Biochemistry Carbohydrates Lipids Unit 3 Macromolecules l A

Biochemistry: Carbohydrates & Lipids Unit 3

Macromolecules l A. What are they? Very large molecules that make l 1. _________________ most of the structure of the body ____________________ l 2. Made of smaller pieces called monomers that can be assembled like _____ legos to form a variety of structures. A large chain of monomers is called a polymer _____. l Mono- one Poly - many

Carbohydrates

I. Macromolecules l B. Carbohydrates monosaccharide (sugar) l 1. Monomer: __________ polysaccharide (starch) l 2. Polymer: ___________ rings of carbon with l 3. Structure: ___________ oxygen and hydrogen attached; CH 2 O ________________ energy, plant structure l 4. Uses: __________ sucrose, cellullose l 5. Examples: ________

Carbohydrates l. Carbohydrates include: l Simple sugars: small sugar molecules in soft drinks l. Complex Carbohydrates: Long starch molecules in pasta and potatoes

Monosaccharides -OSE ending means SUGAR l. Glucose is found in sports drinks Fructose is found in fruits Honey contains both glucose & fructose Galactose is called “milk sugar” 6

Question? ? ? l. What does the suffix –ose mean? l. Sugar l. Where are glucose molecules found? l. In sodas, candies, any sweet snacks l. Where would you find fructose? l. Fruit 7
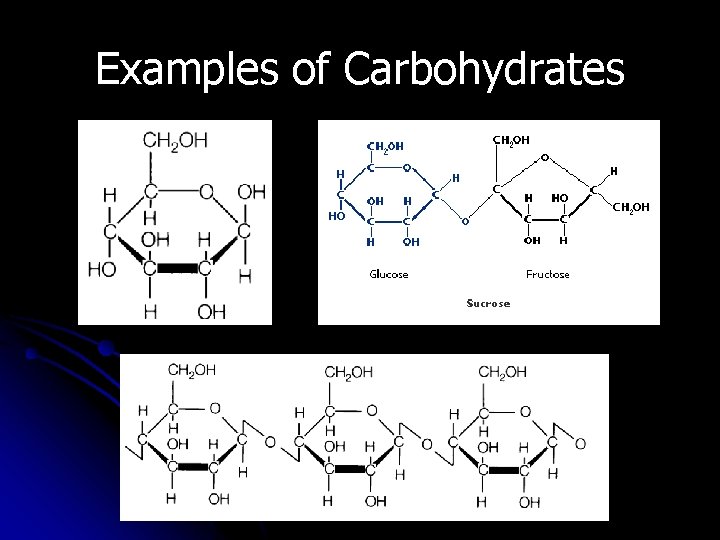
Examples of Carbohydrates

Disaccharides Di = 2 l. A disaccharide is a double sugar They’re made by joining two monosaccharides Involves removing a water molecule (condensation) 9

Disaccharides l Common disaccharides include: Ø Sucrose (table sugar) Ø Lactose (Milk Sugar) Ø Maltose (Grain sugar) 10

Disaccharides 11

Polysaccharides l. Complex carbohydrates Composed of many sugar monomers linked together Polymers of monosaccharide chains 12
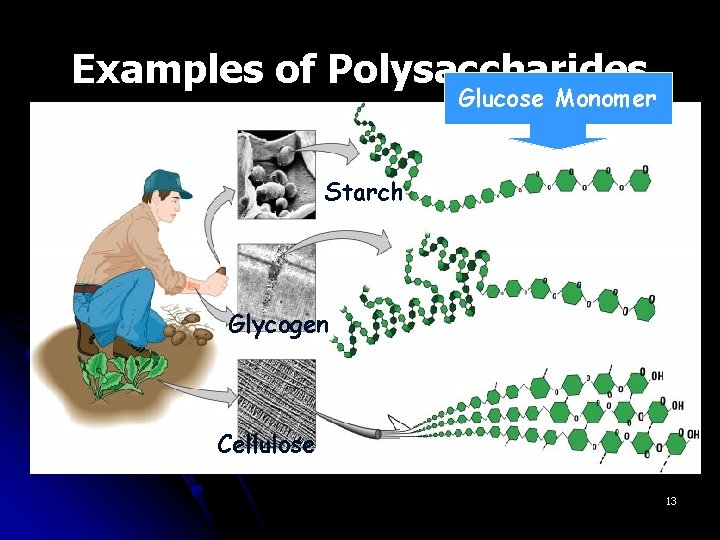
Examples of Polysaccharides Glucose Monomer Starch Glycogen Cellulose 13

Question? ? l. What does the prefix poly- mean again? Mono-? l. Many, one l. Polymers what? lmany of monosaccharide chains means sugar monomers linked together 14

Lipids

II. Macromolecules Lipids fatty acid l 1. Monomer: _____ lipid or fat l 2. Polymer: ______ 3 long chains of carbon l 3. Structure: ___________ hydrogen on a glycerol molecule ________________ energy, structure, warmth l 4. Uses: ____________ fat, oil, cholesterol l 5. Examples: __________

Function of Lipids l Fats store energy, helps to insulate the body, cushion and protect organs, and makes up the cell membrane (lipid bilayer) 17

Lipids l Lipids are molecules that consist of long hydrocarbon chains. Attaching the three chains together is usually a glycerol molecule. Lipids are NONpolar.

