Biochemistry Lecture 10 Lipids Membranes Diversity of Lipids

Biochemistry Lecture 10 Lipids & Membranes

Diversity of Lipids • Stored forms of energy • derivatives of fatty acids • oxidation is highly exergonic • Structure • high heat capacity • mechanical protection (absorb shocks) • esterified fatty acids at cell membranes • water repellant • Signals and cofactors • fused carbon rings alter the location and function of fatty acids 3
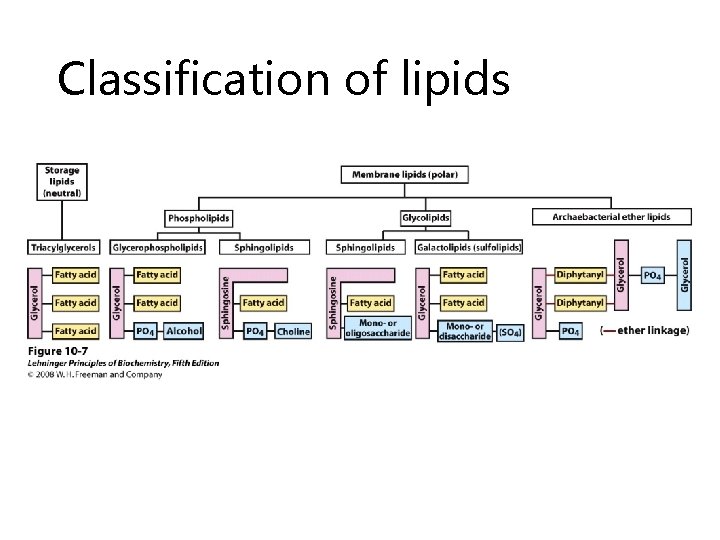
Classification of lipids

