Biochemistry Lectures rd 3 stage Anbar UniversityCollege of

Biochemistry Lectures rd 3 stage Anbar University-College of Pharmacy-Clinical Laboratory Sciences Department Dr. Esraa. A. AL-ANI 2019 -2020

carbohydrates • carbohydrates, one of the four major classes of biomolecules along with proteins, nucleic acids, and lipids. Carbohydrates are aldehyde or ketone compounds with multiple hydroxyl groups. • They make up most of the organic matter on Earth because of their extensive roles in all forms of life : 1 - carbohydrates serve as energy stores, fuels, and metabolic intermediates. 2 - ribose and deoxyribose sugars form part of the structural framework of RNA and DNA. 3 - polysaccharides are structural elements in the cell walls of bacteria and plants cellulose, the main constituent of plant cell walls, is one of the most abundant organic compounds in the biosphere. 4 - carbohydrates are linked to many proteins and lipids, where they play key roles in mediating interactions among cells and interactions between cells and other elements in the cellular environment. • A key related property of carbohydrates in their role as mediators of cellular interactions is the tremendous structural diversity possible within this class of molecules. • Carbohydrates are built from monosaccharides, small molecules that typically contain from three to nine carbon atoms and vary in size and in the stereochemical configuration at one or more carbon centers. • These monosaccharides may be linked together to form a large variety of oligo saccharide structures.
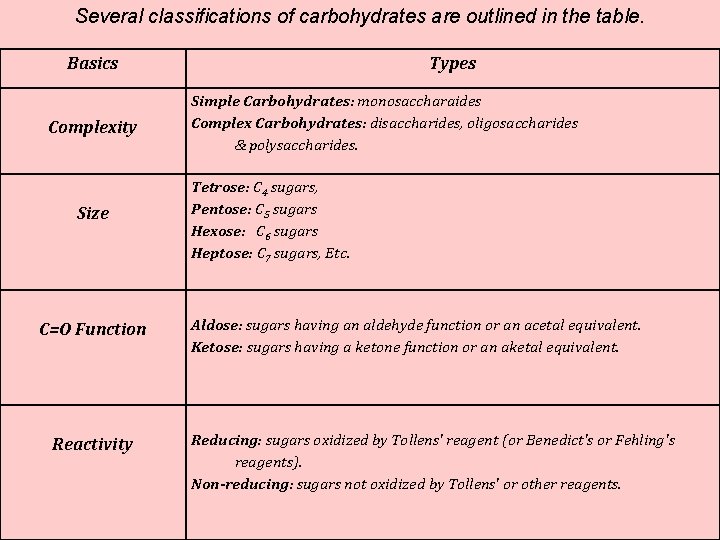
Several classifications of carbohydrates are outlined in the table. Basics Complexity Size C=O Function Reactivity Types Simple Carbohydrates: monosaccharaides Complex Carbohydrates: disaccharides, oligosaccharides & polysaccharides. Tetrose: C 4 sugars, Pentose: C 5 sugars Hexose: C 6 sugars Heptose: C 7 sugars, Etc. Aldose: sugars having an aldehyde function or an acetal equivalent. Ketose: sugars having a ketone function or an aketal equivalent. Reducing: sugars oxidized by Tollens' reagent (or Benedict's or Fehling's reagents). Non-reducing: sugars not oxidized by Tollens' or other reagents.
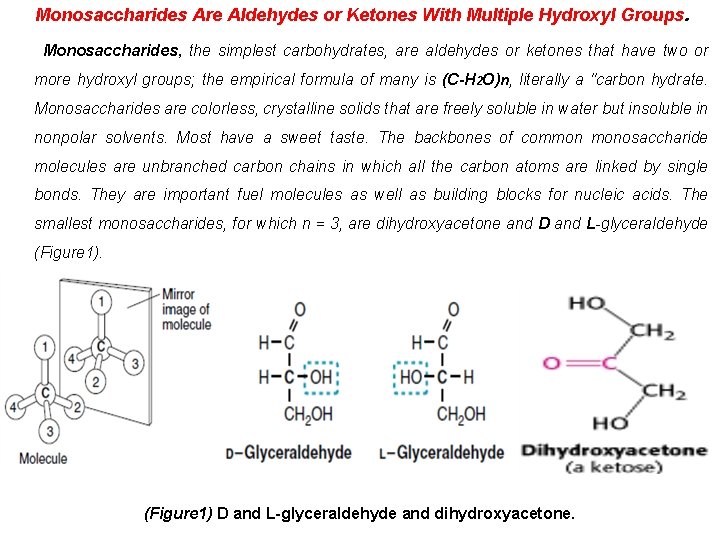
Monosaccharides Are Aldehydes or Ketones With Multiple Hydroxyl Groups. Monosaccharides, the simplest carbohydrates, are aldehydes or ketones that have two or more hydroxyl groups; the empirical formula of many is (C-H 2 O)n, literally a "carbon hydrate. Monosaccharides are colorless, crystalline solids that are freely soluble in water but insoluble in nonpolar solvents. Most have a sweet taste. The backbones of common monosaccharide molecules are unbranched carbon chains in which all the carbon atoms are linked by single bonds. They are important fuel molecules as well as building blocks for nucleic acids. The smallest monosaccharides, for which n = 3, are dihydroxyacetone and D and L-glyceraldehyde (Figure 1) D and L-glyceraldehyde and dihydroxyacetone.