Lipids & Cell Membranes • • • Cell membranes are made of lipids called phospholipids Phospholipids have a head that is polar & attract water (hydrophilic) Phospholipids also have 2 tails that are nonpolar and do not attract water (hydrophobic) 19
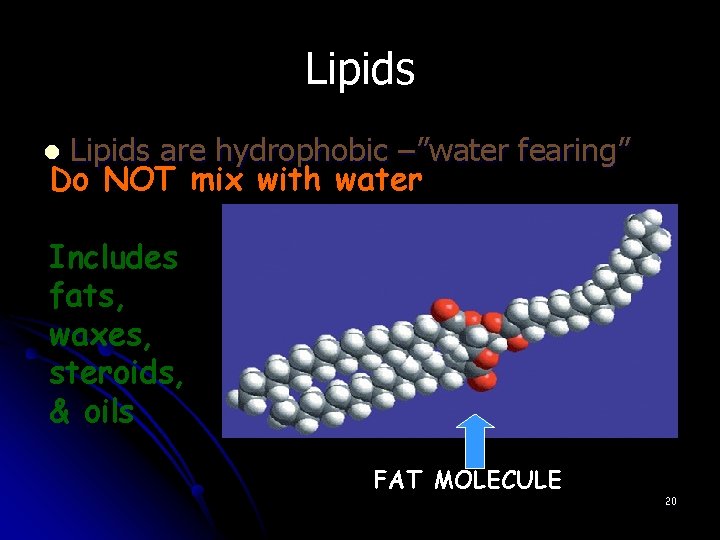
Lipids are hydrophobic –”water fearing” Do NOT mix with water l Includes fats, waxes, steroids, & oils FAT MOLECULE 20
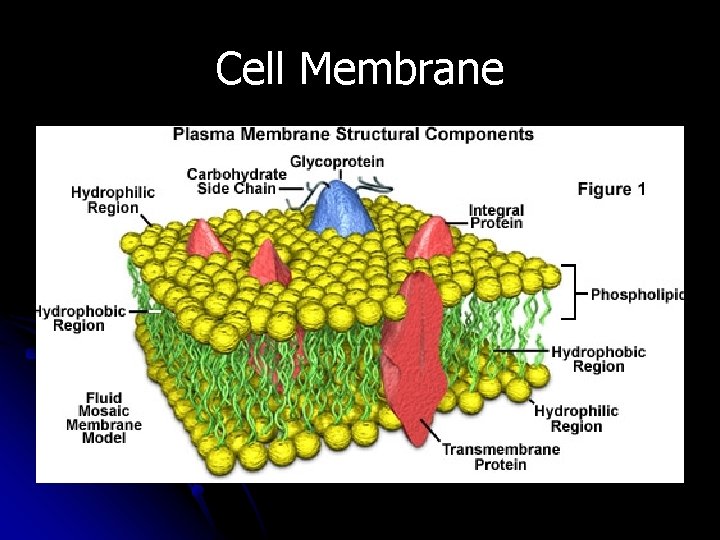
Cell Membrane

Fats in Organisms l Most animal fats have a high proportion of saturated fatty acids & exist as solids at room temperature (butter, margarine, shortening) these are called saturated fats

Fats in Organisms l Most plant oils tend to be low in saturated fatty acids & exist as liquids at room temperature (oils) these are called unsaturated fats
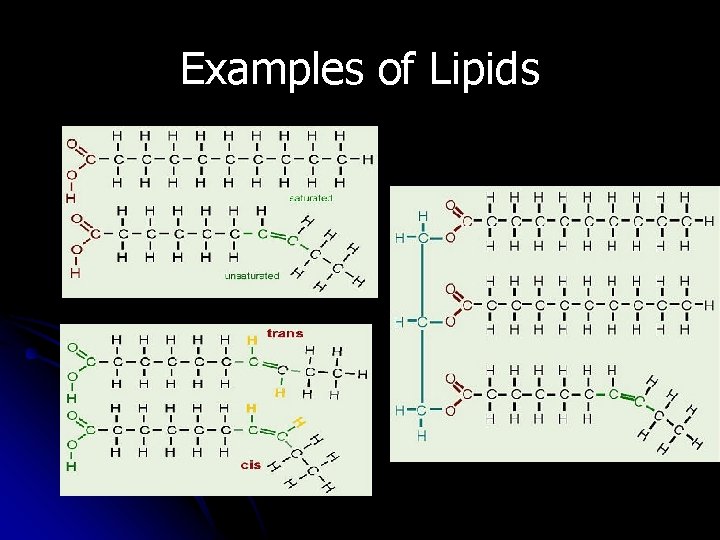
Examples of Lipids

