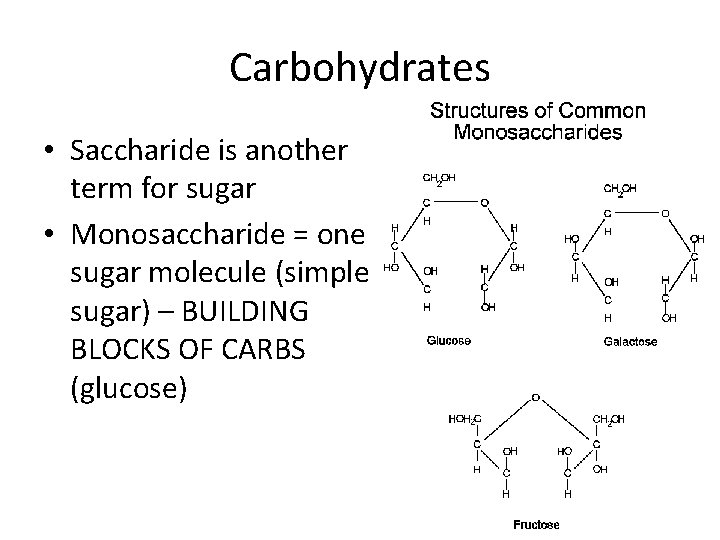
Carbohydrates • Saccharide is another term for sugar • Monosaccharide = one sugar molecule (simple sugar) – BUILDING BLOCKS OF CARBS (glucose)

Carbohydrates • Disaccharide = two sugars combined • Polysaccharide = many sugars combined
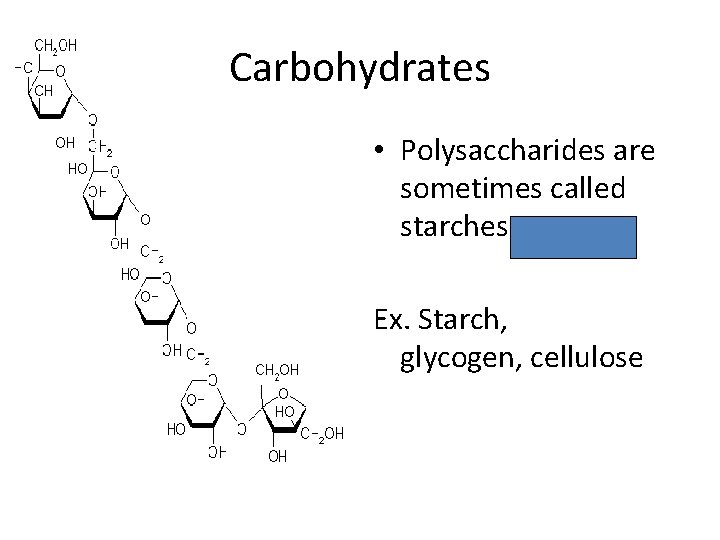
Carbohydrates • Polysaccharides are sometimes called starches Ex. Starch, glycogen, cellulose
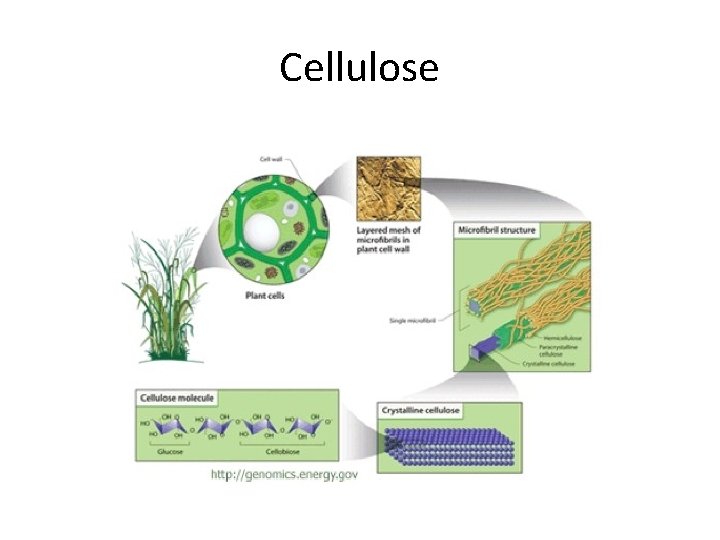
Cellulose