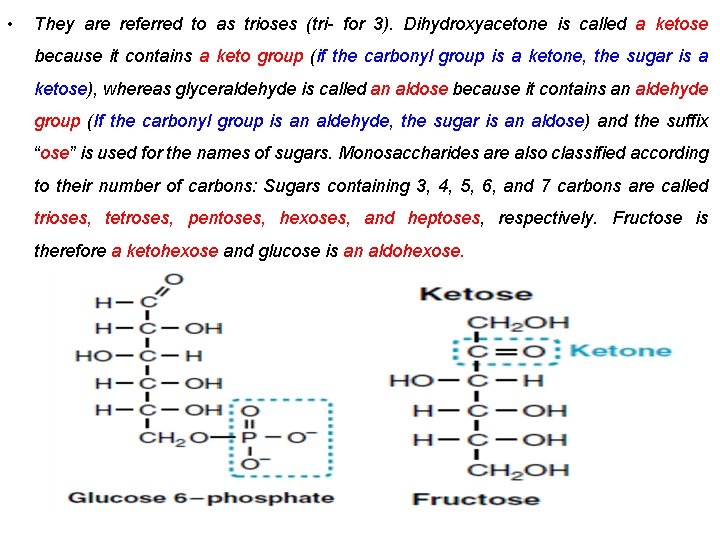
• They are referred to as trioses (tri- for 3). Dihydroxyacetone is called a ketose because it contains a keto group (if the carbonyl group is a ketone, the sugar is a ketose), whereas glyceraldehyde is called an aldose because it contains an aldehyde group (If the carbonyl group is an aldehyde, the sugar is an aldose) and the suffix “ose” is used for the names of sugars. Monosaccharides are also classified according to their number of carbons: Sugars containing 3, 4, 5, 6, and 7 carbons are called trioses, tetroses, pentoses, hexoses, and heptoses, respectively. Fructose is therefore a ketohexose and glucose is an aldohexose.
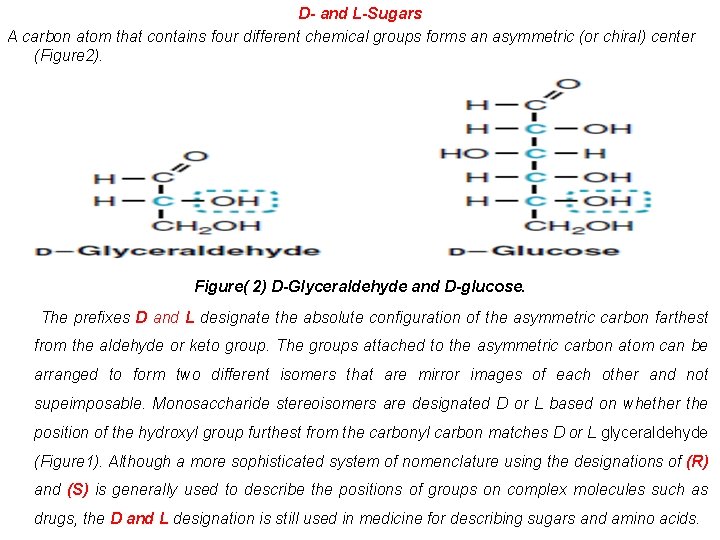
D- and L-Sugars A carbon atom that contains four different chemical groups forms an asymmetric (or chiral) center (Figure 2). Figure( 2) D-Glyceraldehyde and D-glucose. The prefixes D and L designate the absolute configuration of the asymmetric carbon farthest from the aldehyde or keto group. The groups attached to the asymmetric carbon atom can be arranged to form two different isomers that are mirror images of each other and not supeimposable. Monosaccharide stereoisomers are designated D or L based on whether the position of the hydroxyl group furthest from the carbonyl carbon matches D or L glyceraldehyde (Figure 1). Although a more sophisticated system of nomenclature using the designations of (R) and (S) is generally used to describe the positions of groups on complex molecules such as drugs, the D and L designation is still used in medicine for describing sugars and amino acids.
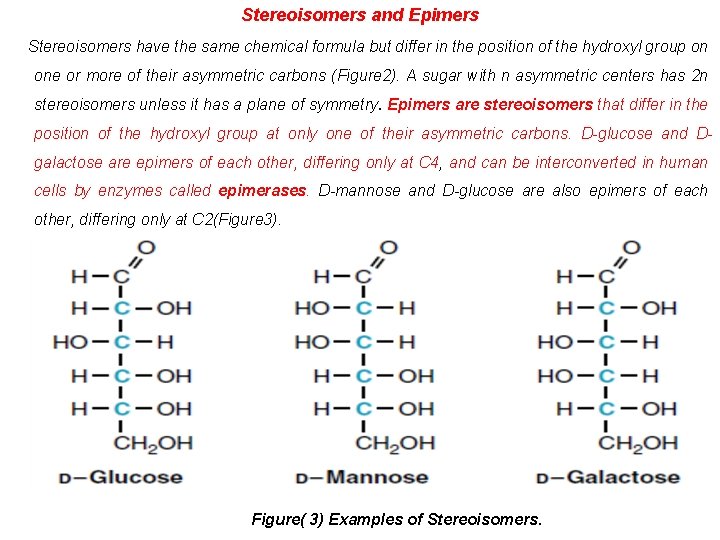
Stereoisomers and Epimers Stereoisomers have the same chemical formula but differ in the position of the hydroxyl group on one or more of their asymmetric carbons (Figure 2). A sugar with n asymmetric centers has 2 n stereoisomers unless it has a plane of symmetry. Epimers are stereoisomers that differ in the position of the hydroxyl group at only one of their asymmetric carbons. D-glucose and Dgalactose are epimers of each other, differing only at C 4, and can be interconverted in human cells by enzymes called epimerases. D-mannose and D-glucose are also epimers of each other, differing only at C 2(Figure 3). Figure( 3) Examples of Stereoisomers.