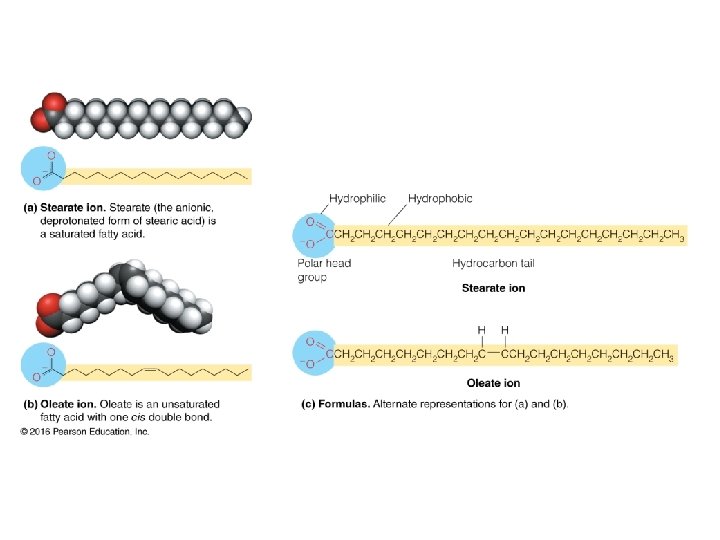
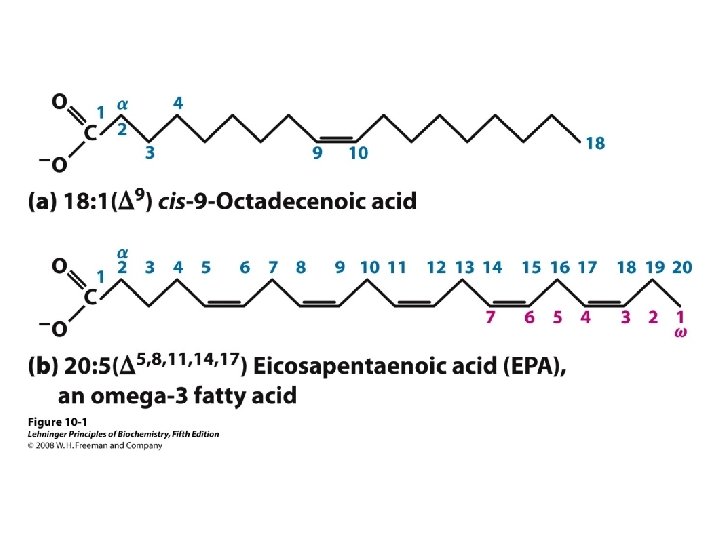
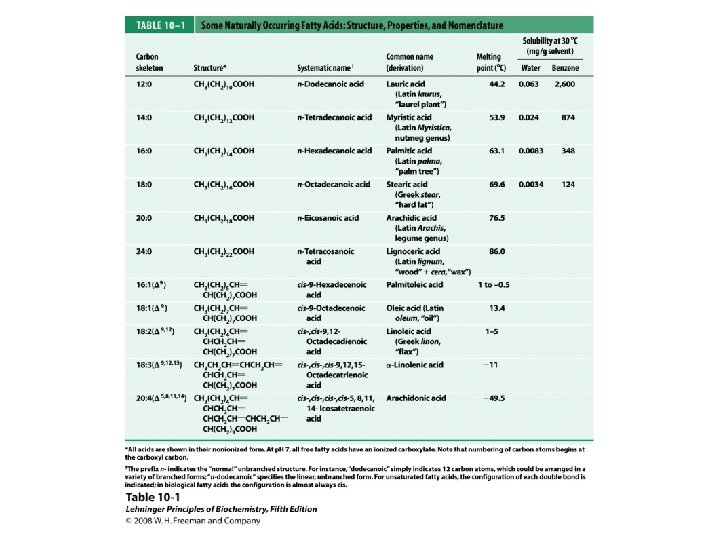
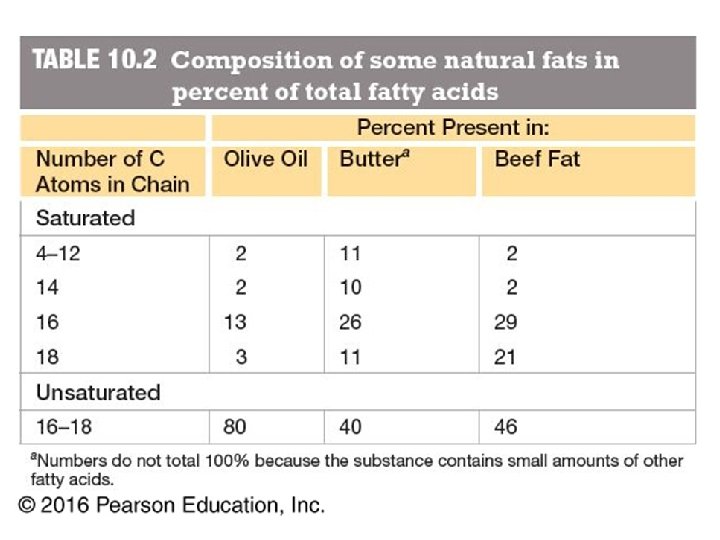
Fatty Acids • Carboxylic acids with hydrocarbon chains containing from 4 to 36 carbons • Almost all natural fatty acids have an even number of carbons • Most natural fatty acids are unbranched • Saturated: no double bonds between carbons in the chain • Monounsaturated: one double bond between carbons in the alkyl chain • Polyunsaturated: more than one double bond in the alkyl chain
