BIOCHEMISTRY CHEMISTRY OF LIFE Atomsbasic unit of matter
BIOCHEMISTRY

CHEMISTRY OF LIFE Atoms-basic unit of matter In 460 BC Democritis coined the term ‘atomos’ which means indivisible
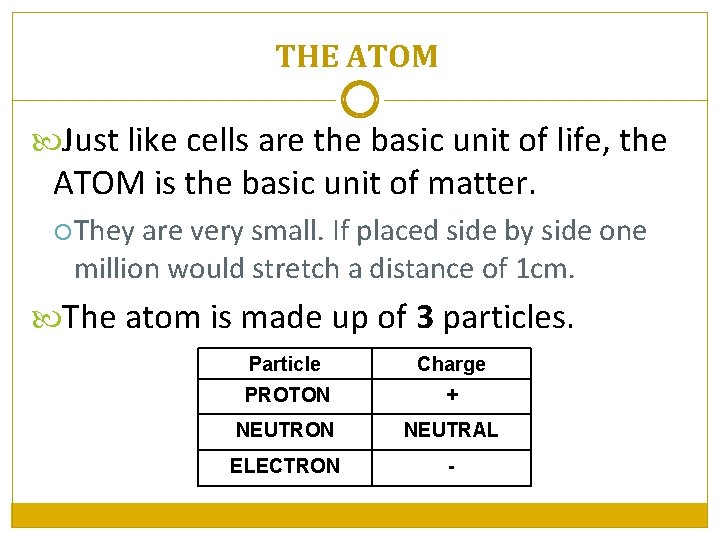
THE ATOM Just like cells are the basic unit of life, the ATOM is the basic unit of matter. They are very small. If placed side by side one million would stretch a distance of 1 cm. The atom is made up of 3 particles. Particle Charge PROTON + NEUTRON NEUTRAL ELECTRON -
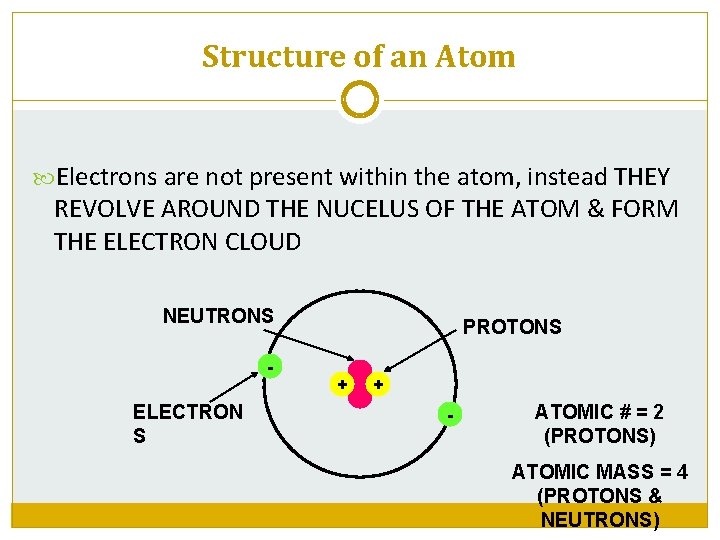
Structure of an Atom Electrons are not present within the atom, instead THEY REVOLVE AROUND THE NUCELUS OF THE ATOM & FORM THE ELECTRON CLOUD NEUTRONS - ELECTRON S PROTONS + + - ATOMIC # = 2 (PROTONS) ATOMIC MASS = 4 (PROTONS & NEUTRONS)

ISOTOPES Isotopes are atoms of the same element that HAVE A DIFFERENT NUMBER OF NEUTRONS Some isotopes are radioactive. This means that their nuclei is unstable and will break down at a CONSTANT RATE over time.

Elements-pure substance that consists of one type of atom How element are written… He 2 Atomic number 4 Atomic mass
Compounds A compound is pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions

TWO TYPES OF COMPOUNDS Organic - synthesized by cells and contain Carbon – made of carbon skeleton Carbohydrates, Proteins, Lipids, Nucleic Acids Inorganic - usually "support" life - no specific ratio of C, H, and O Water (H 2 O), Carbon Dioxide (CO 2)

4 Types of Organic Compounds Carbohydrates Lipids Proteins Nucleic Acids

Building Macromolecules are large molecules called polymers These polymers are composed of monomers Reactions: Monomers are connection to produce polymers Polymers are broken down into their monomers Enzymes assist in both reactions!