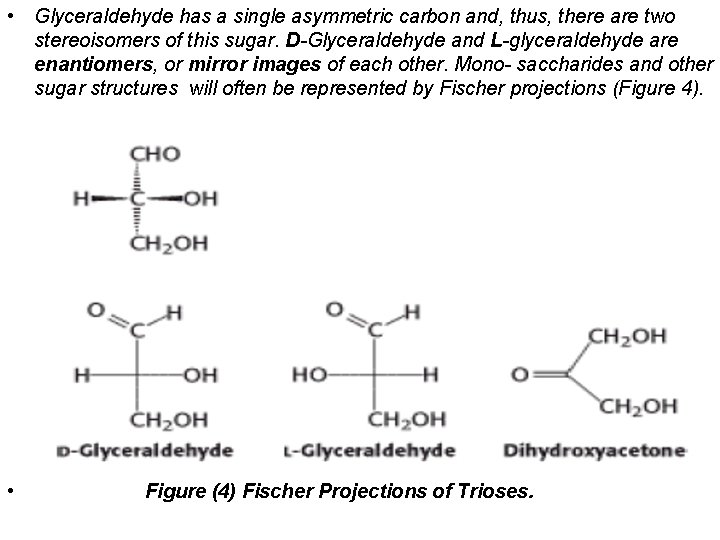
• Glyceraldehyde has a single asymmetric carbon and, thus, there are two stereoisomers of this sugar. D-Glyceraldehyde and L-glyceraldehyde are enantiomers, or mirror images of each other. Mono- saccharides and other sugar structures will often be represented by Fischer projections (Figure 4). • Figure (4) Fischer Projections of Trioses.

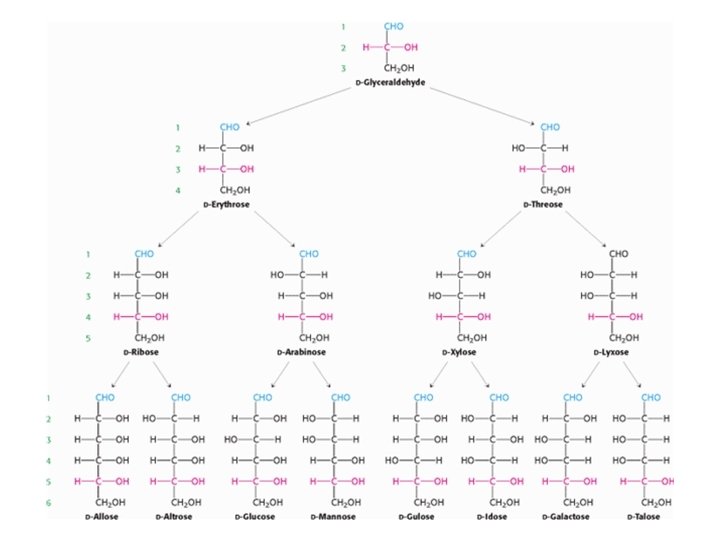
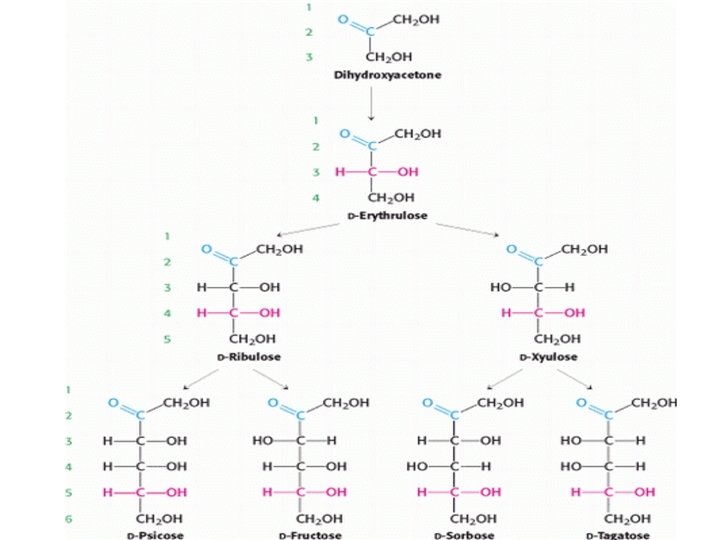
• In a Fischer projection of a molecule, atoms joined to an asymmetric carbon atom by horizontal bonds are in front of the plane of the page, and those joined by vertical bonds are behind. • Fischer projections are useful for depicting carbohydrate structures because they provide clear and simple views of the stereochemistry at each carbon center. • Because simple monosaccharides have multiple asymmetric carbons, they exist as diastereoisomers, isomers that are not mirror images of each other, as well as enantiomers. • Figure (5) shows the common d-aldose sugars. d-Ribose, the carbohydrate component of RNA, is a five-carbon aldose. d-Glucose, d-mannose, and dgalactose are considerable six-carbon aldoses. • Note Dihydroxyacetone is the simplest ketose and ketoses have one fewer asymmetric center than do aldoses with the same number of carbons. DFructose is the most abundant ketohexose. • The stereochemical relation between d-ketoses containing as many as six carbon atoms are shown in Figure (6).
