Biochemistry Heather Mc Dermott Taylor Dunn Chloe Entenberg
Biochemistry Heather Mc. Dermott, Taylor Dunn & Chloe Entenberg
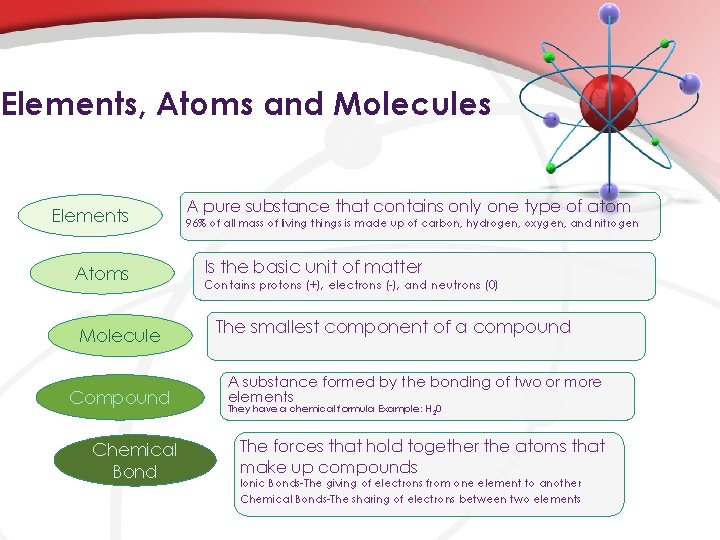
Elements, Atoms and Molecules Elements Atoms Molecule Compound Chemical Bond A pure substance that contains only one type of atom 96% of all mass of living things is made up of carbon, hydrogen, oxygen, and nitrogen Is the basic unit of matter Contains protons (+), electrons (-), and neutrons (0) The smallest component of a compound A substance formed by the bonding of two or more elements They have a chemical formula Example: H 20 The forces that hold together the atoms that make up compounds Ionic Bonds-The giving of electrons from one element to another Chemical Bonds-The sharing of electrons between two elements

• Properties of Water • Polar covalent compound • • Polar = the electrons are unevenly shared between the hydrogen and oxygen atoms Oxygen pulls on the electrons greater than hydrogen • The shared electrons are more likely closer to the oxygen • Partial positive pole and partial negative pole • Strong cohesion and adhesion • • Cohesion = attraction between molecules of the same substance • Adhesion = attraction between molecules of different substances Hydrogen bonding = weak bonds that can be broken easily, responsible for special properties • Waters liquid form is more dense than solid form • When lakes freeze they freeze from the top down
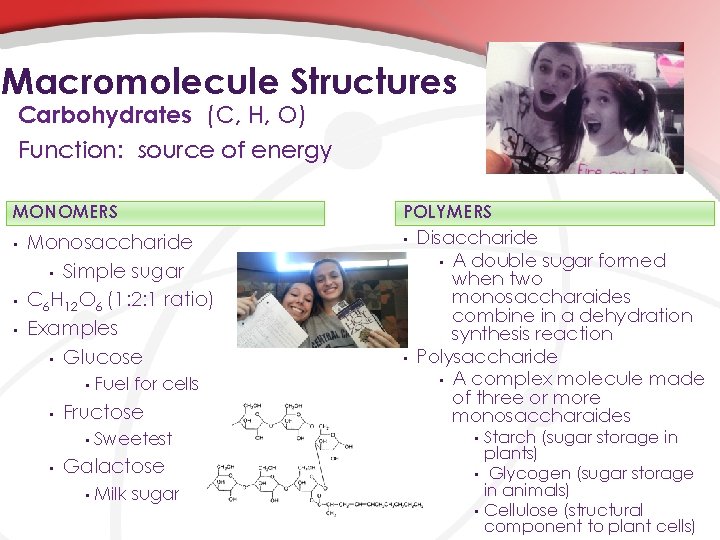
Macromolecule Structures Carbohydrates (C, H, O) Function: source of energy MONOMERS • • • Monosaccharide • Simple sugar C 6 H 12 O 6 (1: 2: 1 ratio) Examples • Glucose • • Fructose • • Fuel for cells Sweetest Galactose • Milk sugar POLYMERS • • Disaccharide • A double sugar formed when two monosaccharaides combine in a dehydration synthesis reaction Polysaccharide • A complex molecule made of three or more monosaccharaides Starch (sugar storage in plants) • Glycogen (sugar storage in animals) • Cellulose (structural component to plant cells) •