Is there anything that all these carbohydrates have in common? • • Sucrose Galactose Glucose Maltose Fructose Ribose Cellulose There subunit or building block is a simple sugar! All of them end in …OSE
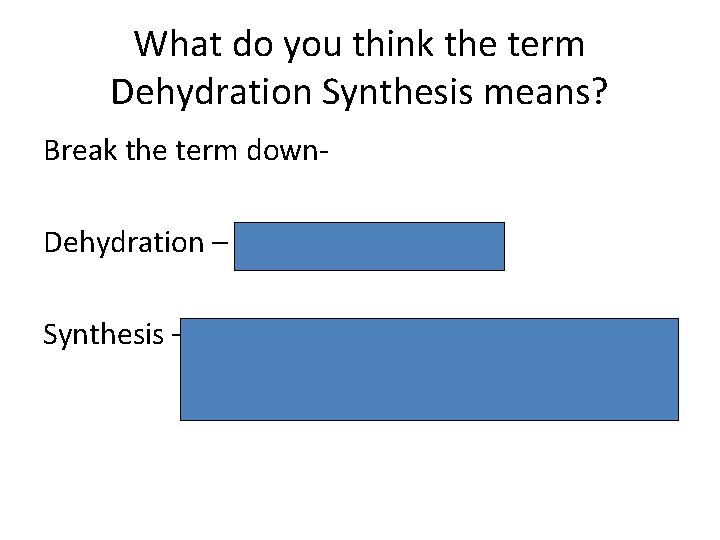
What do you think the term Dehydration Synthesis means? Break the term down. Dehydration – to take water away Synthesis – to put something together, to make something
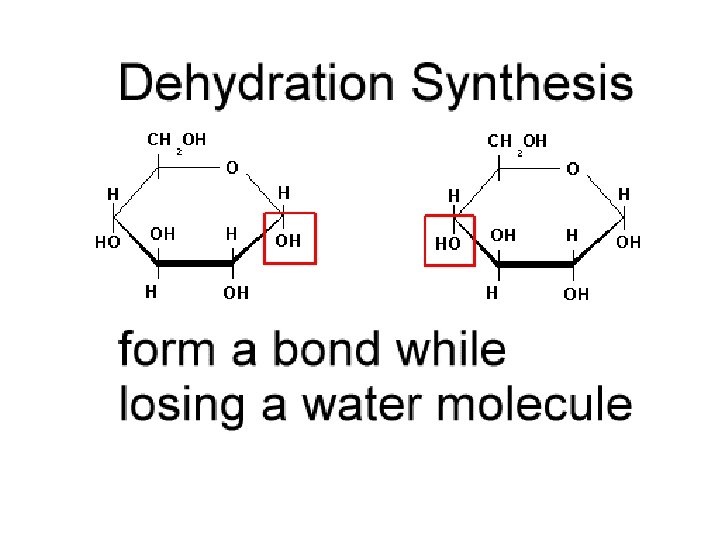
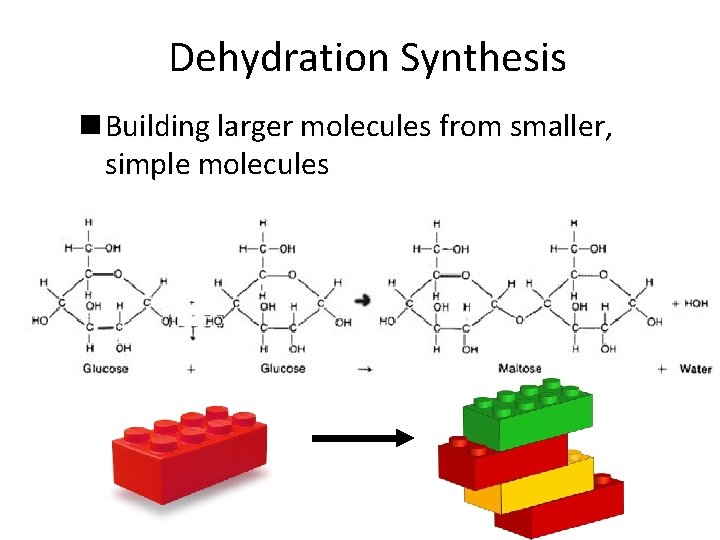
Dehydration Synthesis n Building larger molecules from smaller, simple molecules

Hydrolysis – to add water and break something apart
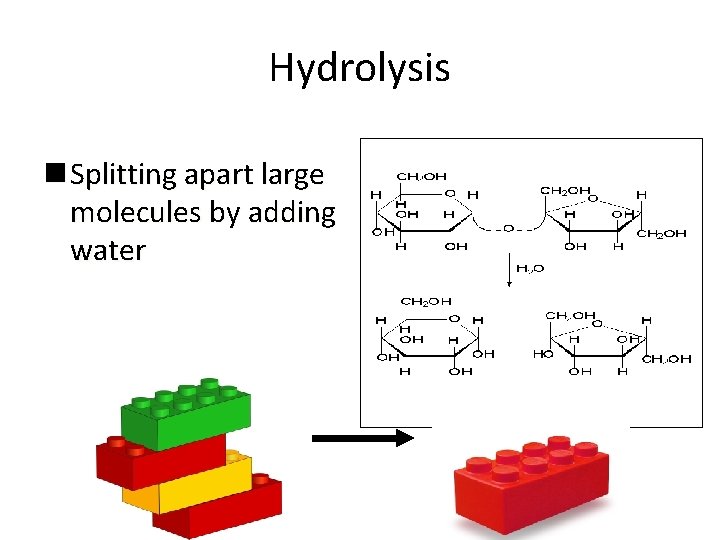
Hydrolysis n Splitting apart large molecules by adding water

CONCEPT CHECK! In plants, simple sugars are least likely to be (1) linked together to form proteins (2) broken down into carbon dioxide and water (3) used as a source of energy (4) stored in the form of starch molecules