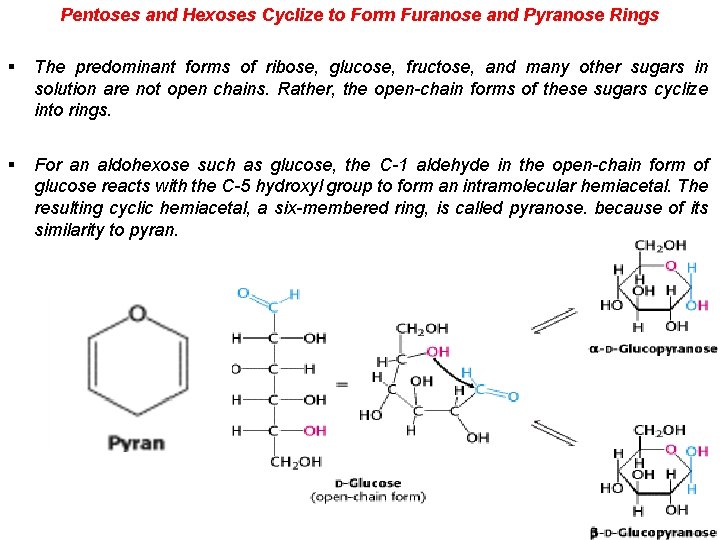
Pentoses and Hexoses Cyclize to Form Furanose and Pyranose Rings § The predominant forms of ribose, glucose, fructose, and many other sugars in solution are not open chains. Rather, the open-chain forms of these sugars cyclize into rings. § For an aldohexose such as glucose, the C-1 aldehyde in the open-chain form of glucose reacts with the C-5 hydroxyl group to form an intramolecular hemiacetal. The resulting cyclic hemiacetal, a six-membered ring, is called pyranose. because of its similarity to pyran.
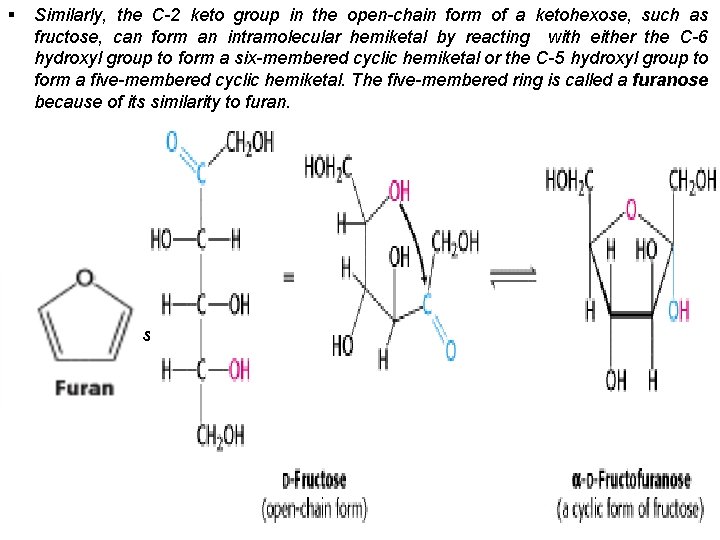
§ Similarly, the C-2 keto group in the open-chain form of a ketohexose, such as fructose, can form an intramolecular hemiketal by reacting with either the C-6 hydroxyl group to form a six-membered cyclic hemiketal or the C-5 hydroxyl group to form a five-membered cyclic hemiketal. The five-membered ring is called a furanose because of its similarity to furan. § The depictions of glucopyranose and fructofuranose are Haworth projections.

v An additional asymmetric center is created when a cyclic hemiacetal is formed. In glucose, C-1, the carbonyl carbon atom in the open-chain form, becomes an asymm etric center. Thus, two ring structures can be formed: α-d-glucopyranose & β-d-gluco pyranose. v For d sugars drawn as Haworth projections, the designation α means that the hydr oxyl group attached to C-1 is below the plane of the ring, β means that it is above the plane of the ring. v The C-1 carbon atom is called the anomeric carbon atom, and the α and β forms are called anomers. An equilibrium mixture of glucose contains approximately one-third α anomer, two-thirds β anomer, and <1% of the open chain form. v The same nomenclature applies to the furanose ring form of fructose, except that α and β refer to the hydroxyl groups attached to C-2, the anomeric carbon atom. v In solution, the hydroxyl group on the anomeric carbon spontaneously (non enzym atically) changes from the α to the β position through a process called mutarotation. When the ring opens, the straight chain aldehyde or ketone is formed. When the ring closes, the hydroxyl group may be in either the α or the β position. This process occurs more rapidly in the presence of cellular enzymes called mutarotases.
