Biochemistry Learning Objectives l l l Know how

Biochemistry

Learning Objectives l l l Know how Monomers relate to Polymers (Pre-AP) Know Hydrolysis & Dehydration synthesis are opposite reactions & what they make Know what monomers make up the 4 major Polymers of biology Know functions/examples of – Carbohydrates – Lipids/Fats – Nucleic Acids – Proteins What Enzymes do and How they Work

Biochemistry: Essential Question: l Which food type is associated to which polymers: l l Polymers – Carbohydrates – Lipids/Fats – Nucleic Acids – Proteins • - Foods: STEAK BREAD BEANS PASTA CHOCOLATE LETTUCE
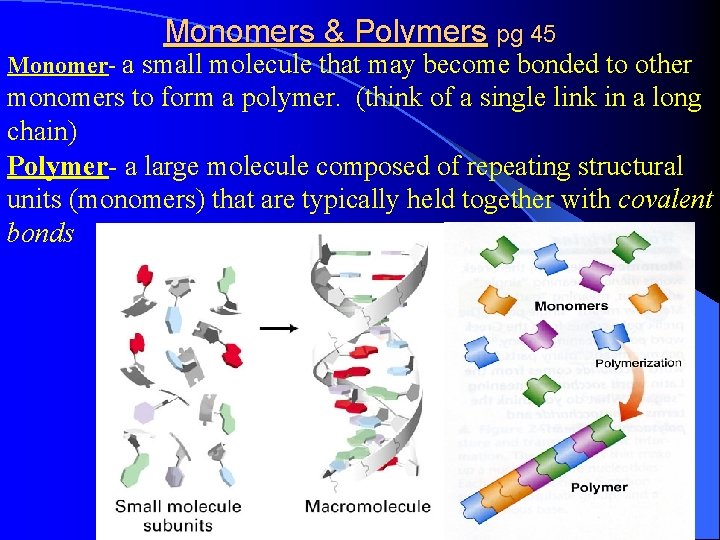
Monomers & Polymers pg 45 Monomer- a small molecule that may become bonded to other monomers to form a polymer. (think of a single link in a long chain) Polymer- a large molecule composed of repeating structural units (monomers) that are typically held together with covalent bonds
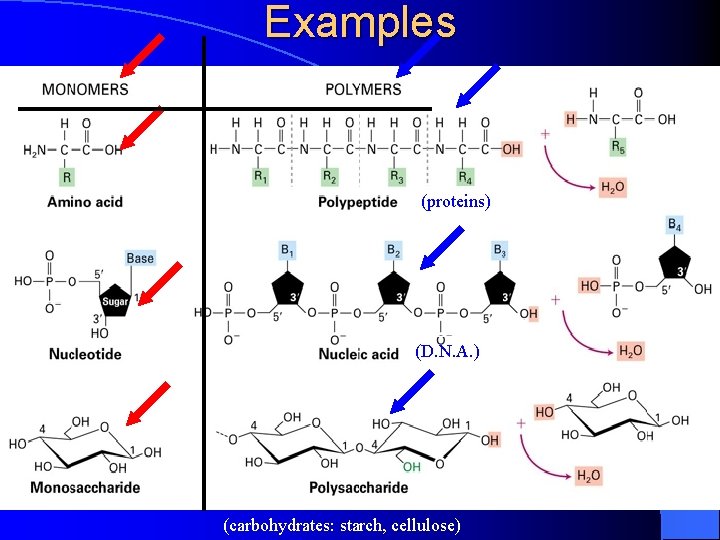
Examples (proteins) (D. N. A. ) (carbohydrates: starch, cellulose)

Polymerization l Polymers are formed by the process of polymerization l Macromolecules are very large polymers 2 ways to go: Creation or Destruction - Can make a polymer from bonding monomers to each other (like links in a chain) - Can break up a polymer into its thousands of monomer units (breaking up the links) THE MOST IMPORTATN INGREDIENT IN THE EITHER CASE IS H 20 Pg 45

Building and Breaking Polymers CONDENSATION Reaction: a chemical reaction where Monomers link to form polymers. l Water is formed during the formation of the polymers l l HYDROLYSIS : the BREAKDOWN of some complex polymers. Hydrolysis is the reversal of a condensation so water is used (consumed) in the reaction https: //biology 4 isc. weebly. com/b-biomolecules. html
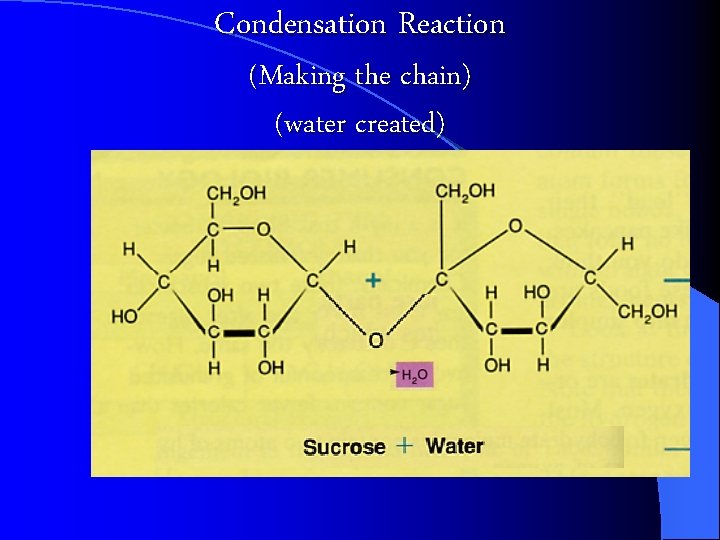
Condensation Reaction (Making the chain) (water created)
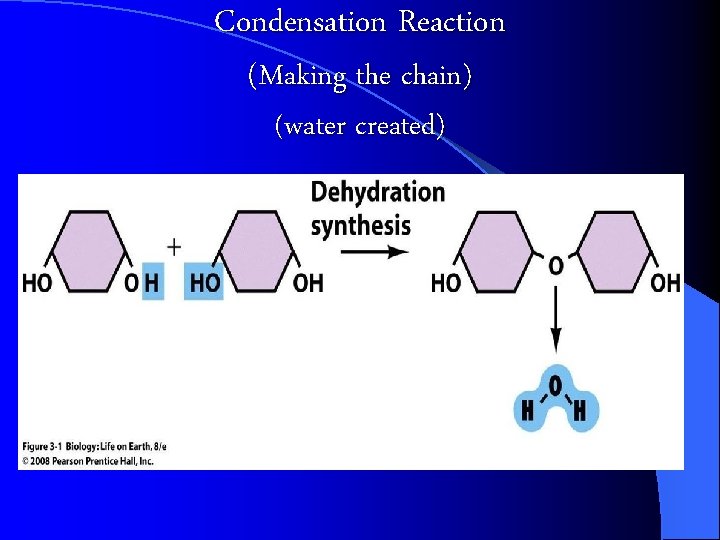
Condensation Reaction (Making the chain) (water created)
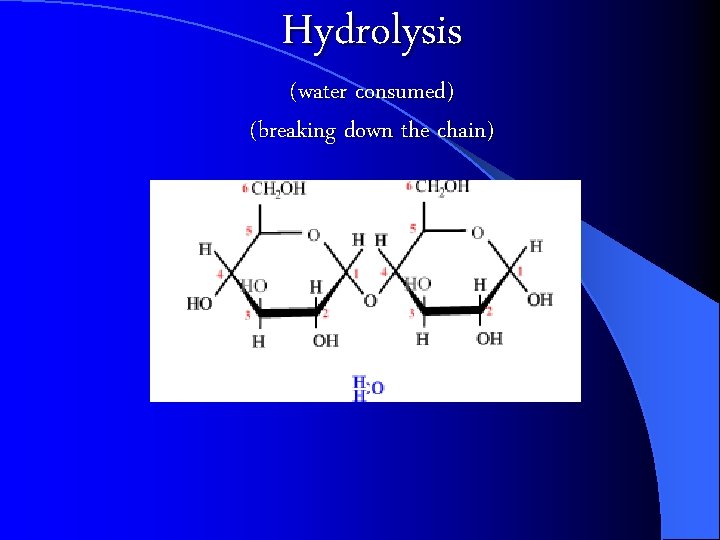
Hydrolysis (water consumed) (breaking down the chain)
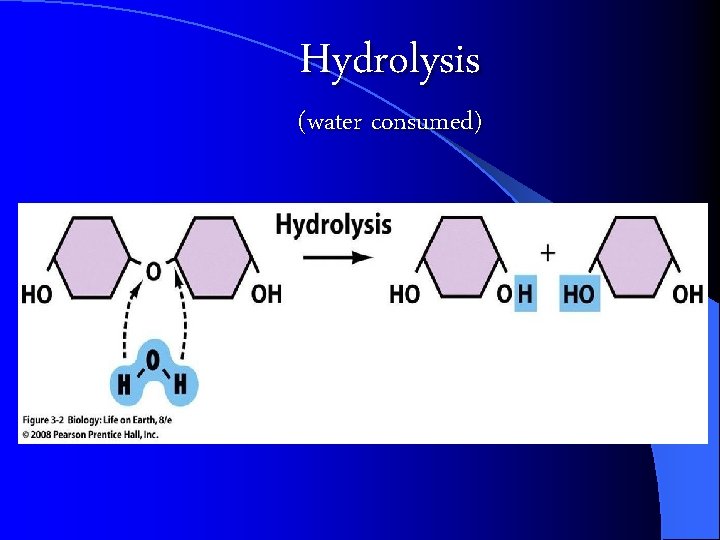
Hydrolysis (water consumed)