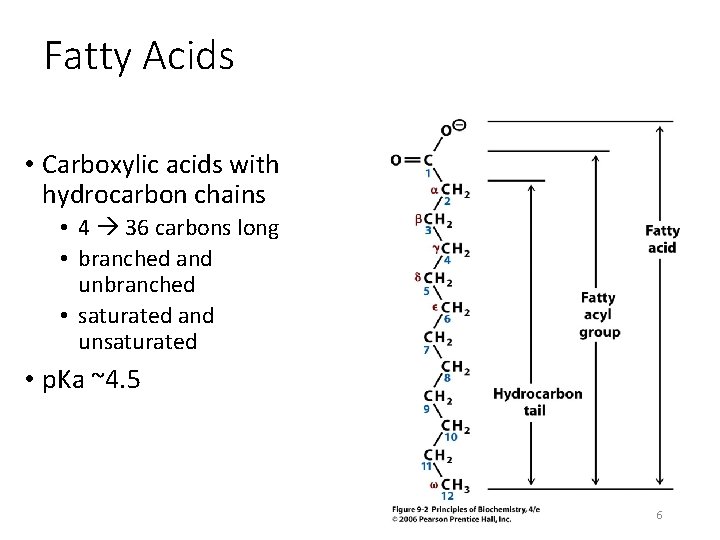
Fatty Acids • Carboxylic acids with hydrocarbon chains • 4 36 carbons long • branched and unbranched • saturated and unsaturated • p. Ka ~4. 5 6
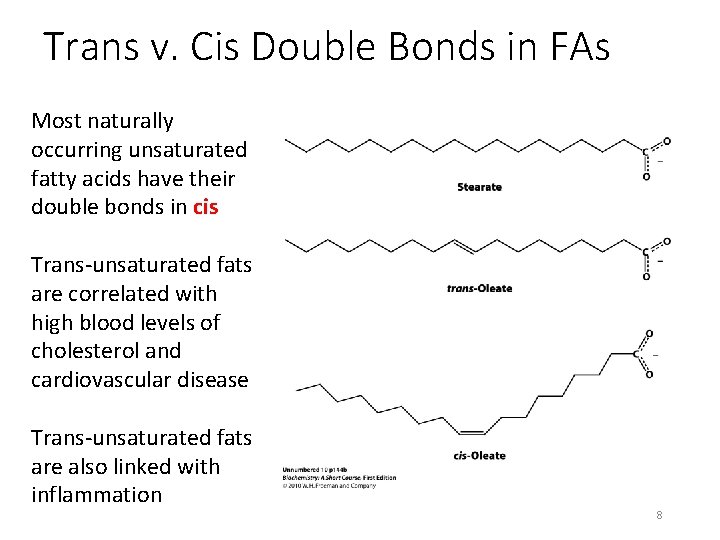
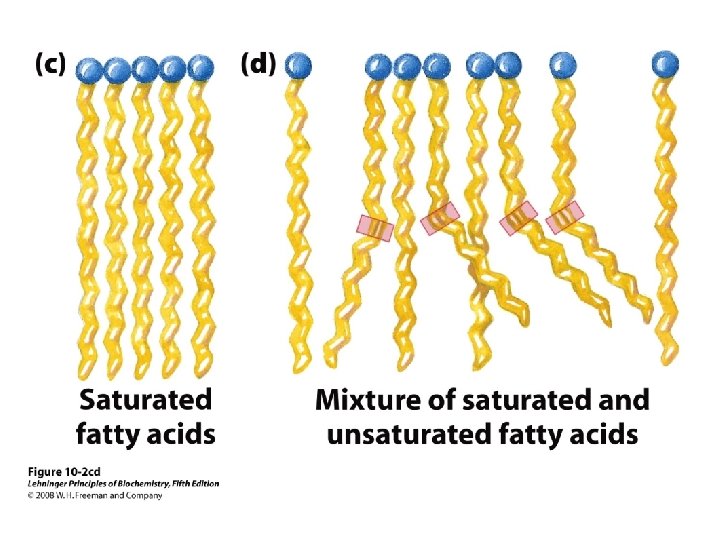
Trans v. Cis Double Bonds in FAs Most naturally occurring unsaturated fatty acids have their double bonds in cis Trans-unsaturated fats are correlated with high blood levels of cholesterol and cardiovascular disease Trans-unsaturated fats are also linked with inflammation 8