Macromolecule Structures Proteins (C, H, O, N) Function: Structural component to living things, helps build muscle mass, controls cell processes and reactions, enzymes MONOMERS • Amino Acid • Contains amino and carboxyl groups (NH 2 and COOH) • R = radical, different for each type of amino acid POLYMERS Protein • Amino acids joined by dehydration synthesis to form dipeptides and polypeptides • Process 1) Form a amino acid chain 2) Twist amino acid chain 3) Fold amino acid chain 4) Bring 2 -4 amino acid chains together •
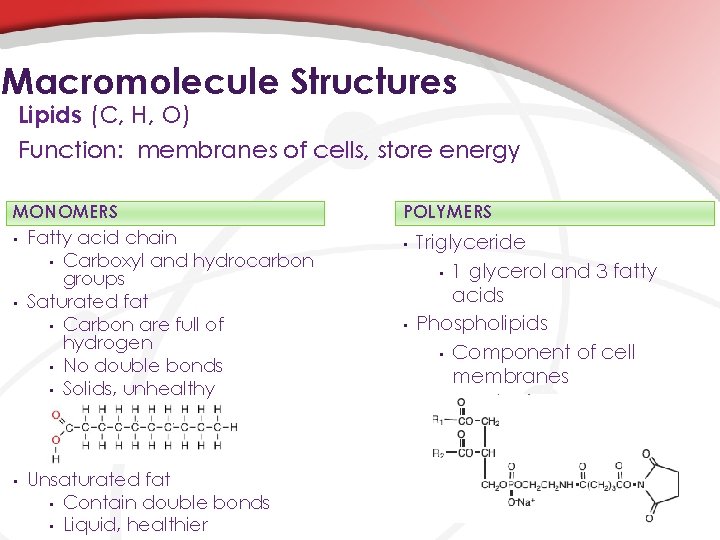
Macromolecule Structures Lipids (C, H, O) Function: membranes of cells, store energy MONOMERS • • • Fatty acid chain • Carboxyl and hydrocarbon groups Saturated fat • Carbon are full of hydrogen • No double bonds • Solids, unhealthy Unsaturated fat • Contain double bonds • Liquid, healthier POLYMERS • • Triglyceride • 1 glycerol and 3 fatty acids Phospholipids • Component of cell membranes
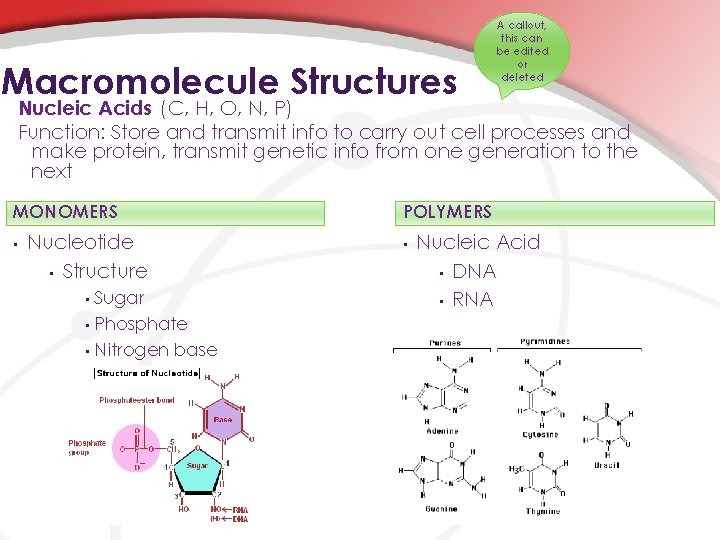
Macromolecule Structures A callout, this can be edited or deleted Nucleic Acids (C, H, O, N, P) Function: Store and transmit info to carry out cell processes and make protein, transmit genetic info from one generation to the next MONOMERS • Nucleotide • Structure Sugar • Phosphate • Nitrogen base • POLYMERS • Nucleic Acid • DNA • RNA

- Slides: 9