The 4 Major Polymers of Biology l 1. Carbohydrates (C-H-O 1: 2: 1 ratio) l 2. Lipids (C-H-O) l 3. Nucleic Acid Chains (C-H-O-N-P) l 4. Proteins (C-H-O-N)…sometimes S

Saccharide = Sugars MONOSACCHARIDES are simple sugars (1 ring) l Know Mono vs Di vs Poly l l GLUCOSE l GALACTOSE = sugar found in milk l FRUCTOSE = fruit sugar l Chemical composition (C 6 H 12 O 6)
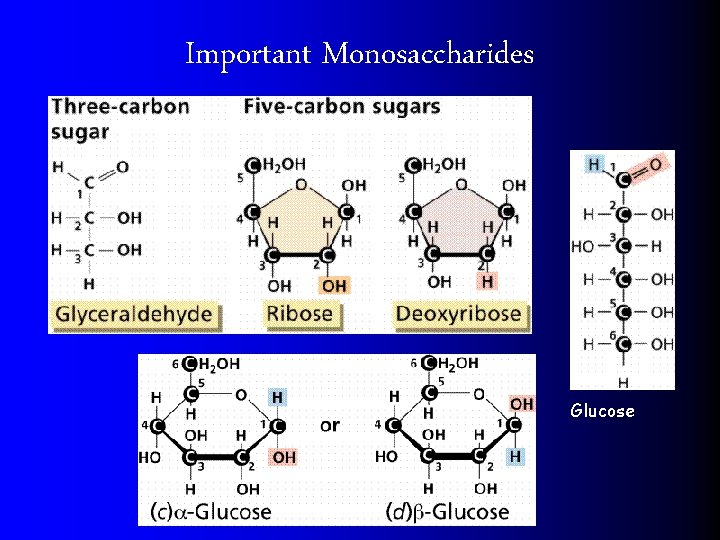
Important Monosaccharides Glucose
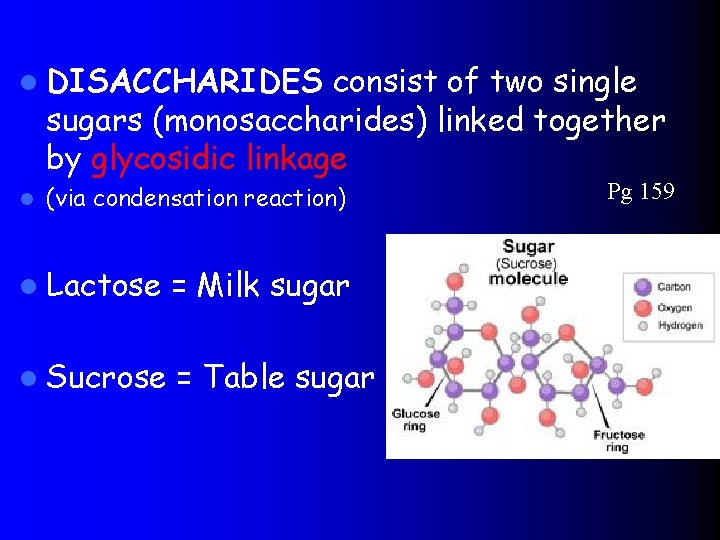
l DISACCHARIDES consist of two single sugars (monosaccharides) linked together by glycosidic linkage l (via condensation reaction) l Lactose = Milk sugar l Sucrose = Table sugar Pg 159
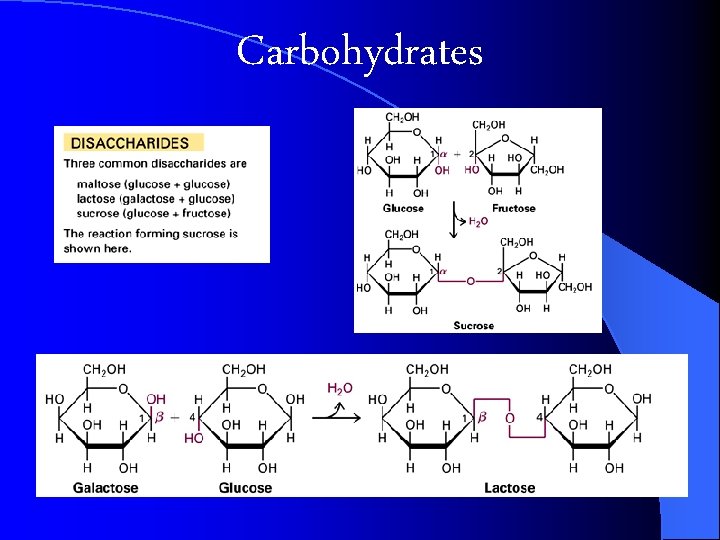
Carbohydrates
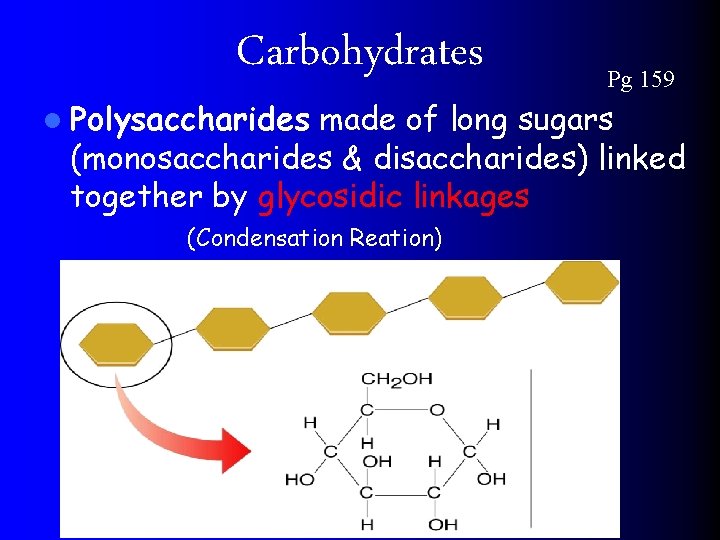
Carbohydrates l Polysaccharides Pg 159 made of long sugars (monosaccharides & disaccharides) linked together by glycosidic linkages (Condensation Reation)
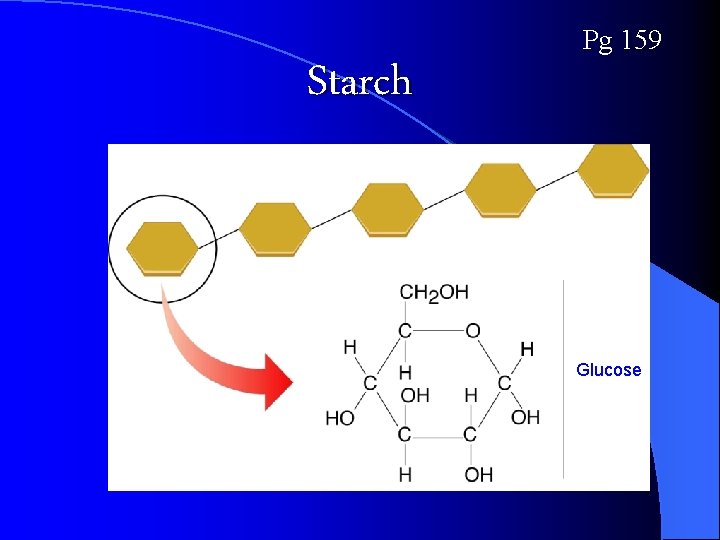
Starch Pg 159 Glucose