Dehydration Synthesis Combining simple molecules to form a more complex one with the removal of water Ex. monosaccharide + monosaccharide ----> disaccharide + water C 6 H 12 O 6 + C 6 H 12 O 6 ----> C 12 H 22 O 11 + H 2 O Polysaccharides are formed from repeated dehydration syntheses of water


Hydrolysis Addition of WATER to a compound to SPLIT it into smaller subunits (also called chemical digestion) ex. disaccharide + H 2 O ---> monosaccharide + monosaccharide C 12 H 22 O 11 + H 2 O ---> C 6 H 12 O 6 + C 6 H 12 O 6


CARBOHYDRATES Consist of C, H, O in a 1: 2: 1 ratio Main source of ENERGY for living things Function- to breakdown of sugar during digestion Supplies immediate energy to cells; excess is stored as complex sugars in cells

3 Types of Carbohydrates 1. Monosaccharides 2. Disaccharides 3. Polysaccharides

Monosaccharides Monomer is a simple sugar All have the formula C 6 H 12 O 6 Also all have a single ring structure Examples: Glucose, Fructose, and Galactose Shape of molecule determines how it reacts
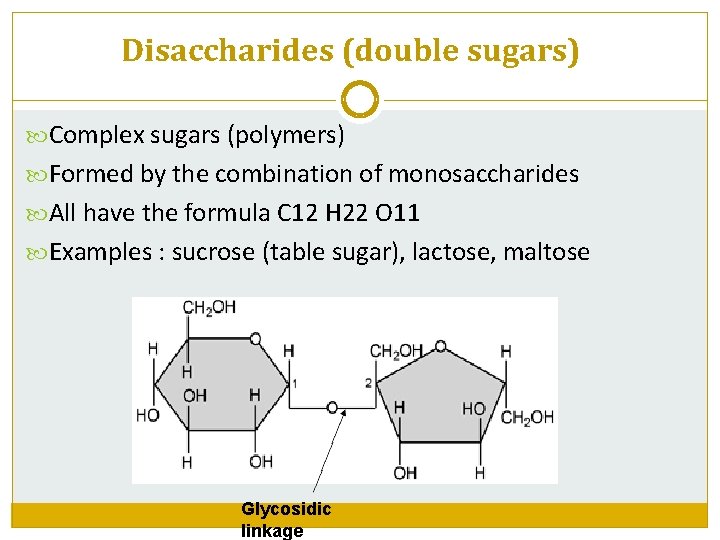
Disaccharides (double sugars) Complex sugars (polymers) Formed by the combination of monosaccharides All have the formula C 12 H 22 O 11 Examples : sucrose (table sugar), lactose, maltose Glycosidic linkage
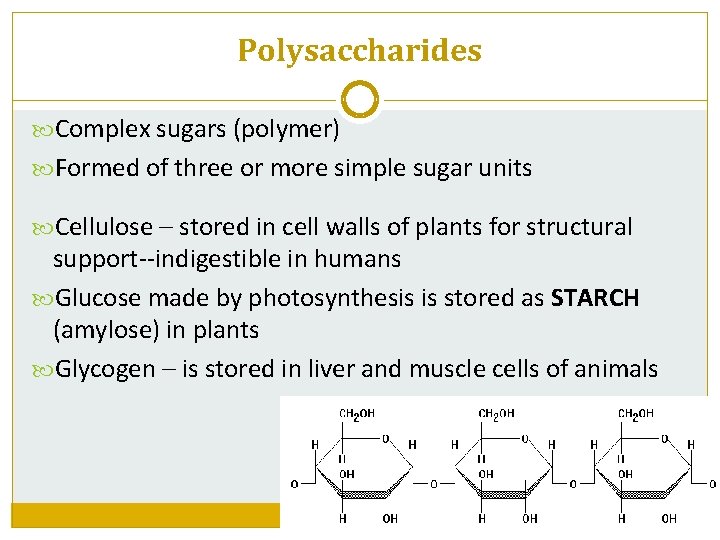
Polysaccharides Complex sugars (polymer) Formed of three or more simple sugar units Cellulose – stored in cell walls of plants for structural support--indigestible in humans Glucose made by photosynthesis is stored as STARCH (amylose) in plants Glycogen – is stored in liver and muscle cells of animals