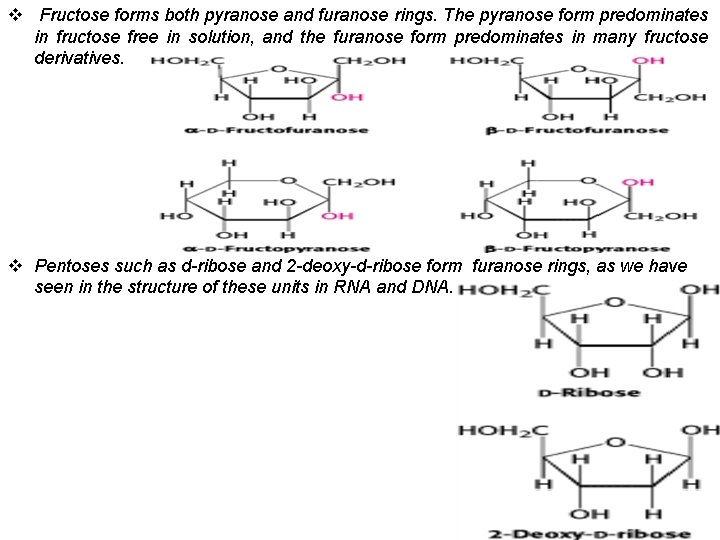
v Fructose forms both pyranose and furanose rings. The pyranose form predominates in fructose free in solution, and the furanose form predominates in many fructose derivatives. v Pentoses such as d-ribose and 2 -deoxy-d-ribose form furanose rings, as we have seen in the structure of these units in RNA and DNA.
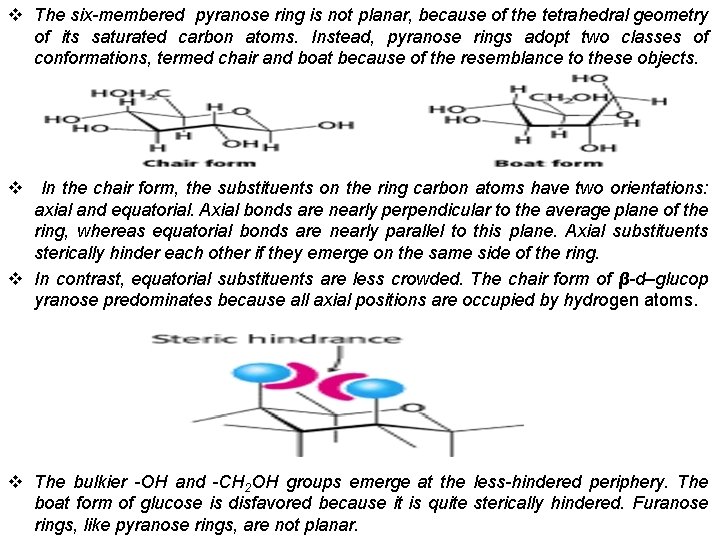
v The six-membered pyranose ring is not planar, because of the tetrahedral geometry of its saturated carbon atoms. Instead, pyranose rings adopt two classes of conformations, termed chair and boat because of the resemblance to these objects. v In the chair form, the substituents on the ring carbon atoms have two orientations: axial and equatorial. Axial bonds are nearly perpendicular to the average plane of the ring, whereas equatorial bonds are nearly parallel to this plane. Axial substituents sterically hinder each other if they emerge on the same side of the ring. v In contrast, equatorial substituents are less crowded. The chair form of β-d–glucop yranose predominates because all axial positions are occupied by hydrogen atoms. v The bulkier -OH and -CH 2 OH groups emerge at the less-hindered periphery. The boat form of glucose is disfavored because it is quite sterically hindered. Furanose rings, like pyranose rings, are not planar.
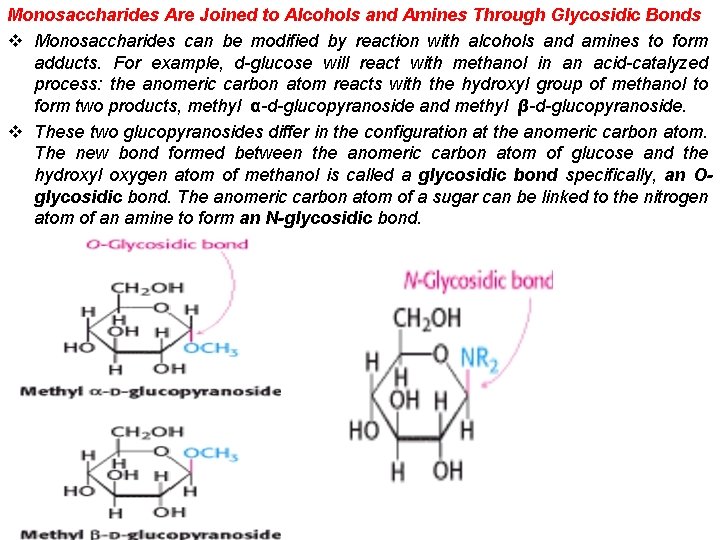
Monosaccharides Are Joined to Alcohols and Amines Through Glycosidic Bonds v Monosaccharides can be modified by reaction with alcohols and amines to form adducts. For example, d-glucose will react with methanol in an acid-catalyzed process: the anomeric carbon atom reacts with the hydroxyl group of methanol to form two products, methyl α-d-glucopyranoside and methyl β-d-glucopyranoside. v These two glucopyranosides differ in the configuration at the anomeric carbon atom. The new bond formed between the anomeric carbon atom of glucose and the hydroxyl oxygen atom of methanol is called a glycosidic bond specifically, an Oglycosidic bond. The anomeric carbon atom of a sugar can be linked to the nitrogen atom of an amine to form an N-glycosidic bond.