Carbohydrate Examples: Starch - Plants convert excess sugars into starches for long-term ENERGY storage (no FAT in plants) l Glycogen -Animals store glucose in the form of glycogen in the liver and muscles to be used as quick energy storage l Cellulose -a structural polysaccharide contained in the cell walls of plants (why plants can stand upright) l Chitin – a polysaccharide found in the cell walls of fungi and the exoskeletons of insects and arthropods l

Lipids (a. k. a. fats) Pg 160 l Lipids are large, NONPOLAR organic molecules that do NOT dissolve in water l Monomers are: l Oils, fats, waxes, and steroids are lipid based l Lipid molecules use less OXYGEN than carbohydrates to store energy efficiently (great for insulation too) l Used a lot in the cell/plasma membrane 3 Fatty acids & 1 Glycerol
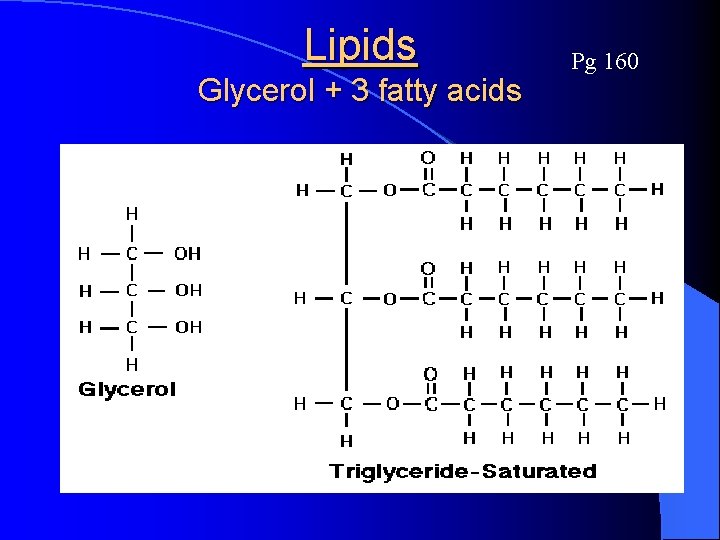
Lipids Glycerol + 3 fatty acids Pg 160