Lipids (Fats) Consist of C, H, O Waxes, oils, fats, steroids (cholesterol & sex hormones) hydrophobic – insoluble in water

Functions of Lipids Energy Storage – breakdown of lipids provides long-term energy supply; excess is stored in fat cells; yields twice as much energy as carbohydrates Component of cell membrane (phospholipids) Provides cushions, insulates and waterproofing (wax)
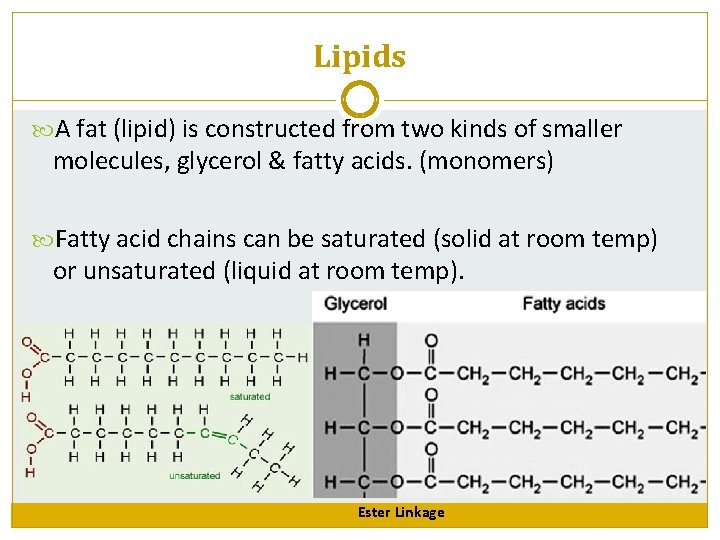
Lipids A fat (lipid) is constructed from two kinds of smaller molecules, glycerol & fatty acids. (monomers) Fatty acid chains can be saturated (solid at room temp) or unsaturated (liquid at room temp). Ester Linkage

Nucleic Acids consist of C, H, O, N, P Polymer of nucleotides (monomer) stores and transmits genetic information Made of 3 parts: 1. 5 -Carbon Sugar 2. Phosphate Group 3. Nitrogenous Base Examples of nongenetic nucleotides - plays a major role in cell metabolism 1. ATP (adenosine triphosphate) – carries energy for cellular activities 2. Subunits of coenzymes – enzyme helpers that transport hydrogen atoms plus electrons from one reaction site to another (examples – NAD+, FAD)
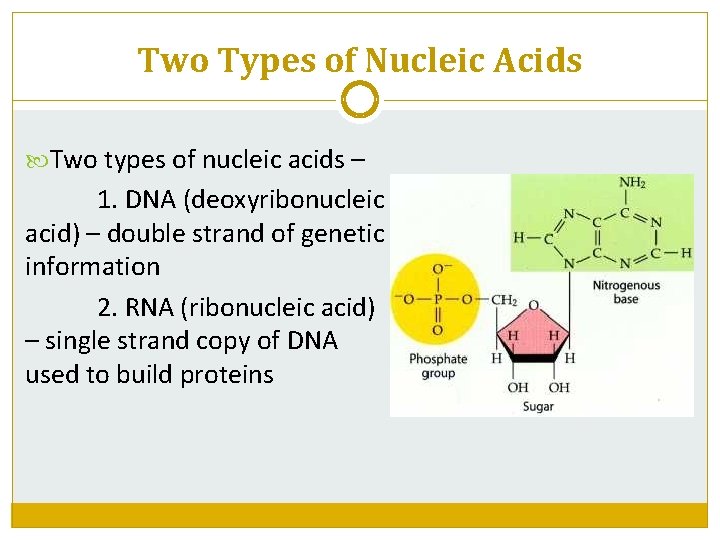
Two Types of Nucleic Acids Two types of nucleic acids – 1. DNA (deoxyribonucleic acid) – double strand of genetic information 2. RNA (ribonucleic acid) – single strand copy of DNA used to build proteins