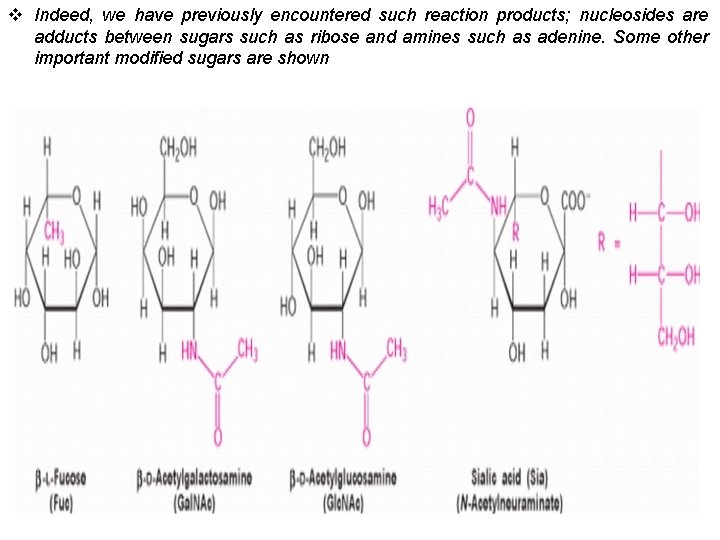
v Indeed, we have previously encountered such reaction products; nucleosides are adducts between sugars such as ribose and amines such as adenine. Some other important modified sugars are shown
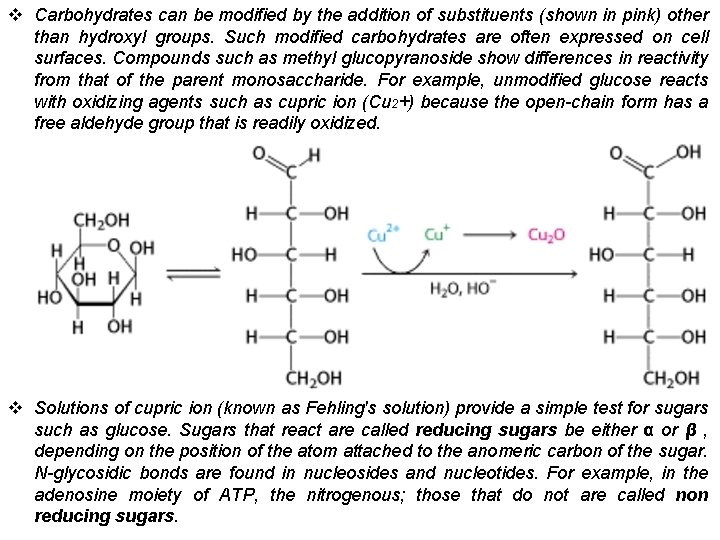
v Carbohydrates can be modified by the addition of substituents (shown in pink) other than hydroxyl groups. Such modified carbohydrates are often expressed on cell surfaces. Compounds such as methyl glucopyranoside show differences in reactivity from that of the parent monosaccharide. For example, unmodified glucose reacts with oxidizing agents such as cupric ion (Cu 2+) because the open-chain form has a free aldehyde group that is readily oxidized. v Solutions of cupric ion (known as Fehling's solution) provide a simple test for sugars such as glucose. Sugars that react are called reducing sugars be either α or β , depending on the position of the atom attached to the anomeric carbon of the sugar. N-glycosidic bonds are found in nucleosides and nucleotides. For example, in the adenosine moiety of ATP, the nitrogenous; those that do not are called non reducing sugars.
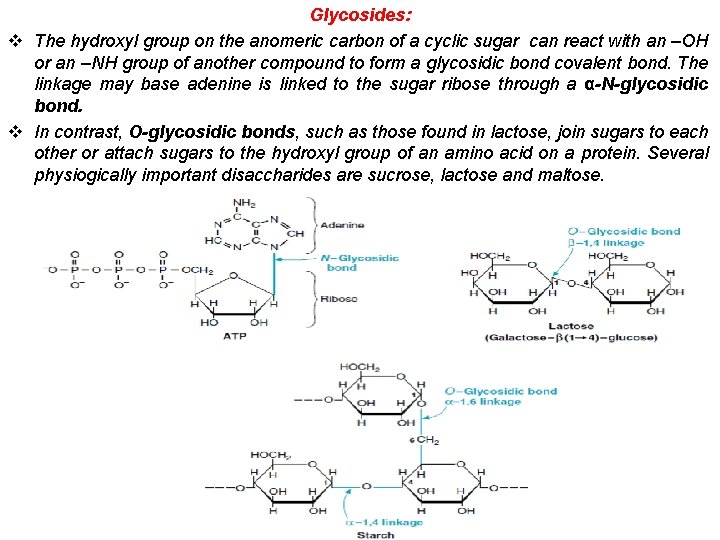
Glycosides: v The hydroxyl group on the anomeric carbon of a cyclic sugar can react with an –OH or an –NH group of another compound to form a glycosidic bond covalent bond. The linkage may base adenine is linked to the sugar ribose through a α-N-glycosidic bond. v In contrast, O-glycosidic bonds, such as those found in lactose, join sugars to each other or attach sugars to the hydroxyl group of an amino acid on a protein. Several physiogically important disaccharides are sucrose, lactose and maltose.