2) Lipids Pg 160 l UNSATURATED FATS are a liquid at room temperature (OILS). l Has a Double bond on the carbon chain l SATURATED FATS are solid at room temperature l NO double bonds on the carbon chain
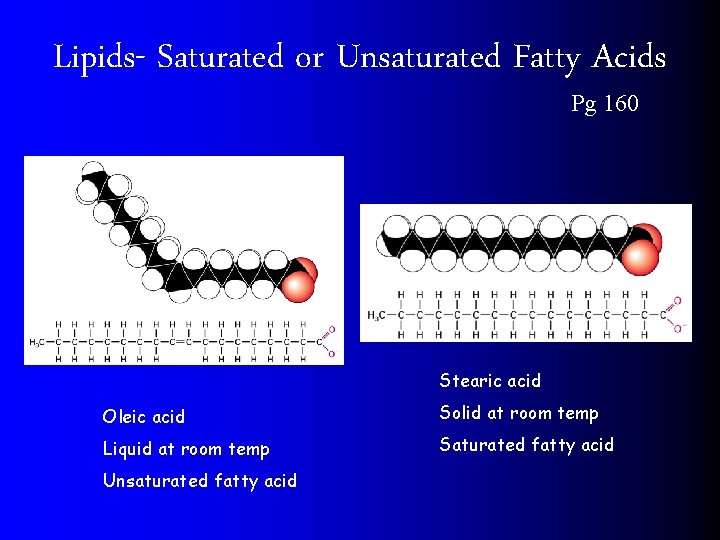
Lipids- Saturated or Unsaturated Fatty Acids Pg 160 Stearic acid Oleic acid Solid at room temp Liquid at room temp Saturated fatty acid Unsaturated fatty acid
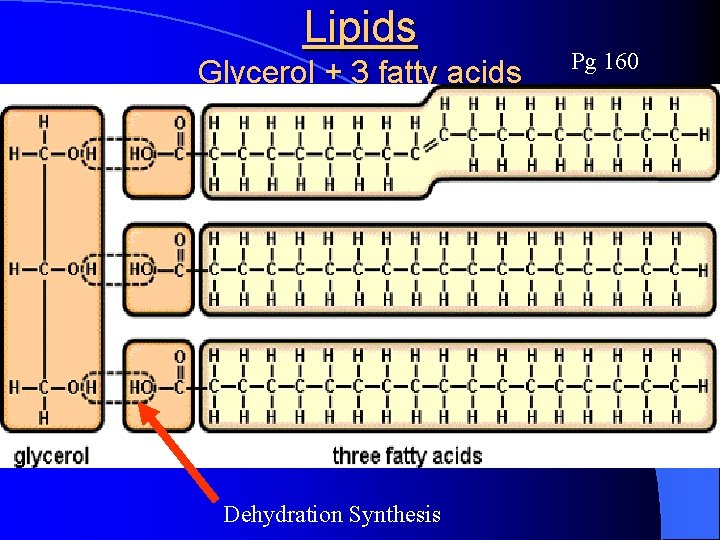
Lipids Glycerol + 3 fatty acids Dehydration Synthesis Pg 160
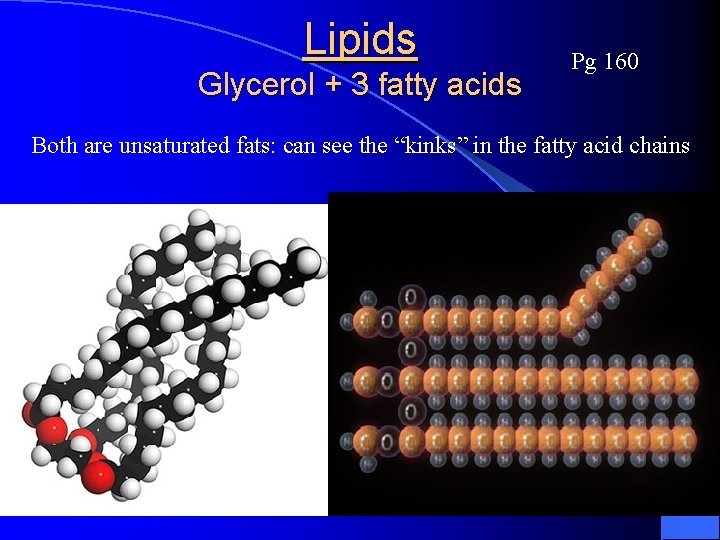
Lipids Glycerol + 3 fatty acids Pg 160 Both are unsaturated fats: can see the “kinks” in the fatty acid chains
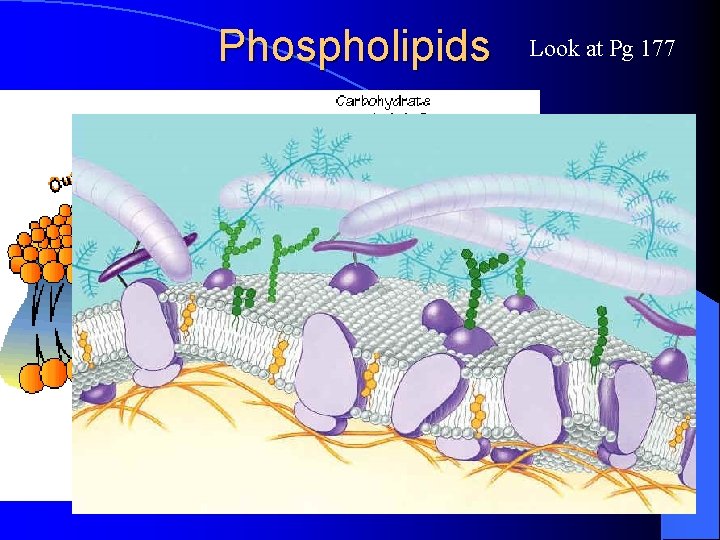
Phospholipids Look at Pg 177
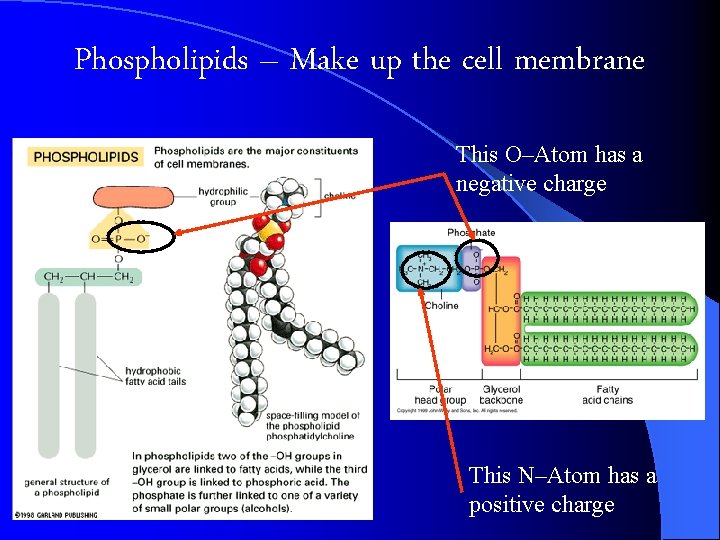
Phospholipids – Make up the cell membrane This O–Atom has a negative charge This N–Atom has a positive charge
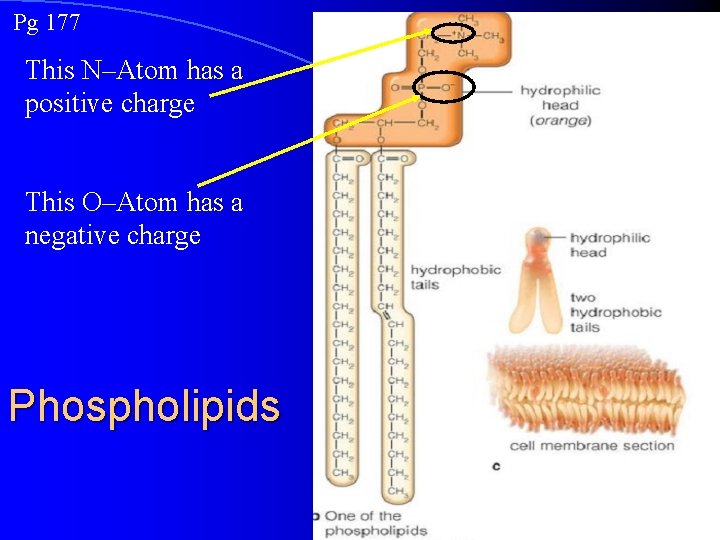
Pg 177 This N–Atom has a positive charge This O–Atom has a negative charge Phospholipids
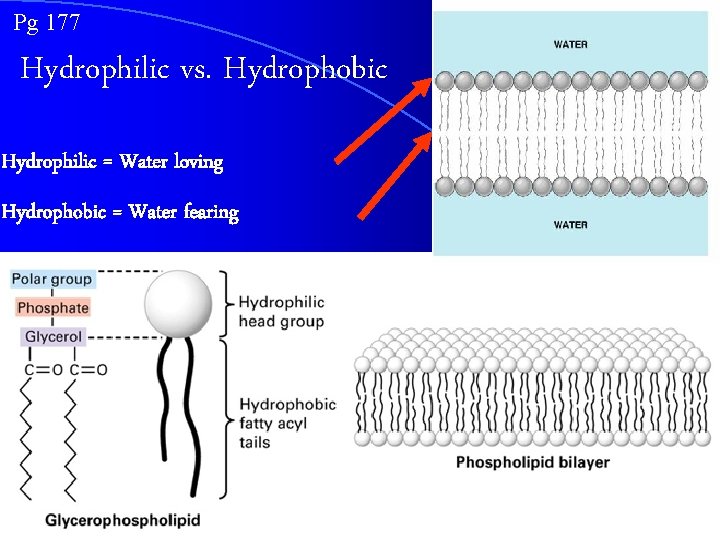
Pg 177 Hydrophilic vs. Hydrophobic Hydrophilic = Water loving Hydrophobic = Water fearing
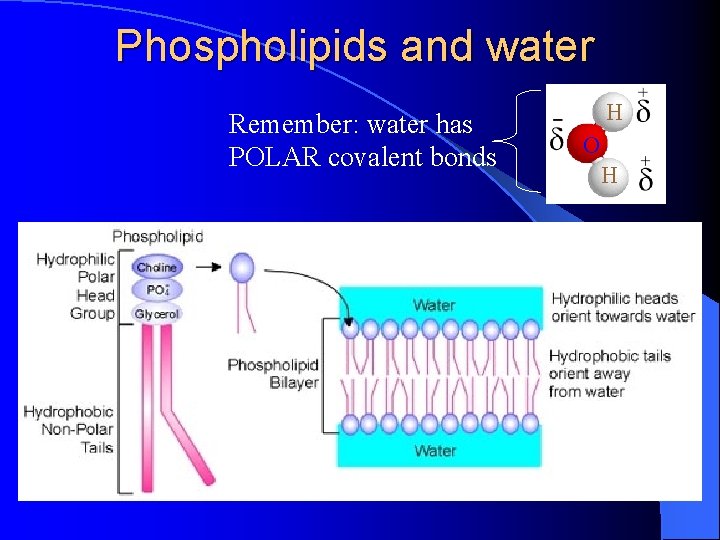
Phospholipids and water Remember: water has POLAR covalent bonds H O H