Proteins Consist of C, H, O, N, S Polymers made of amino acids (monomer) All 20 amino acids have the same structure – but the R group is different. The “R” group may vary in size, shape, charge, hydrophobicity and reactivity. The sequence of amino acids will determine which protein is made Peptide bond forms between amino acids
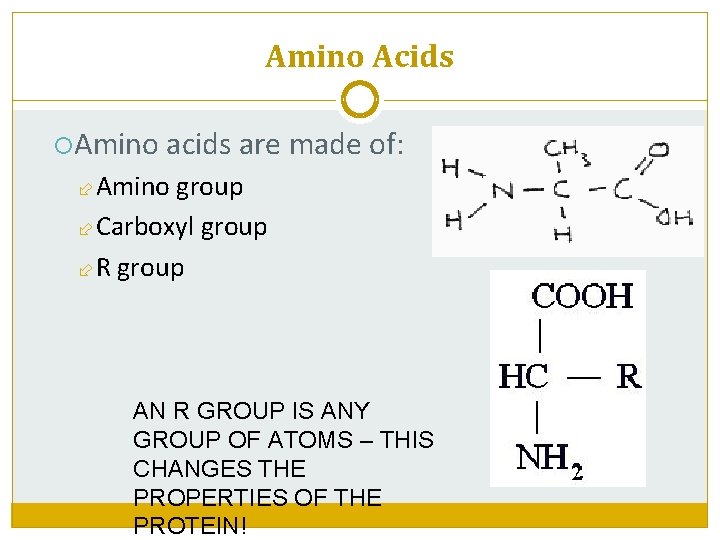
Amino Acids Amino acids are made of: Amino group Carboxyl group R group AN R GROUP IS ANY GROUP OF ATOMS – THIS CHANGES THE PROPERTIES OF THE PROTEIN!

Functions of Proteins Structural element of hair/nails (keratin) & bone/cartilage (collagen) Increase rate of reaction as an enzyme (biological catalyst) Transport and storage of molecules Control of metabolism Receptor proteins - signaling from cell to cell Tissue defense (antibodies)

CHEMICAL REACTIONS A process that changes one set of chemicals into another set of chemicals REACTANTS – elements or compounds that enter into a chemical reaction PRODUCTS – elements or compounds that are produced in a chemical reaction What is something that can speed up a reaction? ENZYMES!

Enzymes are proteins. Enzymes are biological catalyst. Catalyst speed up reactions. These reactions would take place anyway… the enzymes just speed them up! (Most enzyme names end in –ase)
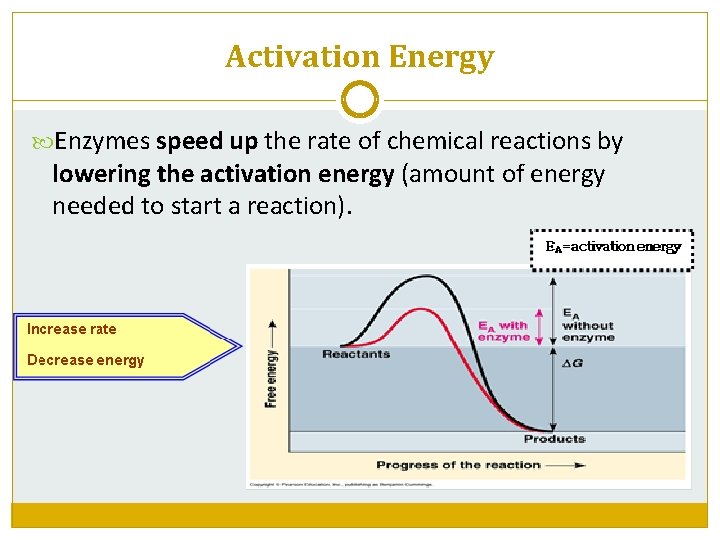
Activation Energy Enzymes speed up the rate of chemical reactions by lowering the activation energy (amount of energy needed to start a reaction).

CHARACTERISTICS of Enzymes do not make anything happen that couldn’t happen on its own, just makes it happen faster. Enzymes are not used up in reactions. They can be used over and over again! Enzymes are only needed in small amounts. Each enzyme is highly selective about its substrate. Enzymes chemically recognize, bind and modify substrates.