Disaccharides and Oligosaccharides v The fact that monosaccharides have multiple hydroxyl groups means that various glycosidic linkages are possible. A disaccharide contains two monosaccharides join ed by an O-glycosidic bond. Three abundant disaccharides are sucrose, lactose, and maltose (common dietary components). v Sucrose (common table sugar) is obtained commercially from cane or beet. The anomeric carbon atoms of a glucose unit and a fructose unit are joined in this disaccharide; the configuration of this glycosidic linkage is α for glucose and β for fructose. Sucrose can be cleaved into its component monosaccharides by the enzy me sucrase.

v Lactose, the disaccharide of milk, consists of galactose joined to glucose by a β-1, 4 glycosidic linkage. Lactose is hydrolyzed to these monosaccharides by lactase in human beings and by β -galactosidase in bacteria. v In maltose, two d-glucose residues are joined by a glycosidic linkage between the α -anomeric form of C-1 on one sugar and the hydroxyl oxygen atom on C-4 of the adjacent sugar. Such a linkage is called an α-1, 4 -glycosidicbond. Maltose comes from the hydrolysis of starch and is in turn hydrolyzed to glucose by maltase. v Sucrase, lactase, and maltase are located on the outer surfaces of epithelial cells lining the small intestine.
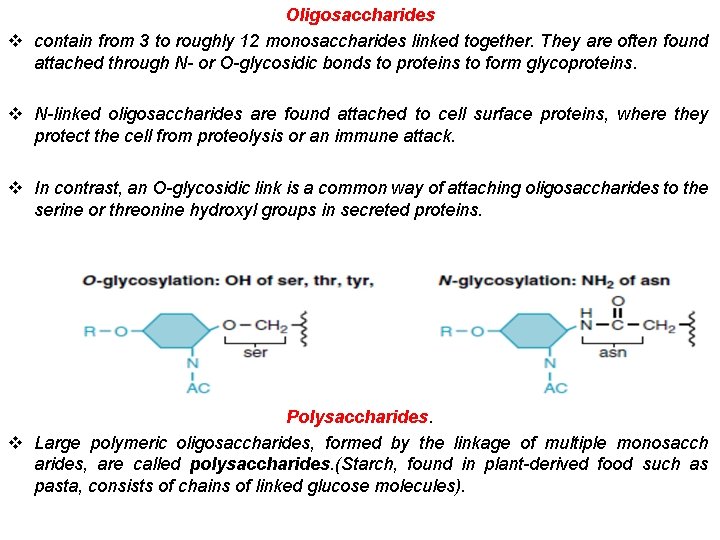
Oligosaccharides v contain from 3 to roughly 12 monosaccharides linked together. They are often found attached through N- or O-glycosidic bonds to proteins to form glycoproteins. v N-linked oligosaccharides are found attached to cell surface proteins, where they protect the cell from proteolysis or an immune attack. v In contrast, an O-glycosidic link is a common way of attaching oligosaccharides to the serine or threonine hydroxyl groups in secreted proteins. Polysaccharides. v Large polymeric oligosaccharides, formed by the linkage of multiple monosacch arides, are called polysaccharides. (Starch, found in plant-derived food such as pasta, consists of chains of linked glucose molecules).
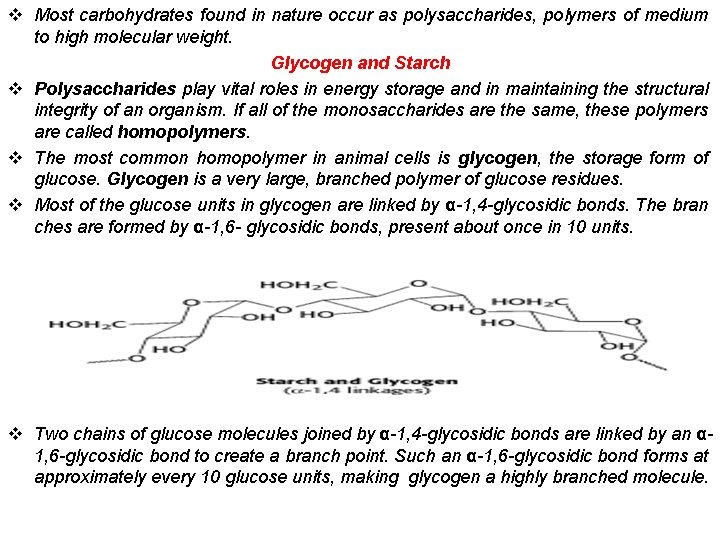
v Most carbohydrates found in nature occur as polysaccharides, polymers of medium to high molecular weight. Glycogen and Starch v Polysaccharides play vital roles in energy storage and in maintaining the structural integrity of an organism. If all of the monosaccharides are the same, these polymers are called homopolymers. v The most common homopolymer in animal cells is glycogen, the storage form of glucose. Glycogen is a very large, branched polymer of glucose residues. v Most of the glucose units in glycogen are linked by α-1, 4 -glycosidic bonds. The bran ches are formed by α-1, 6 - glycosidic bonds, present about once in 10 units. v Two chains of glucose molecules joined by α-1, 4 -glycosidic bonds are linked by an α 1, 6 -glycosidic bond to create a branch point. Such an α-1, 6 -glycosidic bond forms at approximately every 10 glucose units, making glycogen a highly branched molecule.