Functions of Fat 1. As a “Bank” of Energy for harsh times or when food is naturally scarce or when traveling very far (migration) or to fuel rapid growth Ex:

Functions of Fat 2. As Insulation against the cold or wet to keep body temperature stable Blubber (a special lipid) is common in deep aquatic mammals (helps them float too)
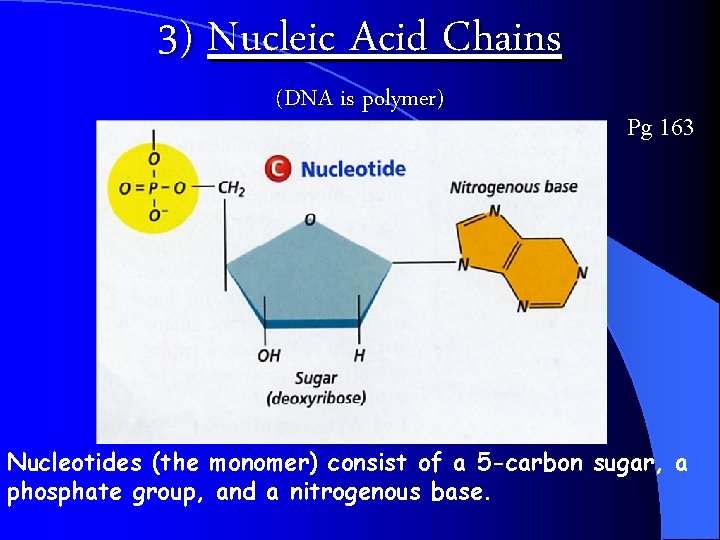
3) Nucleic Acid Chains (DNA is polymer) Pg 163 Nucleotides (the monomer) consist of a 5 -carbon sugar, a phosphate group, and a nitrogenous base.
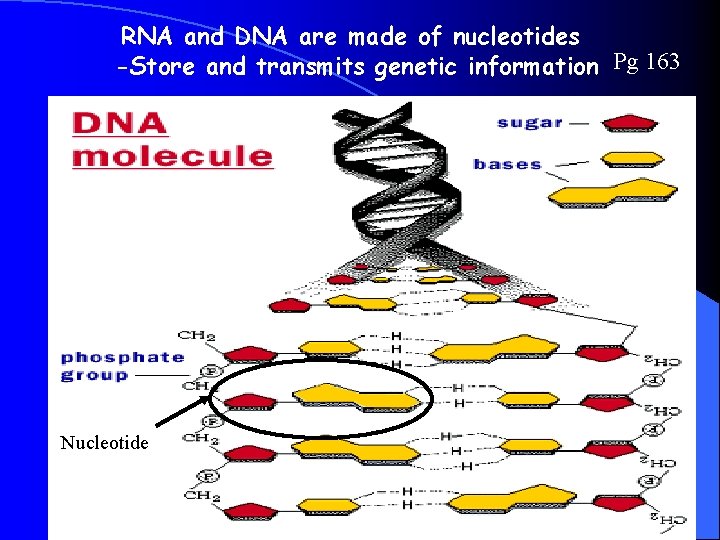
RNA and DNA are made of nucleotides -Store and transmits genetic information Pg 163 Nucleotide
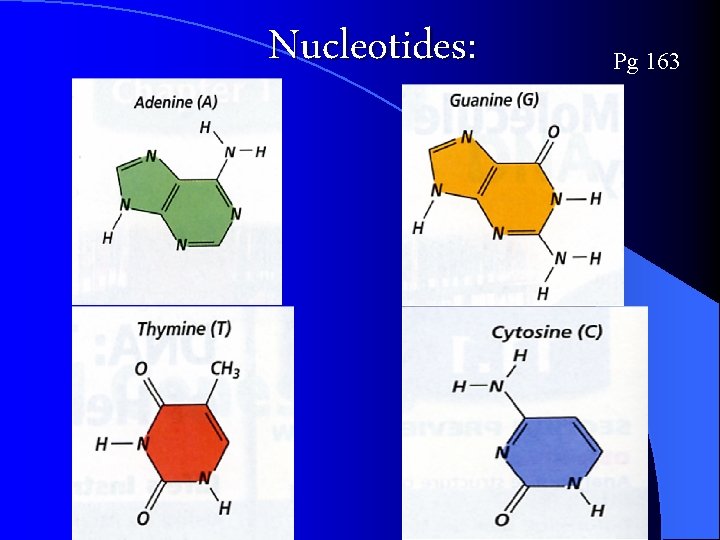
Nucleotides: Pg 163