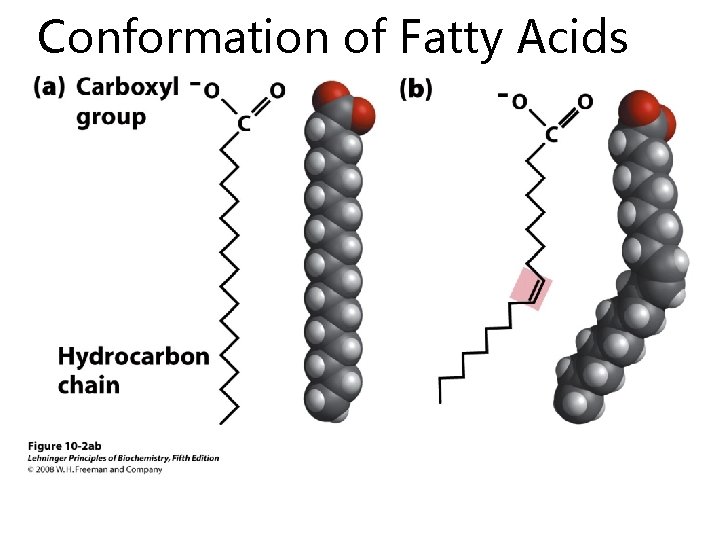
Conformation of Fatty Acids
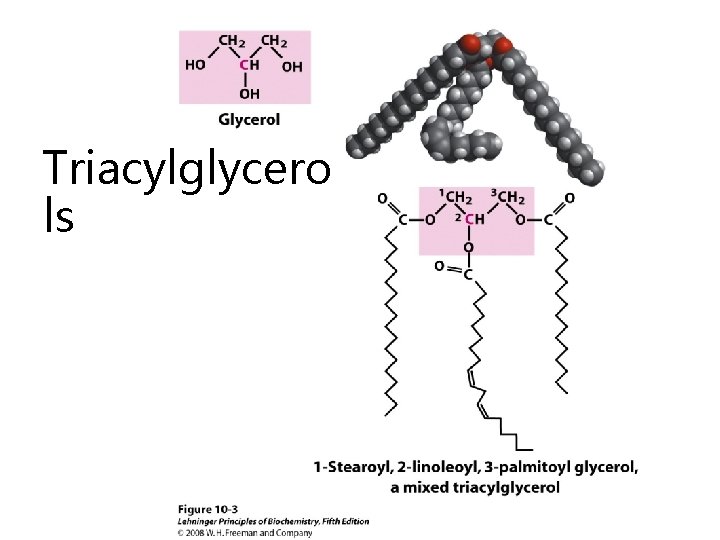
Triacylglycero ls
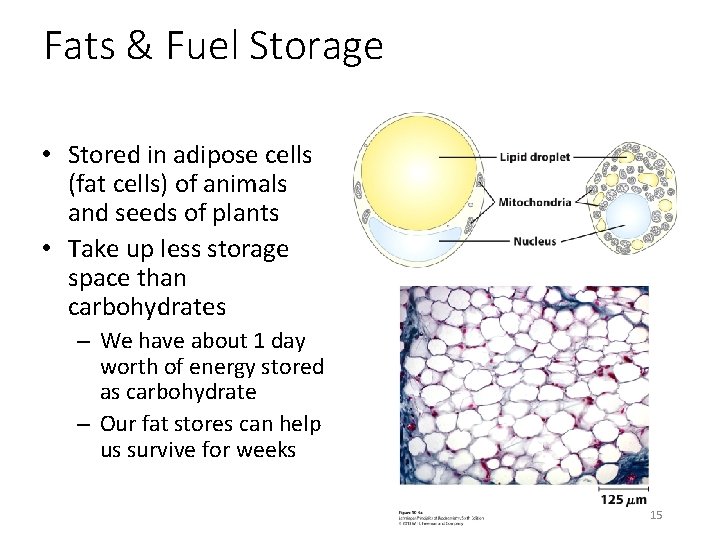
Fats & Fuel Storage • Stored in adipose cells (fat cells) of animals and seeds of plants • Take up less storage space than carbohydrates – We have about 1 day worth of energy stored as carbohydrate – Our fat stores can help us survive for weeks 15

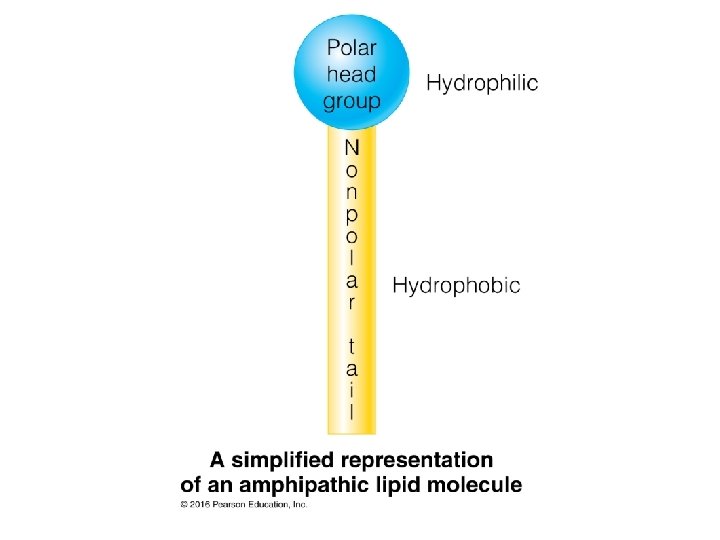