Lipids • Source of stored Energy • Makes up part of the cell membrane

Lipids = Fats and Oils • Food sources of lipids • Butter, Oil, Nuts

Lipids • Fatty Acids - Building block or subunit of Lipids • Glycerol – also helps build a lipid
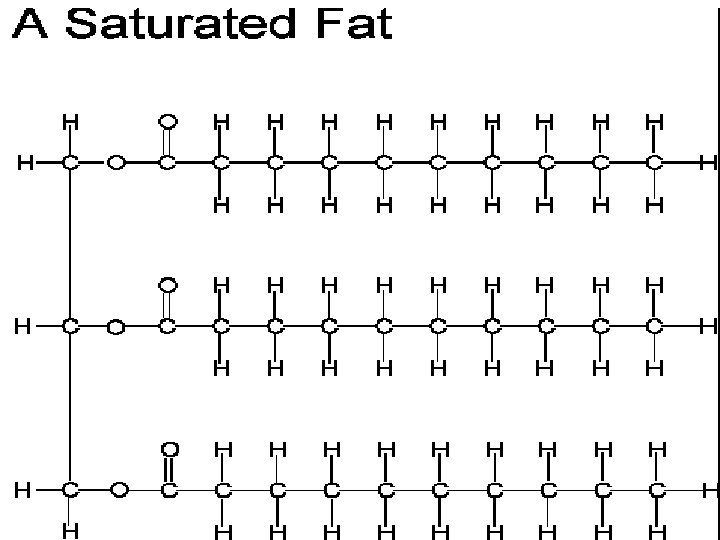
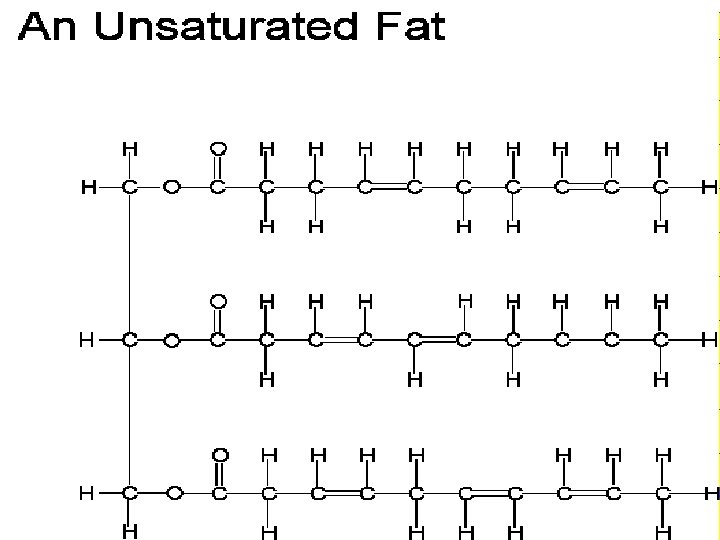
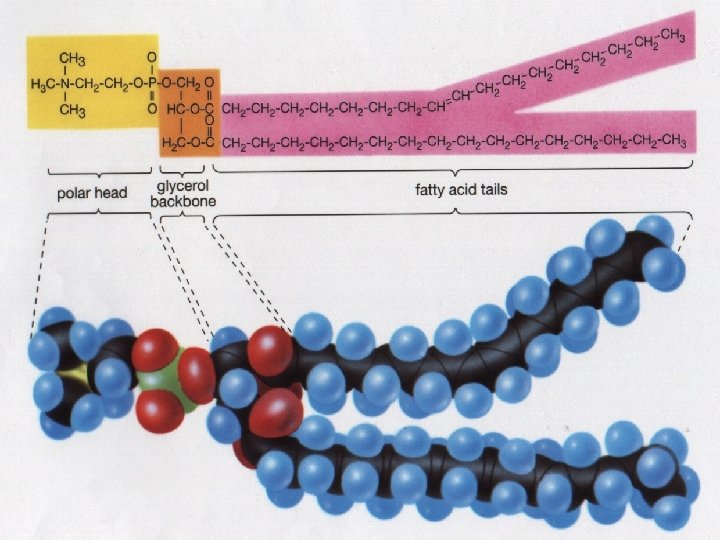
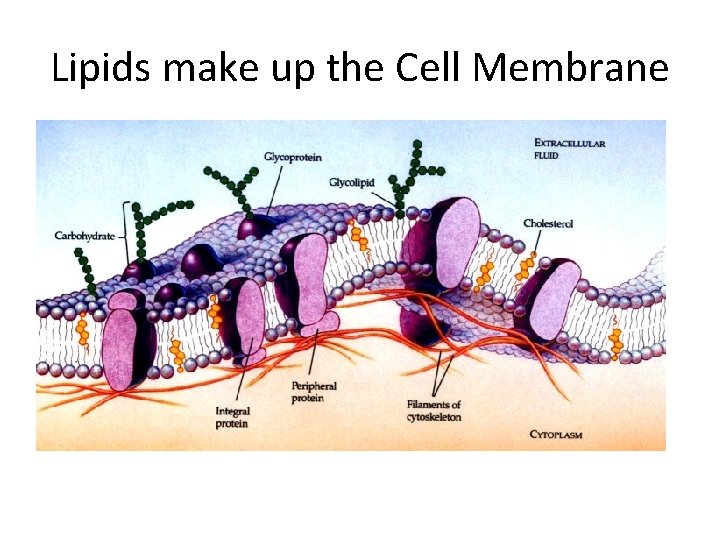
Lipids make up the Cell Membrane
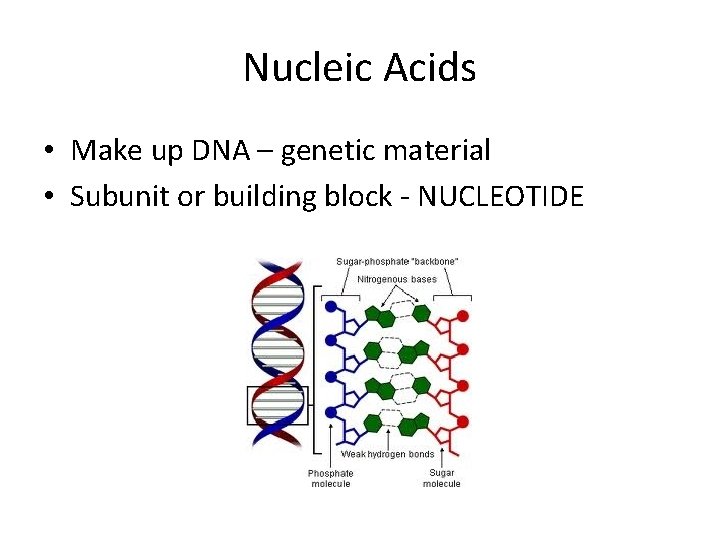
Nucleic Acids • Make up DNA – genetic material • Subunit or building block - NUCLEOTIDE
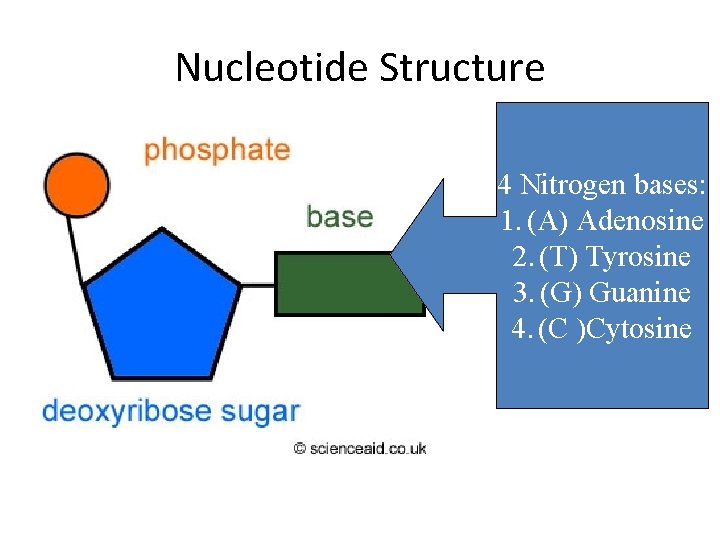
Nucleotide Structure 4 Nitrogen bases: 1. (A) Adenosine 2. (T) Tyrosine 3. (G) Guanine 4. (C )Cytosine