4) ***Proteins*** Pg 161 (monomer are amino acids) l l Proteins are long polymers made up of smaller monomers called AMINO ACIDS Amino Acids differ ONLY in the type of R group they carry Amino acids composed of 3 parts 1. Amino Group 2. Carboxylic group 3. R-group (Makes 20 different amino acids)
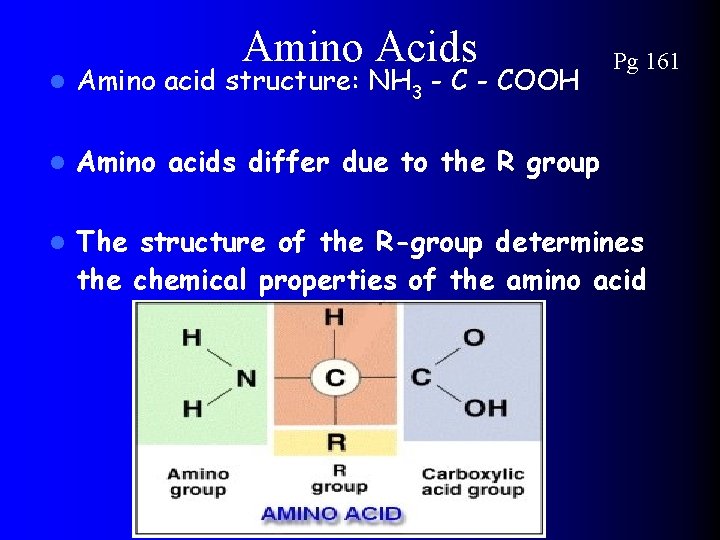
Amino Acids Pg 161 l Amino acid structure: NH 3 - COOH l Amino acids differ due to the R group l The structure of the R-group determines the chemical properties of the amino acid
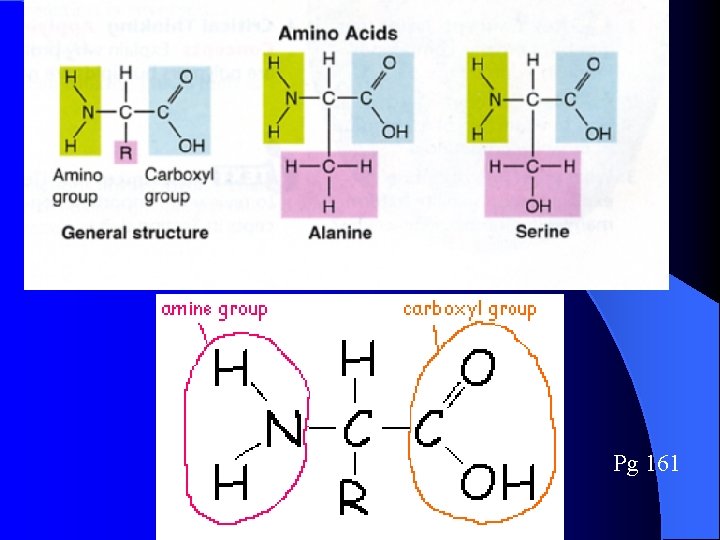
Pg 161