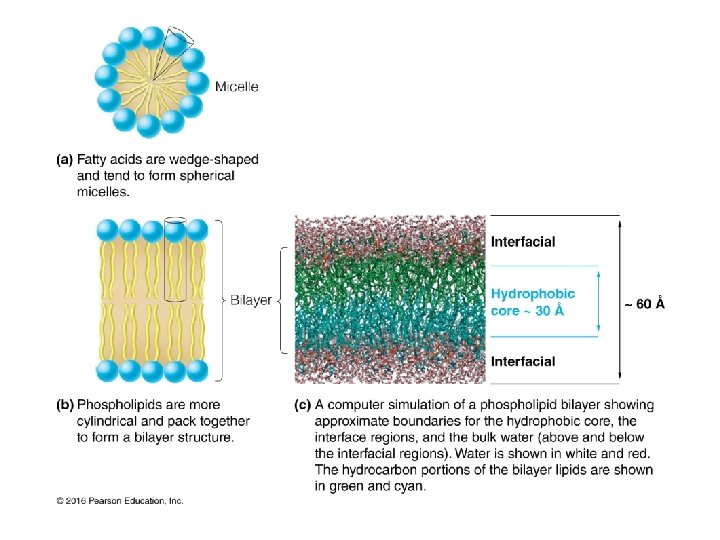
Membranes are Made from Lipids Major membrane lipids: • Phospholipids • Glycerophospholipids • Sphingolipids • Sphingosine • Ceramide • Glycolipids • Cholesterol Membrane lipids are all amphipathic: they have a nonpolar portion and also a polar portion 16
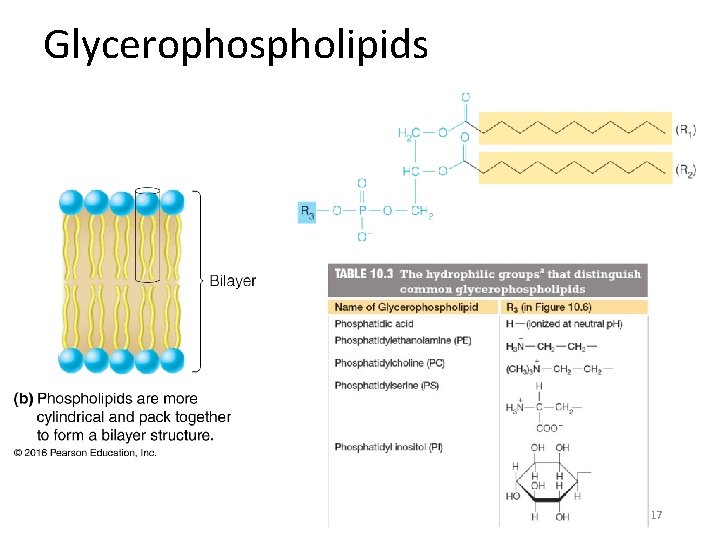
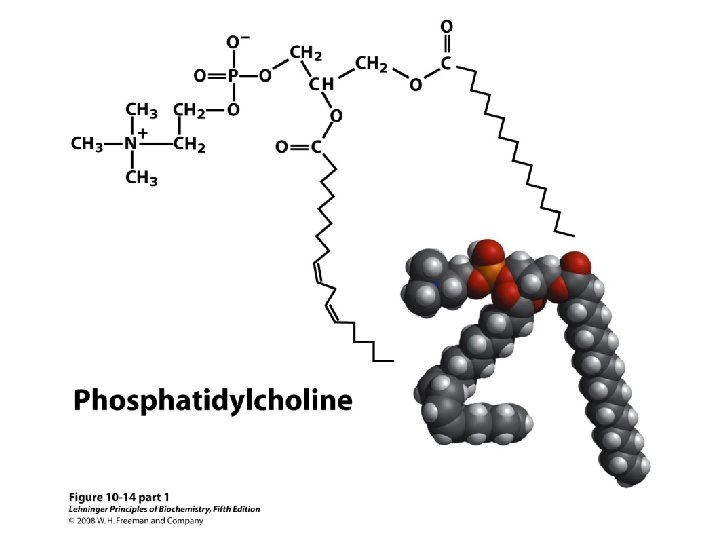
Glycerophospholipids 17
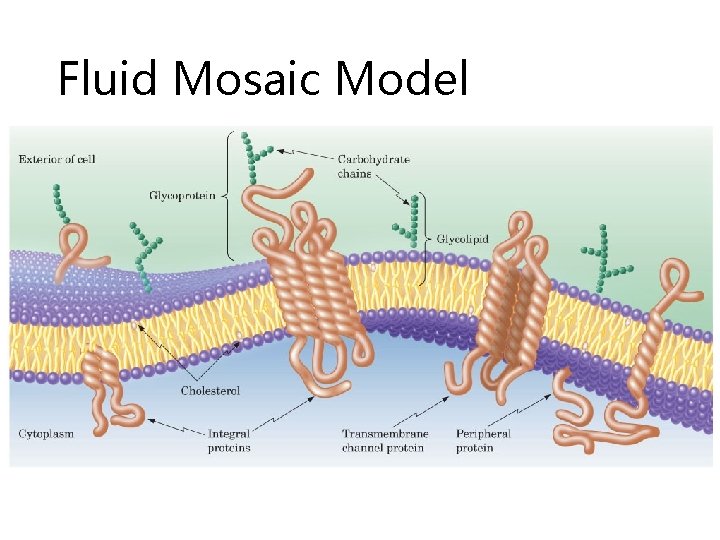
Fluid Mosaic Model Figure 21. 2 The fluid mosaic model of membranes.