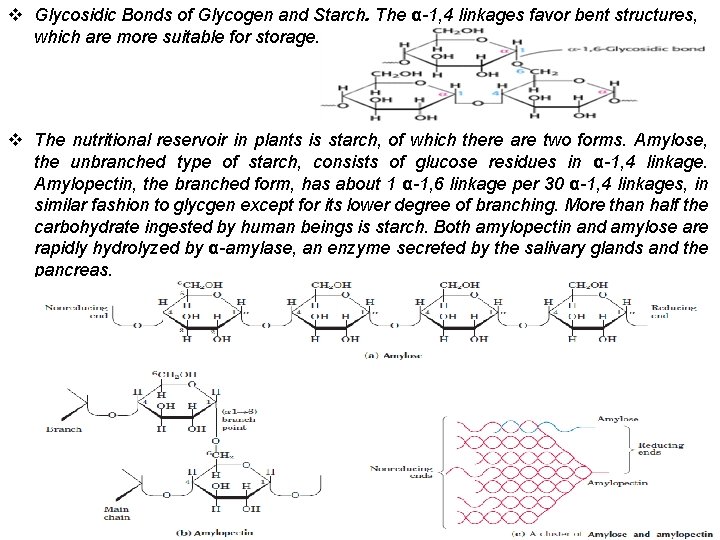
v Glycosidic Bonds of Glycogen and Starch. The α-1, 4 linkages favor bent structures, which are more suitable for storage. v The nutritional reservoir in plants is starch, of which there are two forms. Amylose, the unbranched type of starch, consists of glucose residues in α-1, 4 linkage. Amylopectin, the branched form, has about 1 α-1, 6 linkage per 30 α-1, 4 linkages, in similar fashion to glycgen except for its lower degree of branching. More than half the carbohydrate ingested by human beings is starch. Both amylopectin and amylose are rapidly hydrolyzed by α-amylase, an enzyme secreted by the salivary glands and the pancreas.
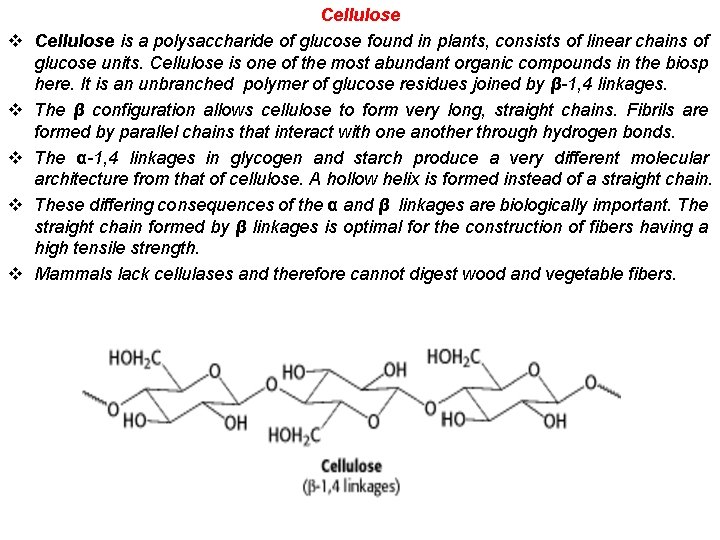
v v v Cellulose is a polysaccharide of glucose found in plants, consists of linear chains of glucose units. Cellulose is one of the most abundant organic compounds in the biosp here. It is an unbranched polymer of glucose residues joined by β-1, 4 linkages. The β configuration allows cellulose to form very long, straight chains. Fibrils are formed by parallel chains that interact with one another through hydrogen bonds. The α-1, 4 linkages in glycogen and starch produce a very different molecular architecture from that of cellulose. A hollow helix is formed instead of a straight chain. These differing consequences of the α and β linkages are biologically important. The straight chain formed by β linkages is optimal for the construction of fibers having a high tensile strength. Mammals lack cellulases and therefore cannot digest wood and vegetable fibers.
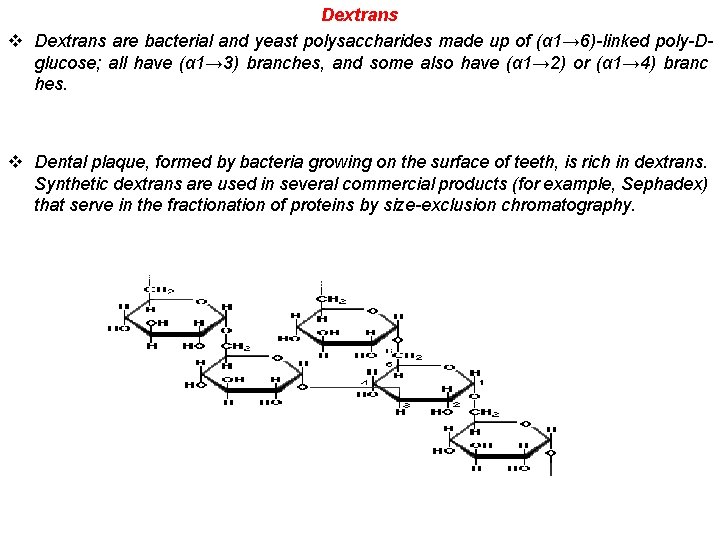
Dextrans v Dextrans are bacterial and yeast polysaccharides made up of (α 1→ 6)-linked poly-Dglucose; all have (α 1→ 3) branches, and some also have (α 1→ 2) or (α 1→ 4) branc hes. v Dental plaque, formed by bacteria growing on the surface of teeth, is rich in dextrans. Synthetic dextrans are used in several commercial products (for example, Sephadex) that serve in the fractionation of proteins by size-exclusion chromatography.
- Slides: 27