20 Amino Acids
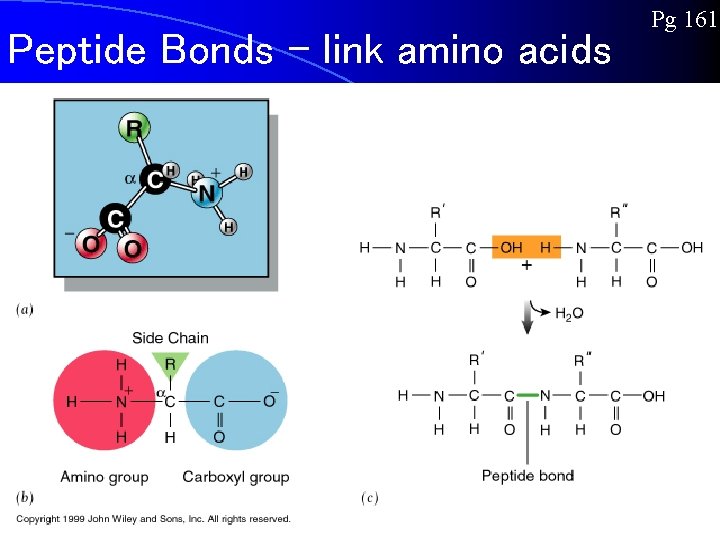
Peptide Bonds – link amino acids Pg 161

Proteins l **Each protein has a specific order of amino acids that allow it form a complex shape** l **Different shapes allow proteins to perform different functions** l Amino Acids can bond to each other, one at a time, forming a long chain called a POLYPEPTIDE. l Proteins are composed of one or more polypeptides.
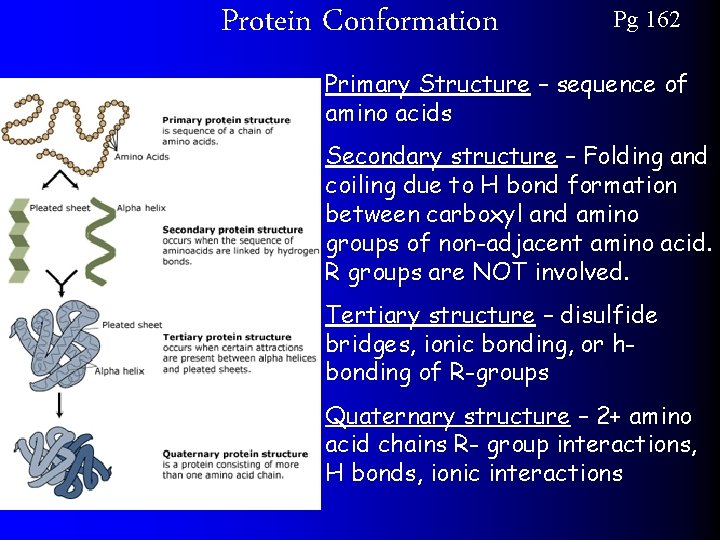
Protein Conformation Pg 162 Primary Structure – sequence of amino acids Secondary structure – Folding and coiling due to H bond formation between carboxyl and amino groups of non-adjacent amino acid. R groups are NOT involved. Tertiary structure – disulfide bridges, ionic bonding, or hbonding of R-groups Quaternary structure – 2+ amino acid chains R- group interactions, H bonds, ionic interactions
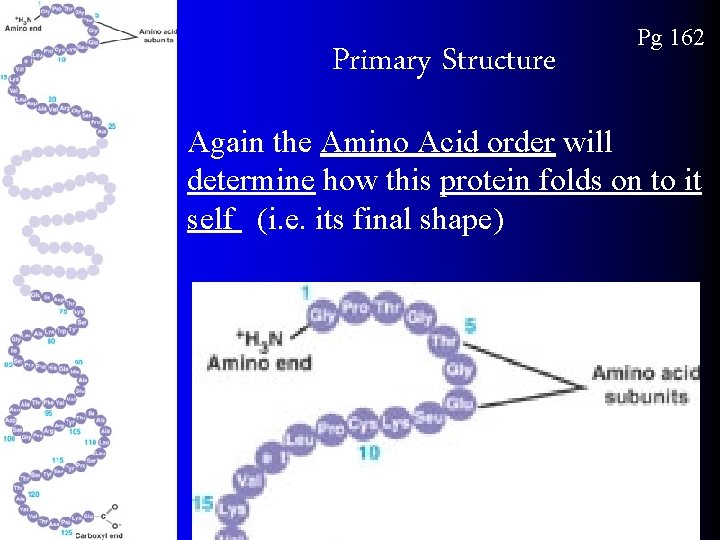
Primary Structure Pg 162 Again the Amino Acid order will determine how this protein folds on to it self (i. e. its final shape)
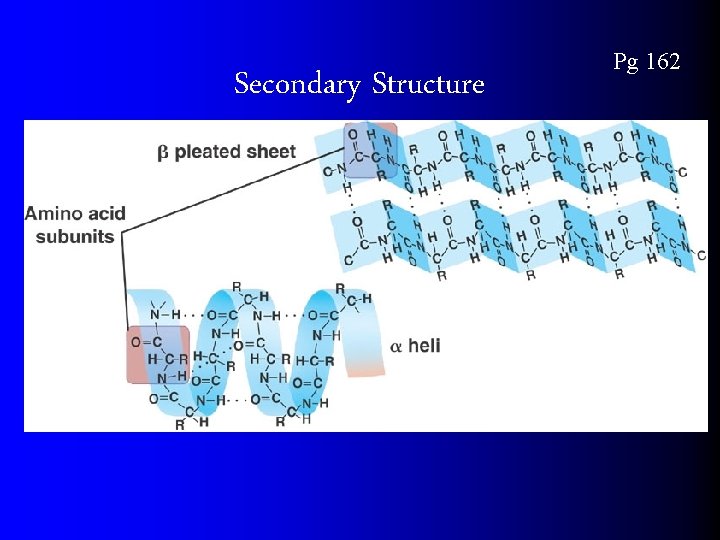
Secondary Structure Pg 162
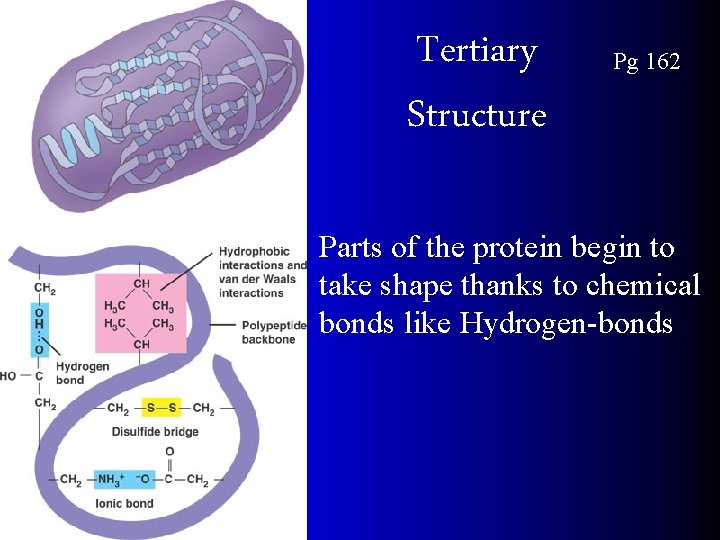
Tertiary Structure Pg 162 Parts of the protein begin to take shape thanks to chemical bonds like Hydrogen-bonds
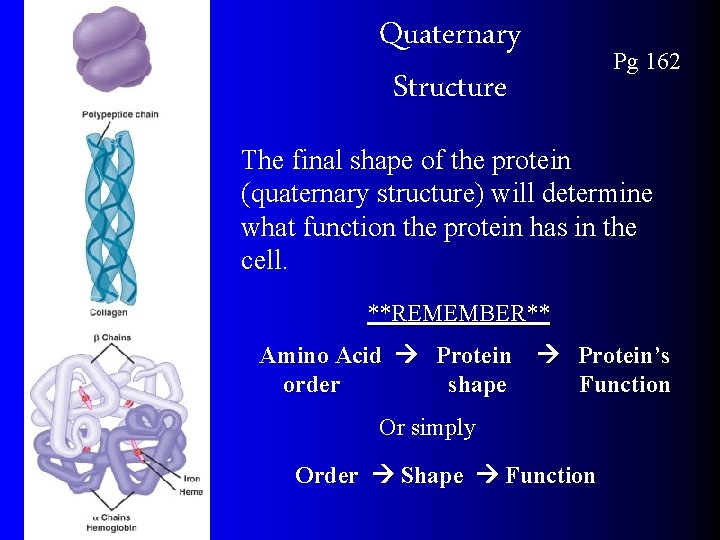
Quaternary Structure Pg 162 The final shape of the protein (quaternary structure) will determine what function the protein has in the cell. **REMEMBER** Amino Acid Protein’s order shape Function Or simply Order Shape Function
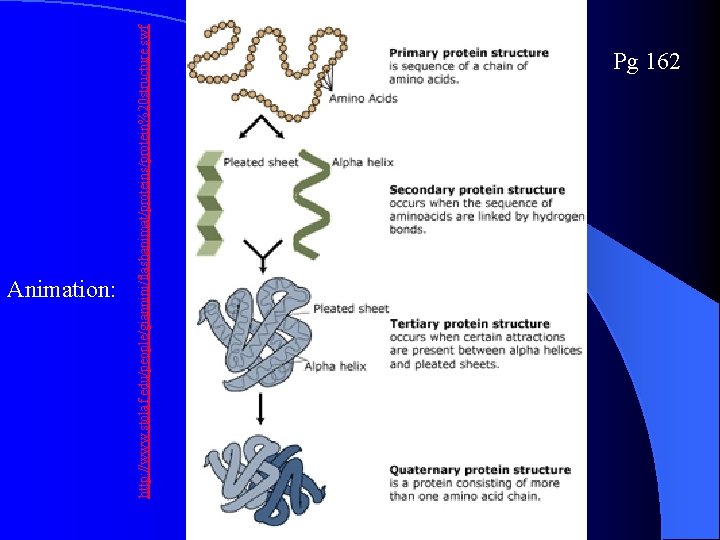
http: //www. stolaf. edu/people/giannini/flashanimat/proteins/protein%20 structure. swf Animation: Pg 162
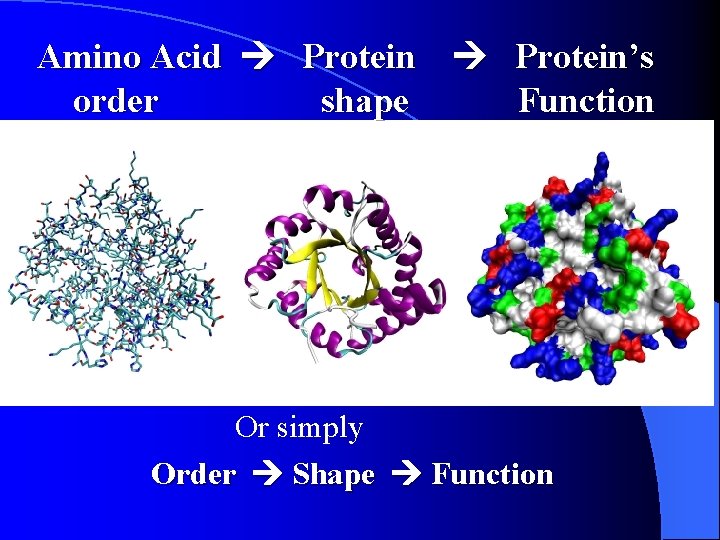
Amino Acid Protein’s order shape Function Or simply Order Shape Function
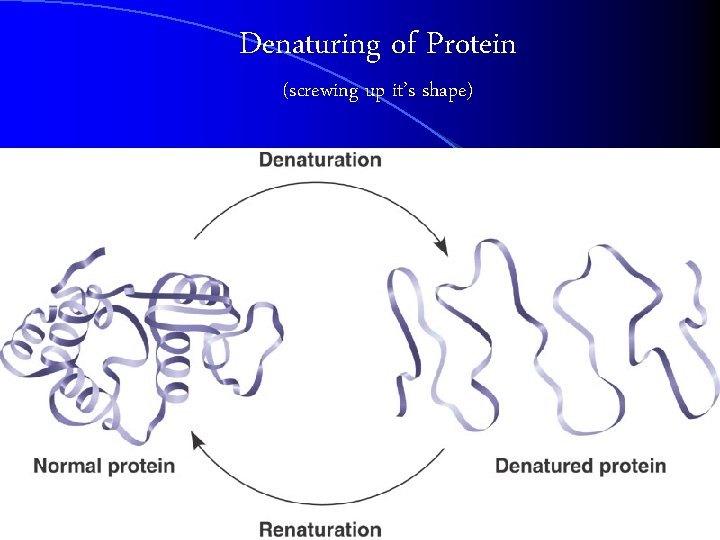
Denaturing of Protein (screwing up it’s shape)