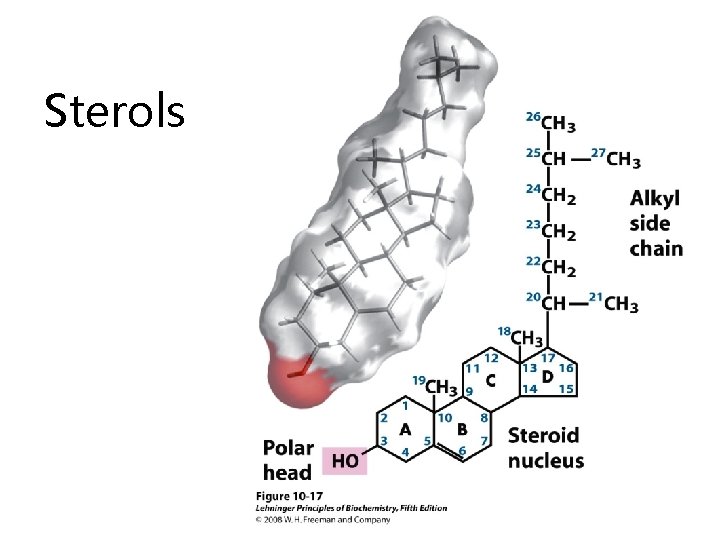
Sterols
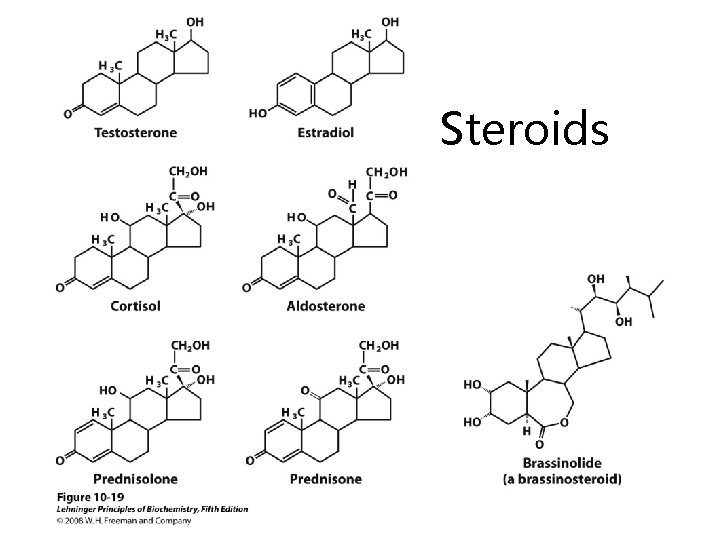
Steroids
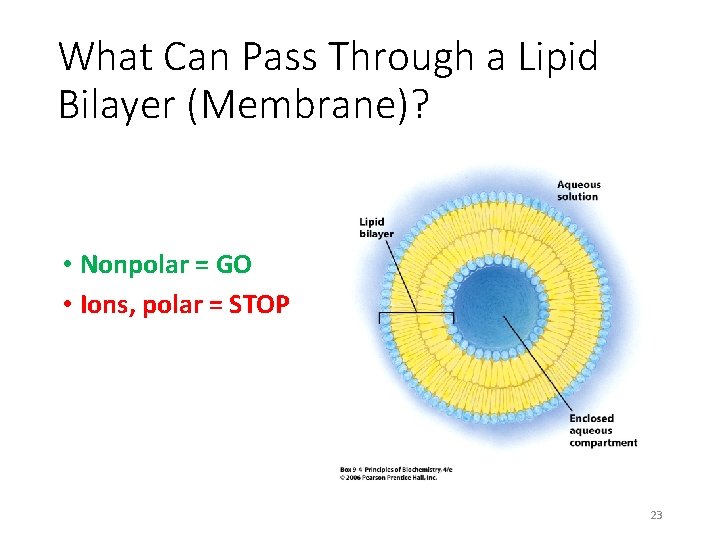
What Can Pass Through a Lipid Bilayer (Membrane)? • Nonpolar = GO • Ions, polar = STOP 23
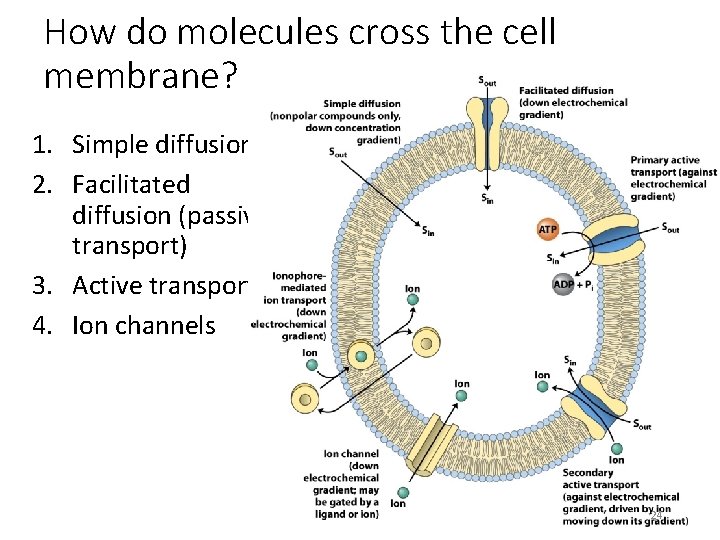
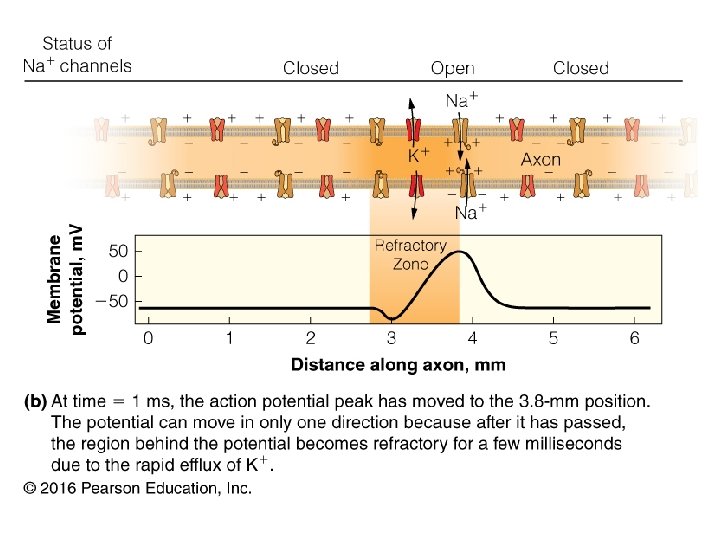
How do molecules cross the cell membrane? 1. Simple diffusion 2. Facilitated diffusion (passive transport) 3. Active transport 4. Ion channels 24
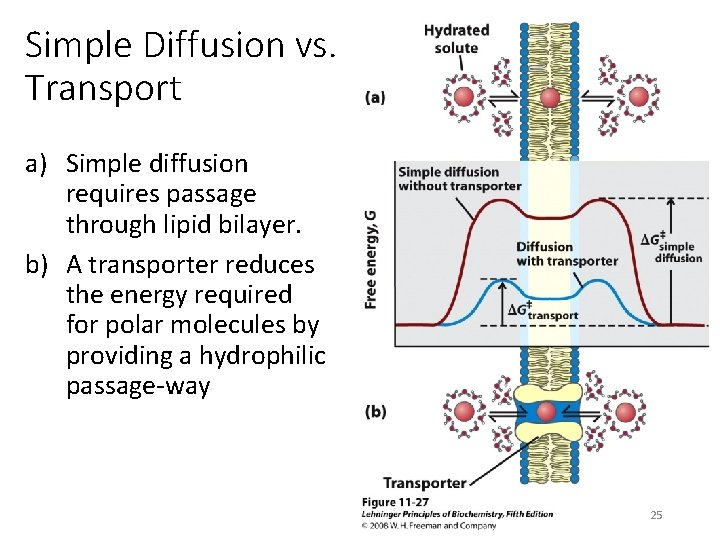