Nucleotides making up DNA Look at the different nitrogen bases:
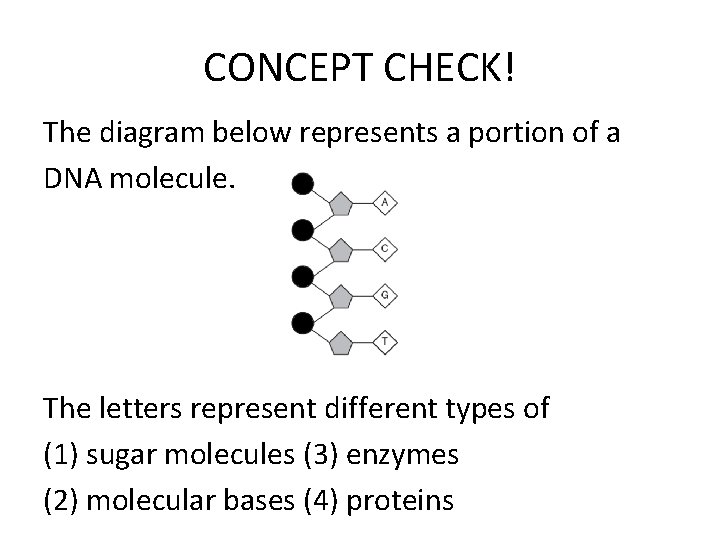
CONCEPT CHECK! The diagram below represents a portion of a DNA molecule. The letters represent different types of (1) sugar molecules (3) enzymes (2) molecular bases (4) proteins
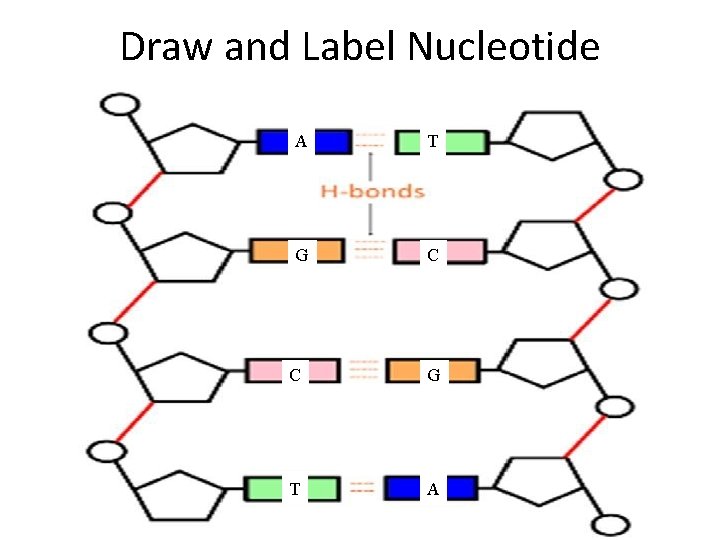
Draw and Label Nucleotide A T G C C G T A

Underneath Concept Check Question – Write the Following • IN DNA, • A always pairs with T • G always pairs with C • *A way to remember it AT Garbage Can
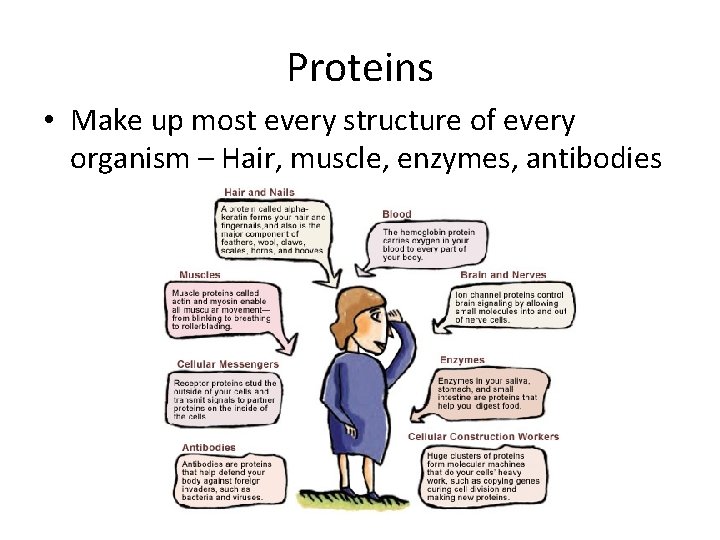
Proteins • Make up most every structure of every organism – Hair, muscle, enzymes, antibodies

Protein Structure • Proteins building blocks, or subunits are called AMINO ACIDS

Food Sources of Protein • • Meat Lentils Tofu Beans

CONCEPT CHECK! Three days after an organism eats some meat, many of the organic molecules originally contained in the meat would be found in newly formed molecules of (1) glucose (3) starch (2) protein (4) oxygen