Denaturing of Protein l Proteins may lose their unqiue 3 D shape (and not work) if there is… l Excessive heat l Changes in p. H (acidic or basic) l Changes in pressure in a cell
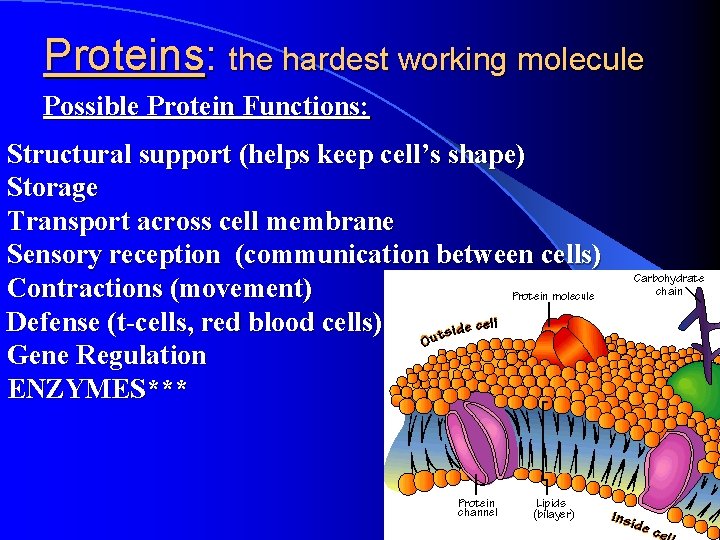
Proteins: the hardest working molecule Possible Protein Functions: Structural support (helps keep cell’s shape) Storage Transport across cell membrane Sensory reception (communication between cells) Contractions (movement) Defense (t-cells, red blood cells) Gene Regulation ENZYMES***

Enzymes Pg 162 **Act as CATALYSTS that can speed up some reactions by more than a billion times!** Enzymes work by a physical fit (Lock and Key) between the enzyme molecule and its SUBSTRATE, the reactant being catalyzed. Enzymes reduces the activation energy for the chemical reaction to occur. After the reaction, the enzyme is released and is unchanged, so it can be used many times Enzyme names end in –”ase”
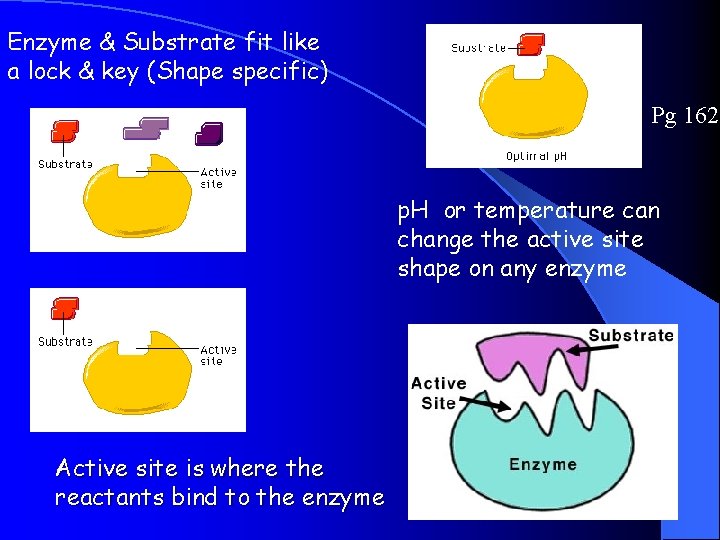
Enzyme & Substrate fit like a lock & key (Shape specific) Pg 162 p. H or temperature can change the active site shape on any enzyme Active site is where the reactants bind to the enzyme
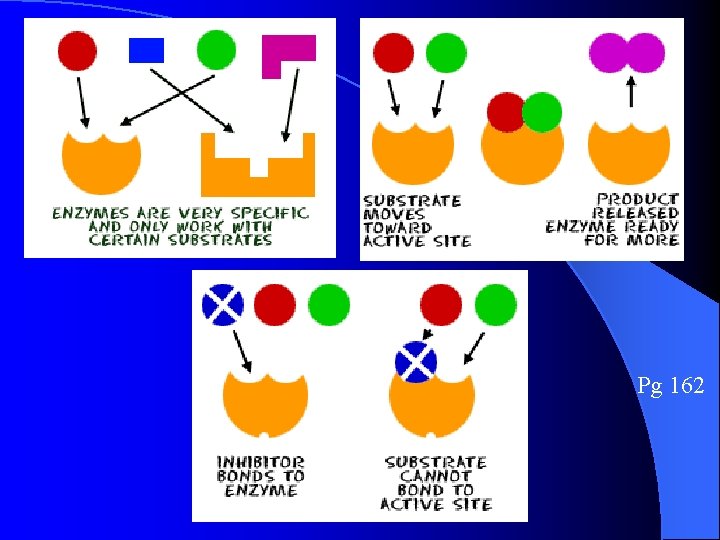
Pg 162
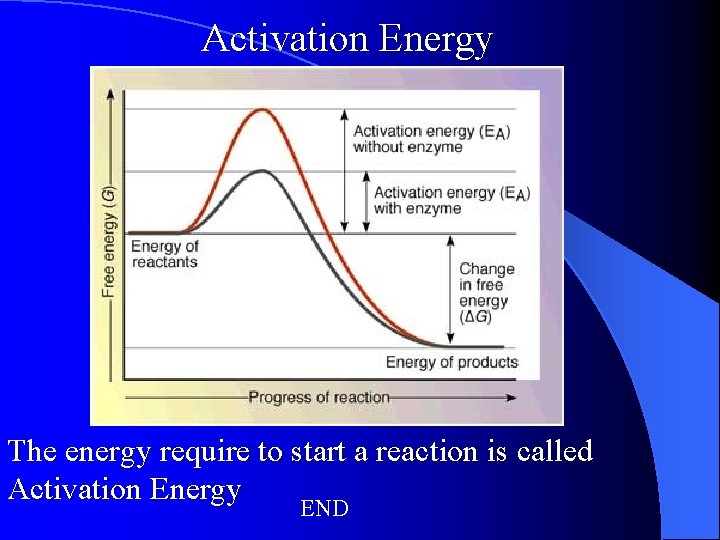
Activation Energy The energy require to start a reaction is called Activation Energy END

- Slides: 56