Simple Diffusion vs. Transport a) Simple diffusion requires passage through lipid bilayer. b) A transporter reduces the energy required for polar molecules by providing a hydrophilic passage-way 25
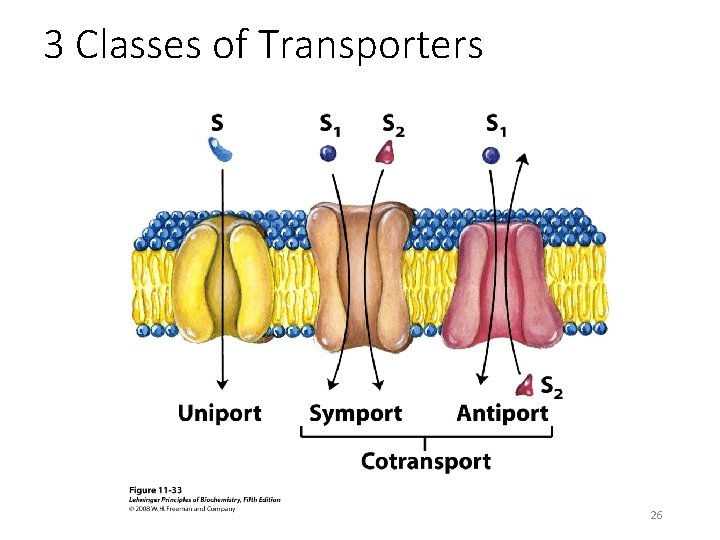
3 Classes of Transporters 26
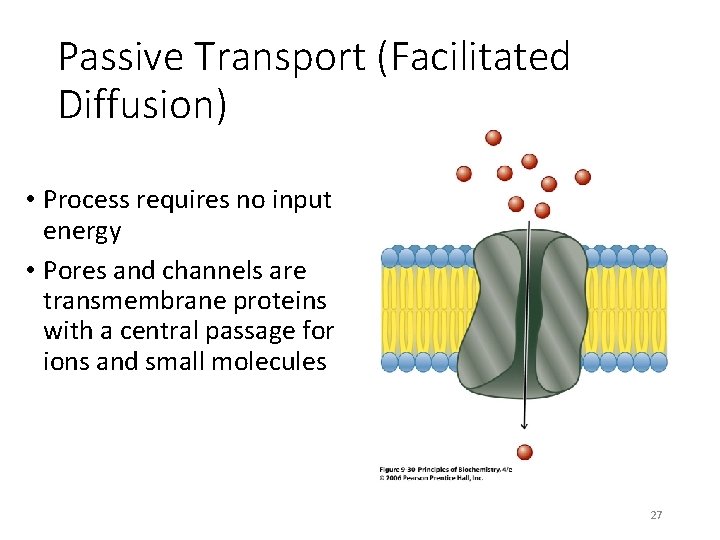
Passive Transport (Facilitated Diffusion) • Process requires no input energy • Pores and channels are transmembrane proteins with a central passage for ions and small molecules 27