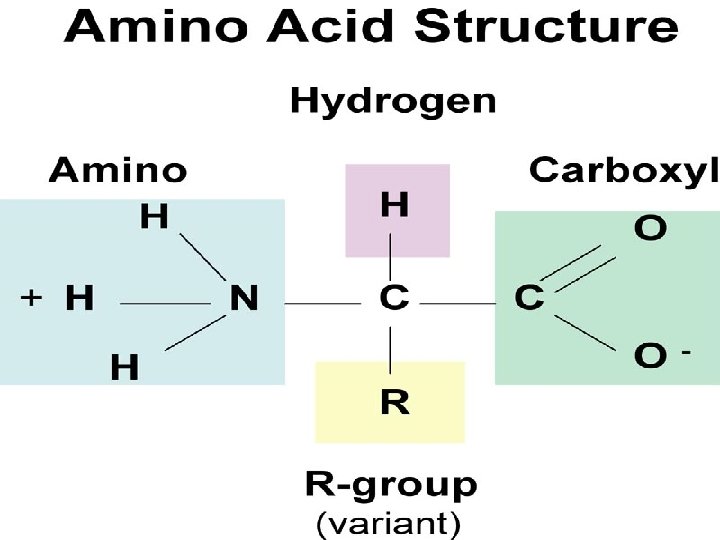
Amino Acid Structure • Four groups 1. amino group 2. carboxyl group 3. Hydrogen group 4. Functional or variable group (R group)

Different Types of Amino Acids

Proteins • One single amino acid is called Monopeptide • Two Dipeptide • Many Polypeptide

• Proteins are held together by peptide bonds
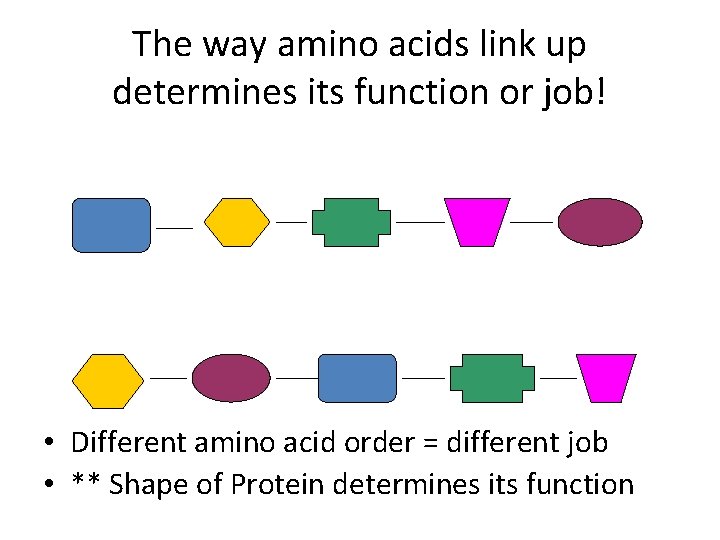
The way amino acids link up determines its function or job! • Different amino acid order = different job • ** Shape of Protein determines its function
Draw an amino acid chain here:

CONCEPT CHECK! Many biological catalysts, proteins, and enzymes are similar in that, in order to function properly, they must (1) interact with each other at a high p. H (2) interact with molecules that can alter their specific bonding patterns (3) contain amino acid chains that fold into a specific shape (4) contain identical DNA base sequences


CONCEPT CHECK! Two proteins in the same cell perform different functions. This is because the two proteins are composed of (1) chains folded the same way and the same sequence of simple sugars (2) chains folded the same way and the same sequence of amino acids (3) chains folded differently and a different sequence of simple sugars (4) chains folded differently and a different sequence of amino acids
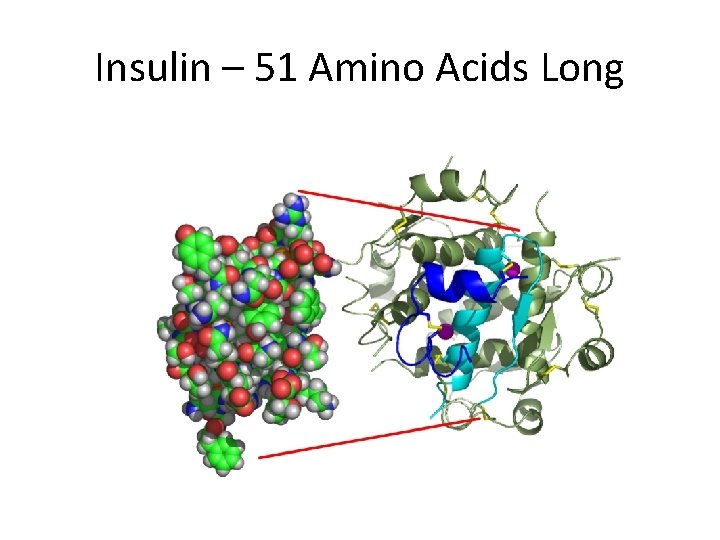
Insulin – 51 Amino Acids Long
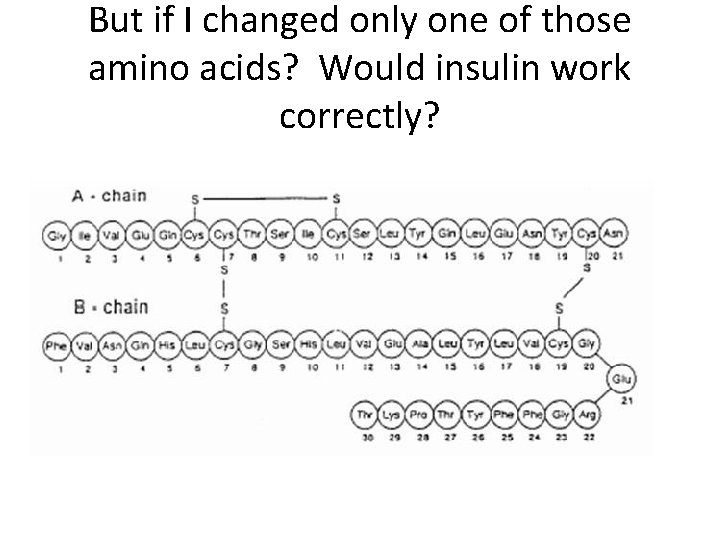
But if I changed only one of those amino acids? Would insulin work correctly?
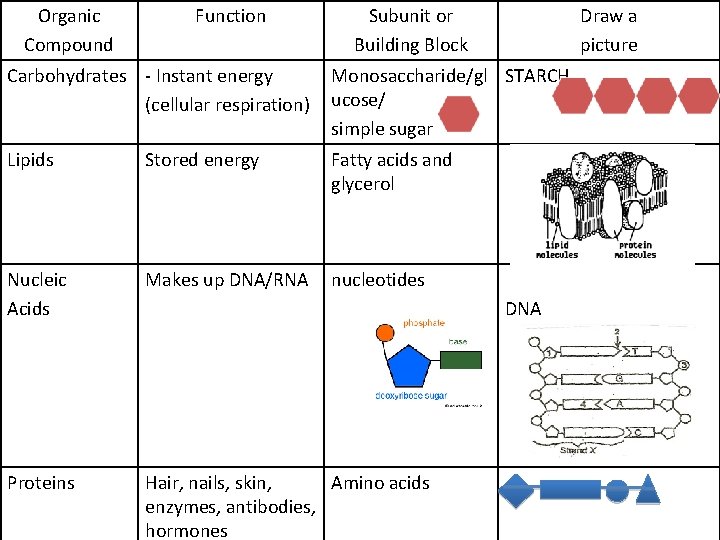
Organic Compound Function Subunit or Building Block Draw a picture Carbohydrates - Instant energy (cellular respiration) Monosaccharide/gl STARCH ucose/ simple sugar Lipids Stored energy Fatty acids and glycerol Nucleic Acids Makes up DNA/RNA nucleotides Proteins Hair, nails, skin, Amino acids enzymes, antibodies, hormones DNA