How Do Enzymes Work? Enzymes have a specific shape. Part of the enzyme matches the shape of the molecule to be reacted called the substrate. The part of the enzyme that binds to the substrate is the active site. When the substrate and enzyme bind temporarily, an enzyme-substrate complex is formed. The activation energy needed for the reaction to occur is reduced. After the reaction is complete, the substrate has formed a new product and the enzyme is released to be reused.
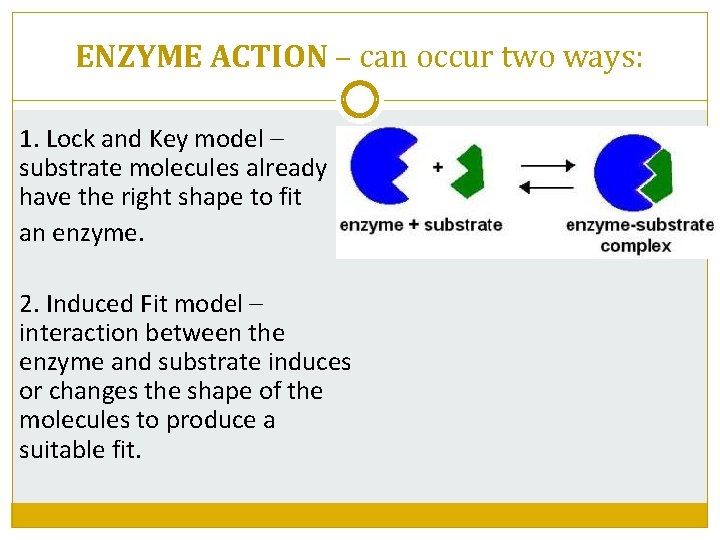
ENZYME ACTION – can occur two ways: 1. Lock and Key model – substrate molecules already have the right shape to fit an enzyme. 2. Induced Fit model – interaction between the enzyme and substrate induces or changes the shape of the molecules to produce a suitable fit.
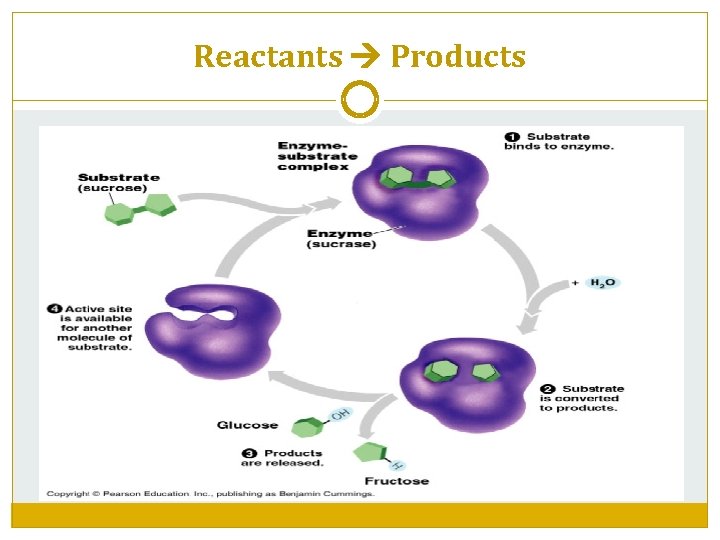
Reactants Products

FACTORS AFFECTING ENZYME ACTIVITY – Temperature p. H Concentration of Enzymes or Substrates Enzyme activators - Co-enzymes Enzyme inhibitors