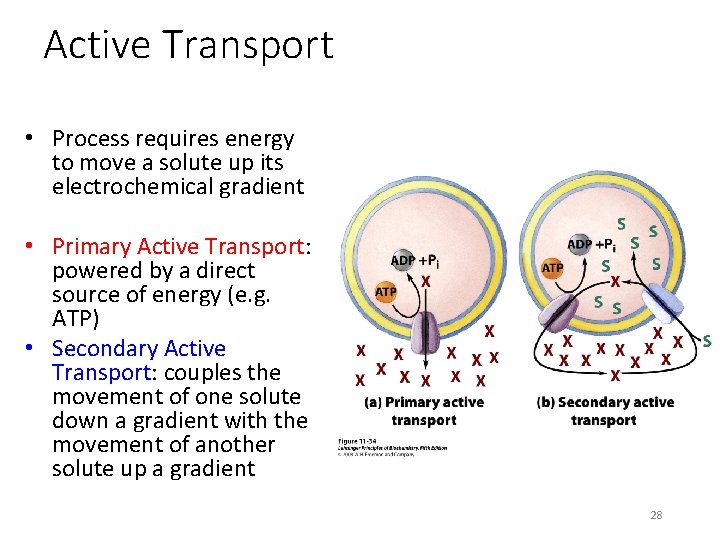
Active Transport • Process requires energy to move a solute up its electrochemical gradient • Primary Active Transport: powered by a direct source of energy (e. g. ATP) • Secondary Active Transport: couples the movement of one solute down a gradient with the movement of another solute up a gradient 28
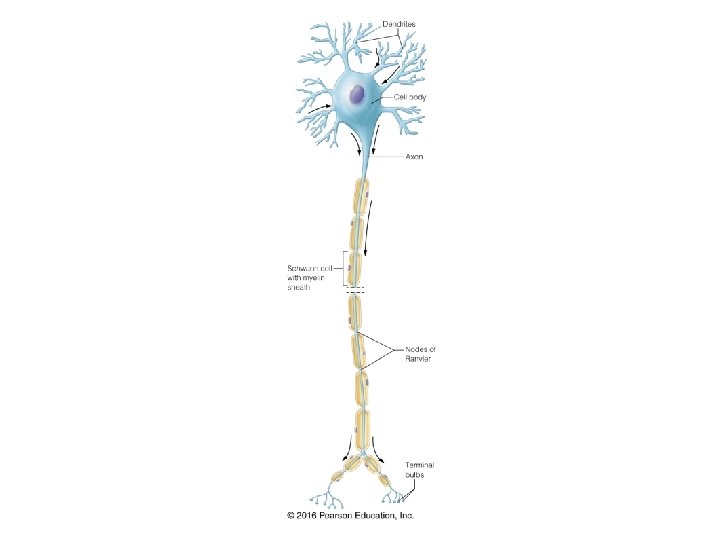
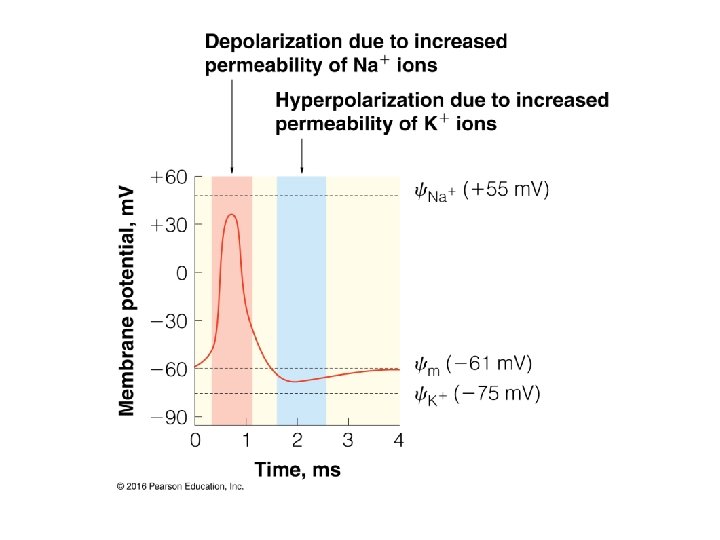
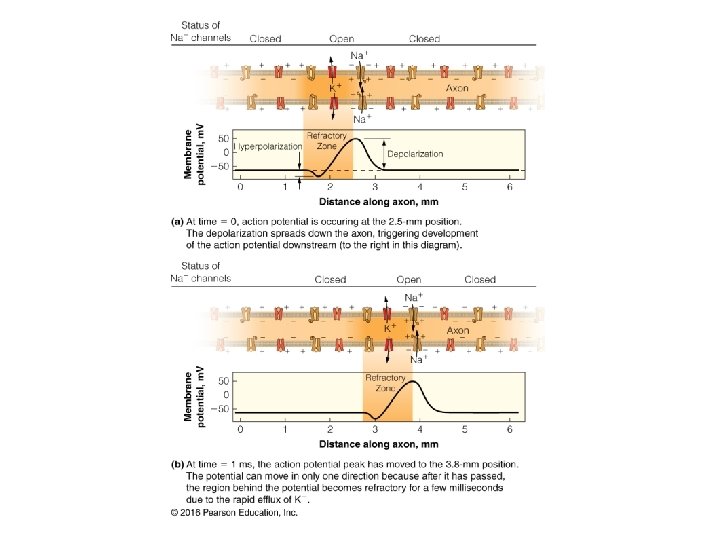
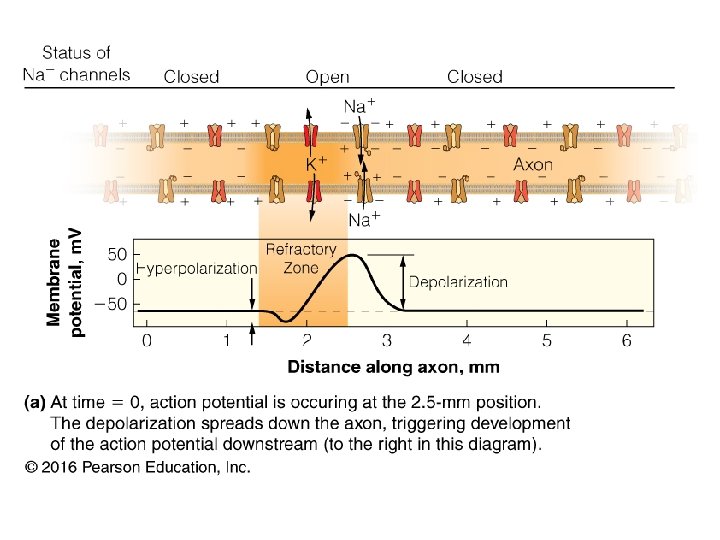
- Slides: 33