Biochemistry Dr Ron Rusay Fall 2001 Copyright 2001

Biochemistry Dr. Ron Rusay Fall 2001 © Copyright 2001 R. J. Rusay

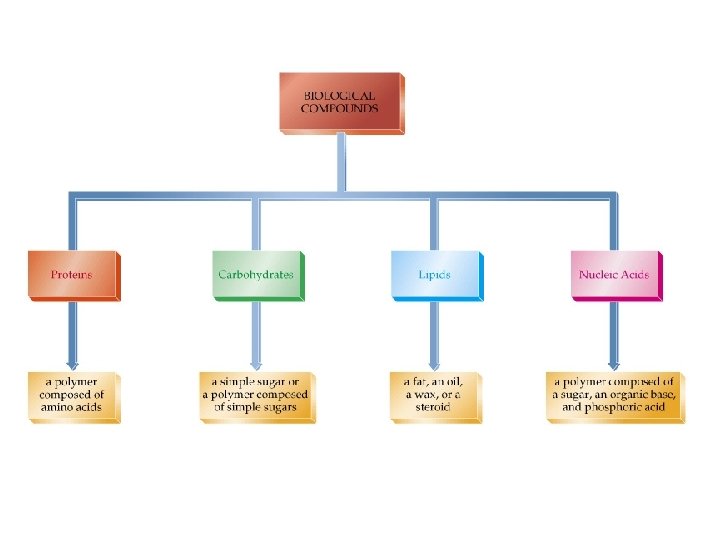
Biochemistry ðThe study of the chemistry of living things. ðThe molecular relationships between physiology, behavior and chemical structures. ðOrganic chemistry and Biochemistry are closely related.

Essential Elements ð There about 30 elements essential to human life, ~ 1/3 of the naturally occuring elements. By mass abundance:

Proteins ðNatural polymers made up of -amino acids (molecular weights range from 6000 to >1, 000 g/mol). ðFibrous Proteins: provide structural integrity and strength to muscle, hair and cartilage. The rotating image above is collagen, a fibrous protein found in skin, bone, muscle and connective tissue. It accounts for about 30% of all protein in humans. Collagen is a molecule made up of 3, 000 amino acids which are evenly distributed in three intertwined, cross-linked chains.

Proteins (continued) ð Globular Proteins: • act as catalysts. • are roughly spherical in shape. ð General Functions: • provide molecular and electron transport. • counter invasion by “foreign” chemicals. • regulate the body’s metabolic systems.

-Amino Acids ð NH 2 always attached to the -carbon (the carbon attached to COOH) ðC = -carbon ð Linking:

Protein Biosynthesis

Protein Bonds & -Amino Acids ð Amino acids combine to form a peptide Bond + H 2 O A peptide linkage ðThere are 20 different amino acids that are commonly found in proteins.

Peptide Bond Formation

Levels of Protein Structure ðPrimary: The sequence of amino acids in the protein chain. Sickle Cell Anemia ðSecondary: The arrangement of the protein chain in the long molecule. Helixes & Sheets. ðTertiary: The overall shape of the protein strand (determined by hydrogen-bonding, dipole-dipole interactions, ionic bonds, covalent bonds and London forces). ð Quaternary: The aggregate shape including all of the strands. Eg. Hemoglobin: 2 and 2

Enzymes ðHighly specialized proteins which catalyze specific biologic reactions.

Enzymes (continued) ðA “lock-and-key” model explains the enzyme’s mechanism. ðA “substrate” and enzyme interact via • • • ðThe H-bonding ionic bonding and/or metal ion-ligand bonding substrate occupies a highly specific “active site” of the enzyme.