Factors Influencing Rate of Enzyme Action Temperature – Enzymes function optimally at certain temperatures. High temps – inactive or denature enzyme Warm temps – increase speed Cold temps – work slowly or not at all
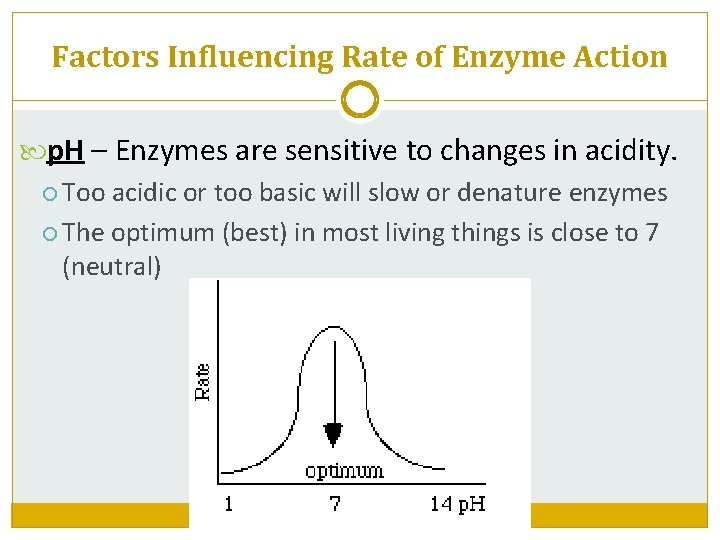
Factors Influencing Rate of Enzyme Action p. H – Enzymes are sensitive to changes in acidity. Too acidic or too basic will slow or denature enzymes The optimum (best) in most living things is close to 7 (neutral)
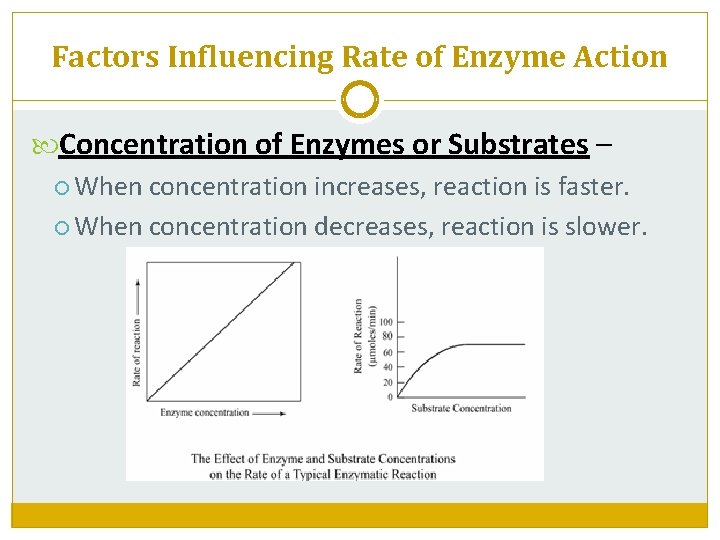
Factors Influencing Rate of Enzyme Action Concentration of Enzymes or Substrates – When concentration increases, reaction is faster. When concentration decreases, reaction is slower.
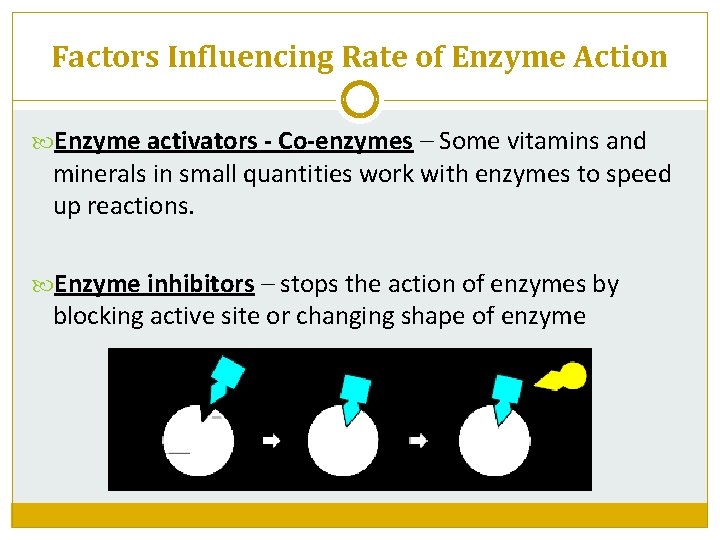
Factors Influencing Rate of Enzyme Action Enzyme activators - Co-enzymes – Some vitamins and minerals in small quantities work with enzymes to speed up reactions. Enzyme inhibitors – stops the action of enzymes by blocking active site or changing shape of enzyme
- Slides: 39