Enzymes ENZYMES ARE SUPER COOL!!!
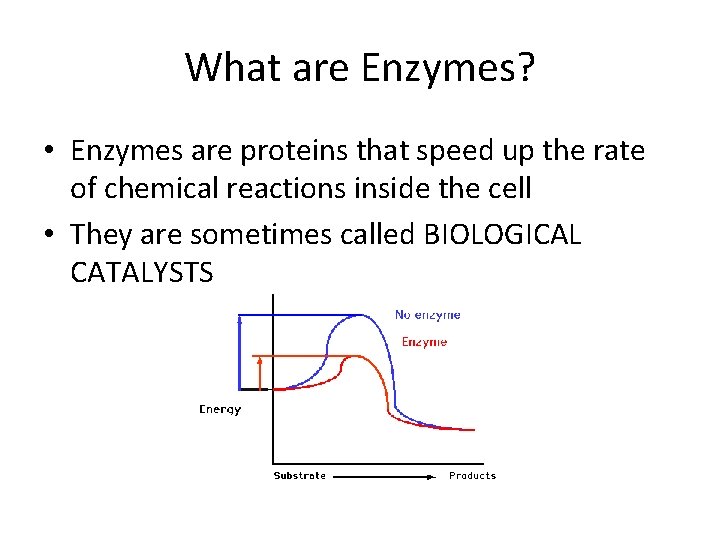
What are Enzymes? • Enzymes are proteins that speed up the rate of chemical reactions inside the cell • They are sometimes called BIOLOGICAL CATALYSTS

What are Enzymes? • Enzymes put things together – SYNTHESIS • Enzymes break things apart – DIGESTION/HYDROLYSIS http: //www. kscience. co. uk /animations/anim_2. htm

Enzymes and chemical reactions • All cells in particular ours, perform a ton of chemical reactions like CELLULAR RESPIRATION • CELLULAR RESPIRATION = ATP • NO ENZYMES = NO ENERGY = YOUR DEAD

WOW…ENZYMES SEEM KIND OF IMPORTANT…LET’S GO BACK A SEC!
Protein Structure • Proteins are made up of AMINO ACIDS

Enzymes are one type of Protein • Enzymes are made up of AMINO ACIDS • There are __20______ types of Amino Acids
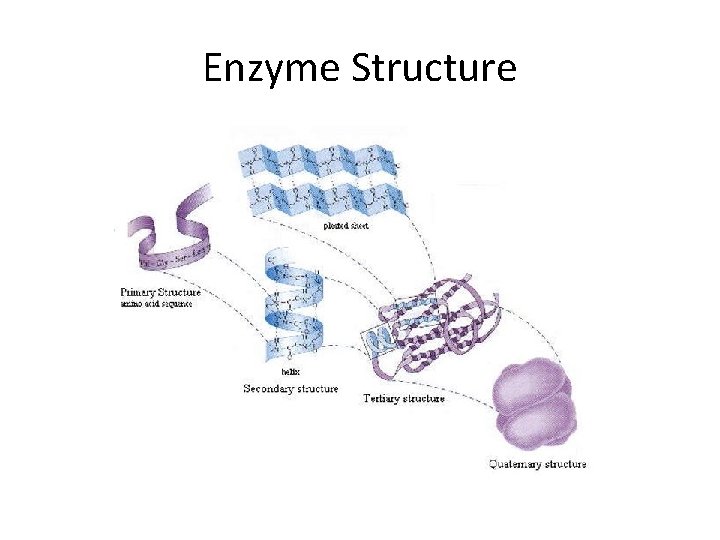
Enzyme Structure
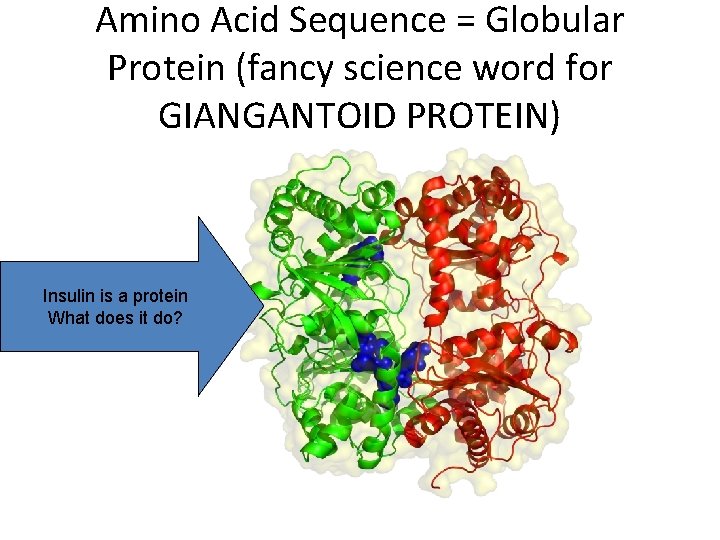
Amino Acid Sequence = Globular Protein (fancy science word for GIANGANTOID PROTEIN) Insulin is a protein What does it do?