Acetylcholinesterase Docking

Carbohydrates ðFormed from the action of light and chlorophyll in green plants: • n CO 2 + n H 2 O -----> Cn (H 2 O)n + n O 2 ðEmpirical formula = CH 2 O ðMonosaccharides (simple sugars) - C 5 : pentoses - ribose, arabinose - C 6 : hexoses - fructose, glucose - Can be aldose or ketose
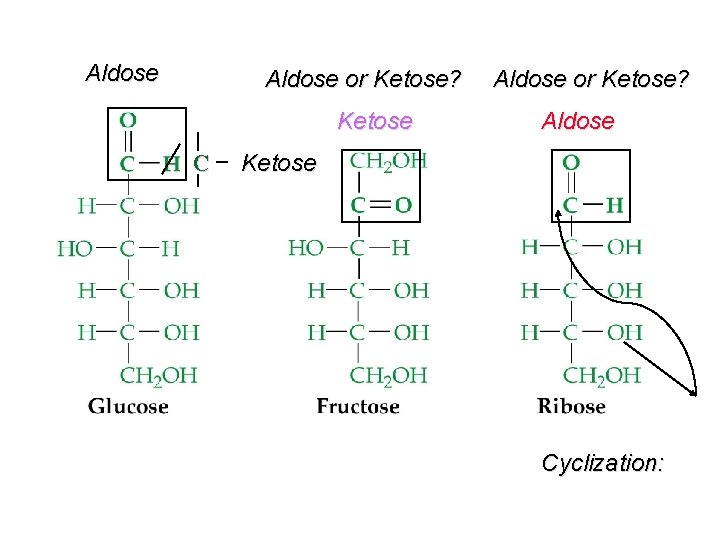
Aldose or Ketose? Ketose C Aldose or Ketose? Aldose Ketose Cyclization:
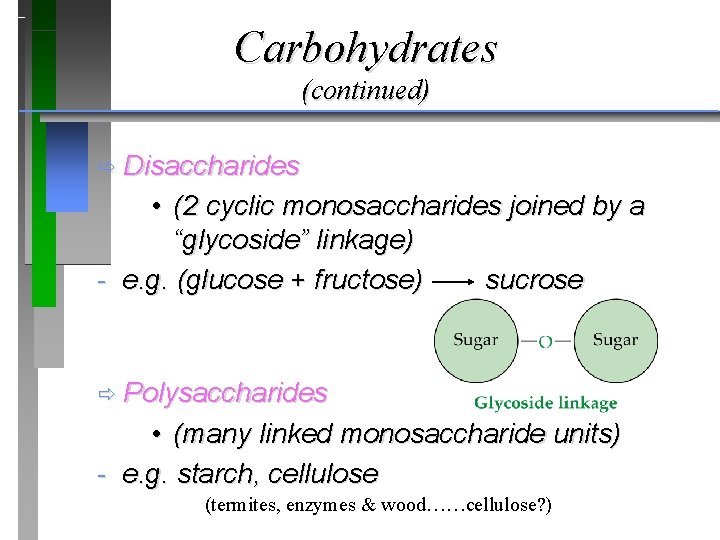
Carbohydrates (continued) ð Disaccharides • (2 cyclic monosaccharides joined by a “glycoside” linkage) - e. g. (glucose + fructose) sucrose ð Polysaccharides • (many linked monosaccharide units) - e. g. starch, cellulose (termites, enzymes & wood……cellulose? )

Nucleic Acids ð DNA: • • • encyclopedia of genetic information. directs protein synthesis. Molar mass = 10 9 Daltons (amu) ð RNA: • • deoxyribonucleic acids assist in protein synthesis. Messenger RNA transfer RNA Molecular weights = 20, 000 to 40, 000

DNA Replication
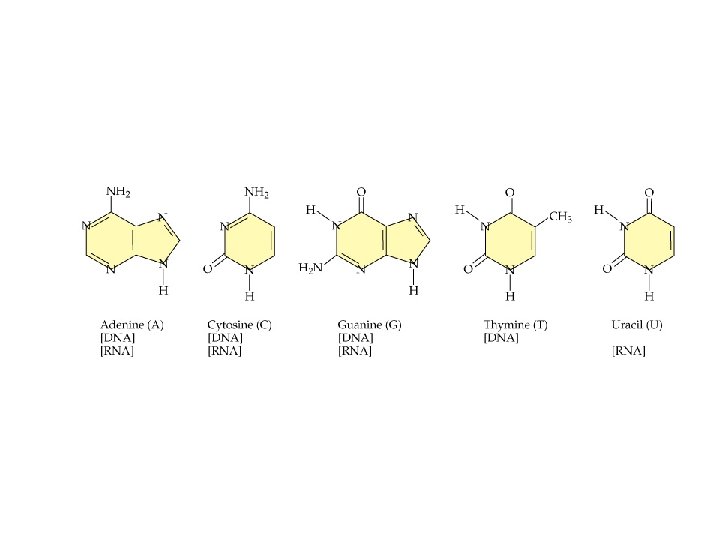
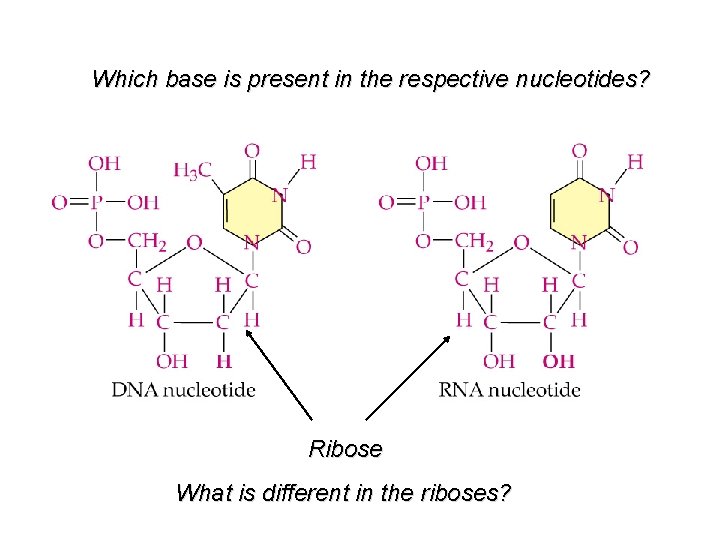
Which base is present in the respective nucleotides? Ribose What is different in the riboses?

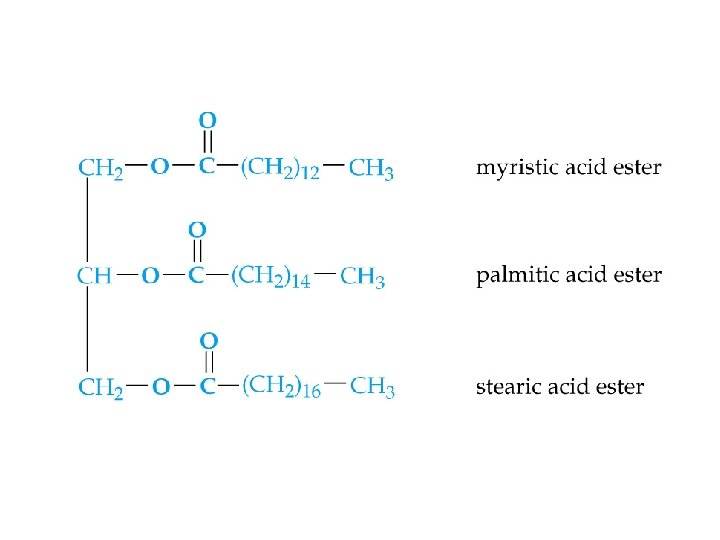
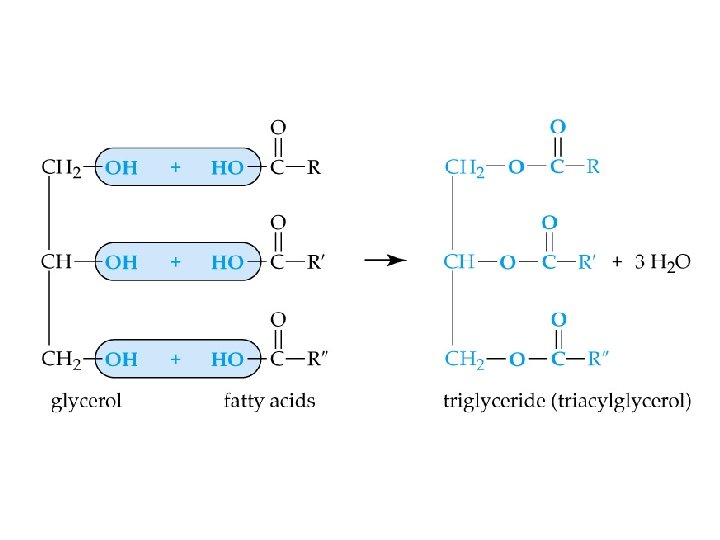
Lipids ðStructurally diverse natural products containing C, H and O ðNon-polar, water-insoluble compounds. • • fats (esters of fatty acids) phospholipids (contain phosphorus) waxes steroids