Amino Acids • The order or sequence of AMINO ACIDS determines the function or job of an enzyme • The shape of the protein determines its function!!!!

OK we’ve seen two different Enzymes already…how many more are there? USE WORD TO FIGURE ONE • In nature there. ASSOCIATIONS are thousands of THIS different OUT! OTHERWISE YOU WOULD HAVE TO MEMORIZE THEM an ALL!!!! enzymes present that help organism Yikes! maintain homeostasis • TIP…. most ENZYMES end in ASE • Amylase breaks down Starch • Protease breaks down Proteins • Lipase breaks down ____LIPIDS____

Enzyme Specificity • Individual enzymes are HIGHLY SPECIFIC • Meaning an enzyme has only one job
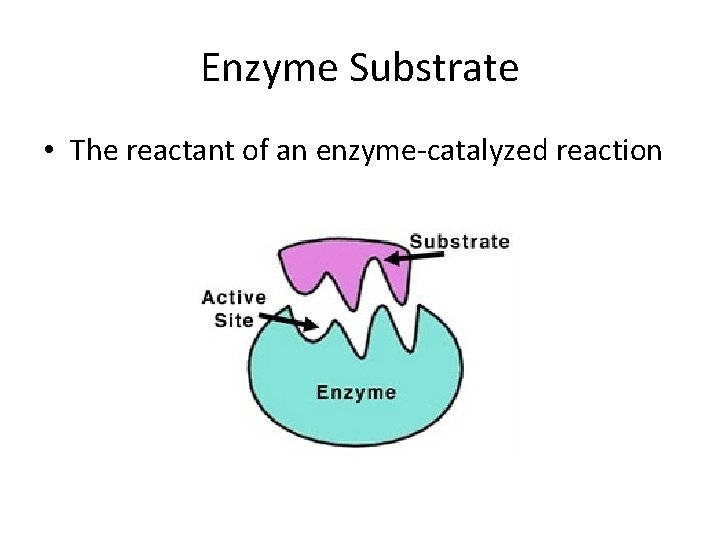
Enzyme Substrate • The reactant of an enzyme-catalyzed reaction
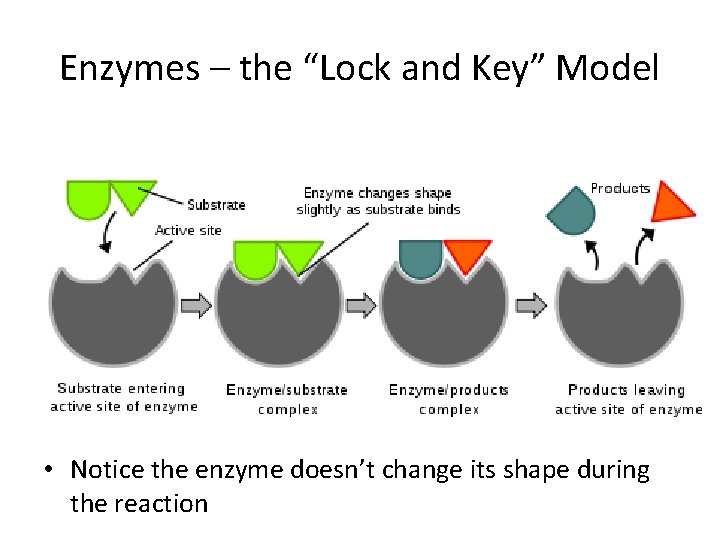
Enzymes – the “Lock and Key” Model • Notice the enzyme doesn’t change its shape during the reaction

Now let’s draw our own Enzyme Reaction…WOW…THIS IS TOO MUCH FUN FOR ONE DAY! Substrates Products Enzyme. Substrate Complex Enzyme Be sure to label the Enzyme, substrate, enzymesubstrate complex, and products