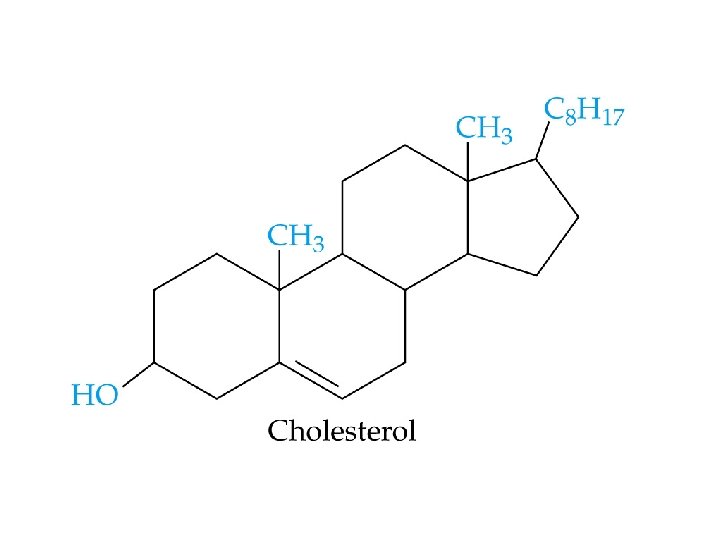
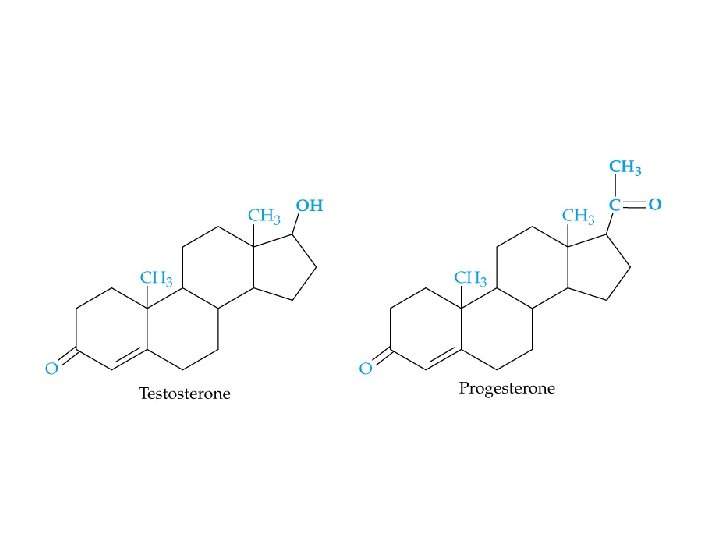
Steroids - Common structure: contains 4 rings - Examples: • • cholesterol adrenocorticoid hormones sex hormones bile acids
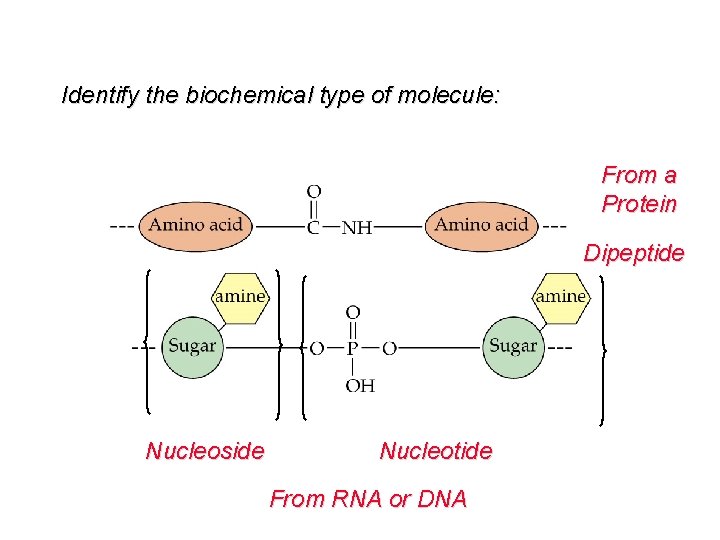
Identify the biochemical type of molecule: From a Protein Dipeptide Nucleoside Nucleotide From RNA or DNA
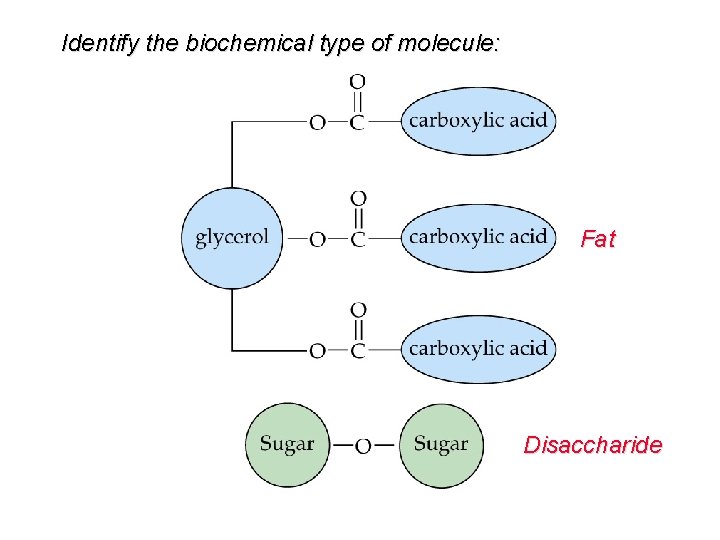
Identify the biochemical type of molecule: Fat Disaccharide
- Slides: 29