FACTORS THAT EFFECT ENYZMES • Proteins, or enzymes, can loose their shape • DENATURE – To change the shape of a protein substrate enzyme substrate Denatured enzyme
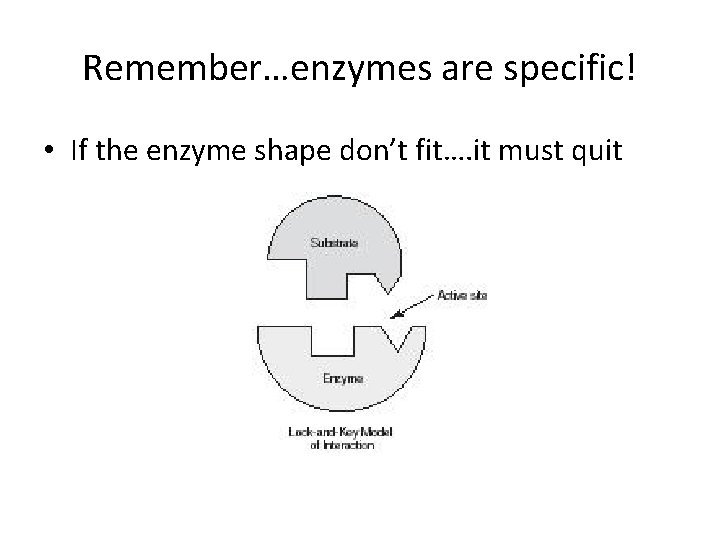
Remember…enzymes are specific! • If the enzyme shape don’t fit…. it must quit
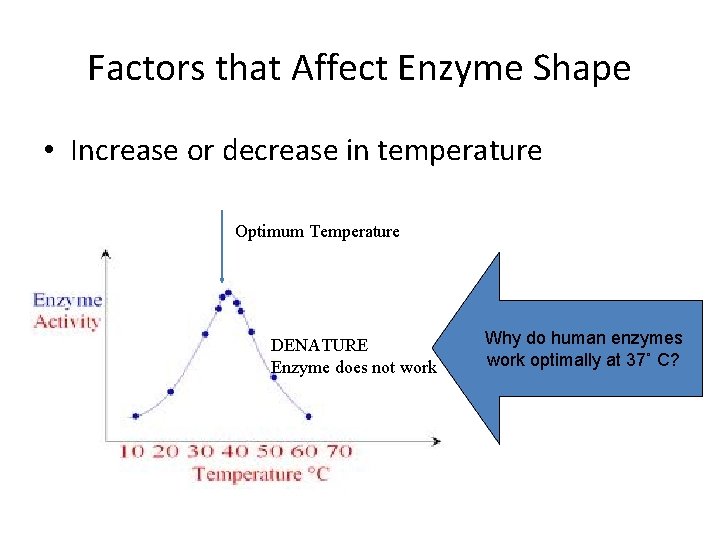
Factors that Affect Enzyme Shape • Increase or decrease in temperature Optimum Temperature DENATURE Enzyme does not work Why do human enzymes work optimally at 37˚ C?
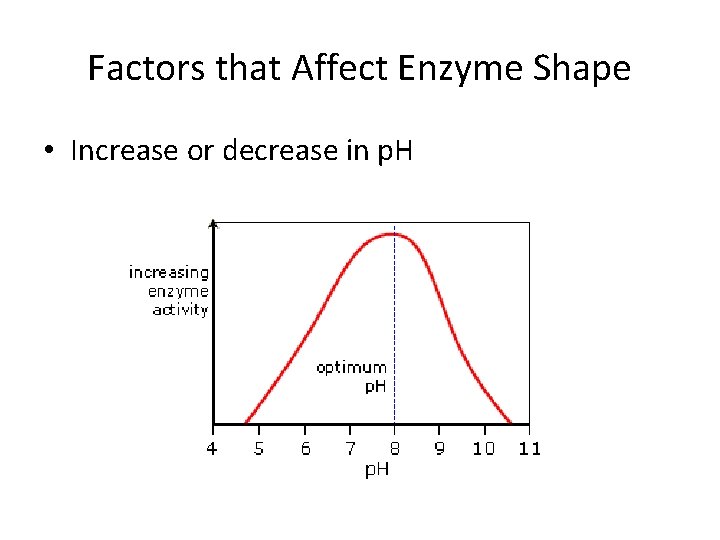
Factors that Affect Enzyme Shape • Increase or decrease in p. H

CONCEPT CHECK! The failure to regulate the p. H of the blood can affect the activity of (1) enzymes that clot blood (2) red blood cells that make antibodies (3) chlorophyll that carries oxygen in the blood (4) DNA that controls starch digestion in the blood
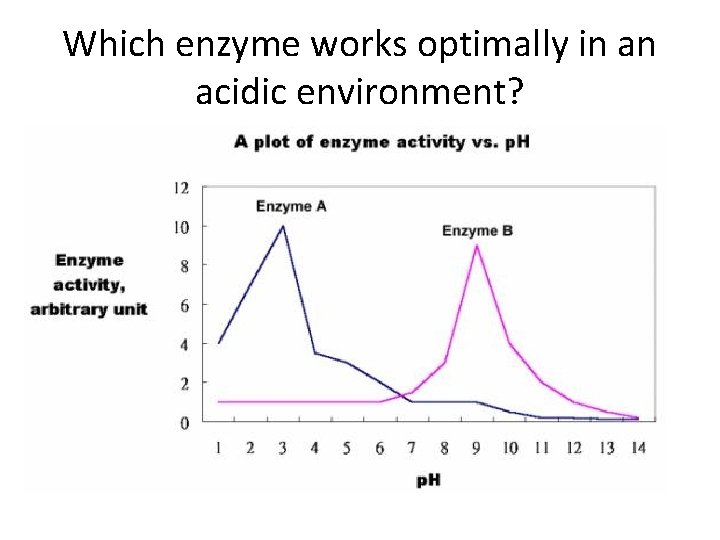
Which enzyme works optimally in an acidic environment?
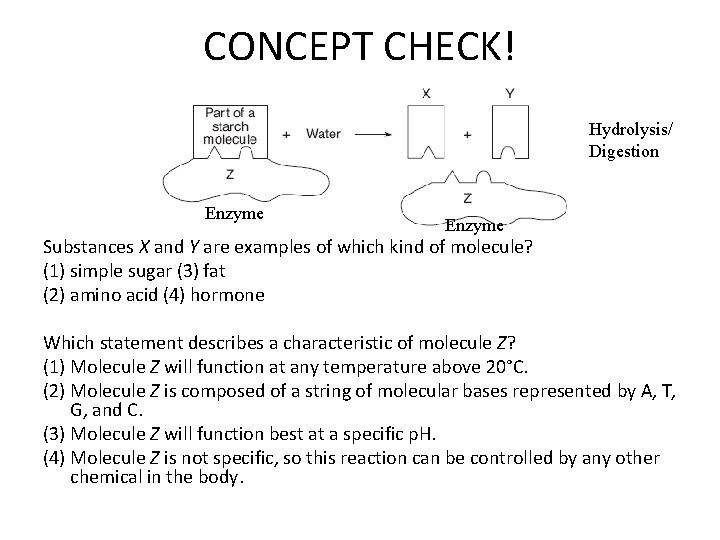
CONCEPT CHECK! Hydrolysis/ Digestion Enzyme Substances X and Y are examples of which kind of molecule? (1) simple sugar (3) fat (2) amino acid (4) hormone Which statement describes a characteristic of molecule Z? (1) Molecule Z will function at any temperature above 20°C. (2) Molecule Z is composed of a string of molecular bases represented by A, T, G, and C. (3) Molecule Z will function best at a specific p. H. (4) Molecule Z is not specific, so this reaction can be controlled by any other chemical in the body.

NOW YOU CAN CREATE YOUR OWN ENZYMES! NICE WORK!
- Slides: 104