Take the Quiz on Edmodo l Log in and complete the short quiz on edmodo. com

Biochemistry: Proteins & Nucleic Acids Unit 3

III. Macromolecules l D. Protein amino acid (20) l 1. Monomer: ________ protein or polypeptide l 2. Polymer: ___________ central carbon atom with l 3. Structure: ___________________________ hydrogen, amine, carboxyl, & R groups structure, emergency energy l 4. Uses: ____________ skin, insulin, enzymes l 5. Examples: __________

Four Types of Proteins Storage Structural Contractile Transport 28

Proteins l Proteins are building blocks of structures called amino acids. Proteins are what your DNA codes to make A peptide bond forms between amino acids by dehydration synthesis. l Dehydration synthesis = the building up of large molecules by removing water molecules l

Examples of Proteins
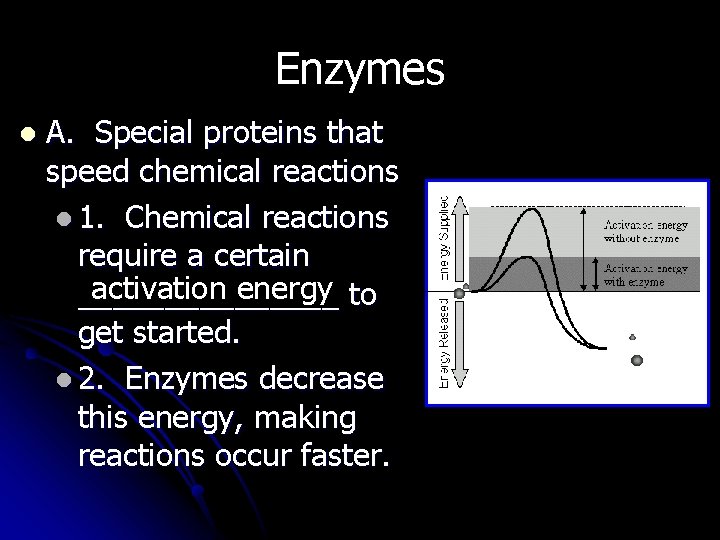
Enzymes l A. Special proteins that speed chemical reactions l 1. Chemical reactions require a certain activation energy to ________ get started. l 2. Enzymes decrease this energy, making reactions occur faster.

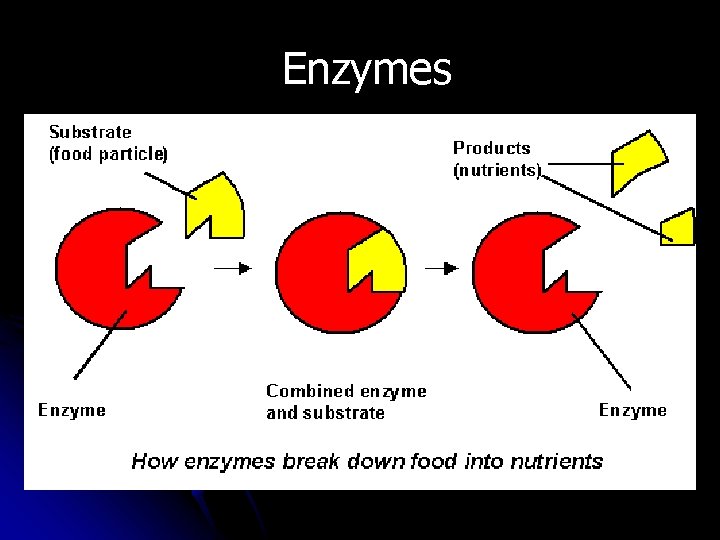
Enzymes l B. Lock-and-Key Model l 1. Enzymes are not used up by the reaction, but each can only work on one enzyme specificity reaction (________). l 2. This is called the lock-and-key model key of enzymes. An enzyme is like a _____ which can open exactly one _____. lock If you want to “unlock” another reaction, you need a different enzyme.

Enzymes l C. Factors which affect enzymes Temperature l 1. _______--enzymes, like all proteins, change shape when exposed to heat or cold. Each has an optimal temperature range. p. H l 2. ____--all enzymes have an optimal range of p. H. Example: stomach Concentration l 3. _______--having more enzymes makes the reaction faster.
Enzymes

Biochemistry: Nucleic Acid & ATP Unit 3

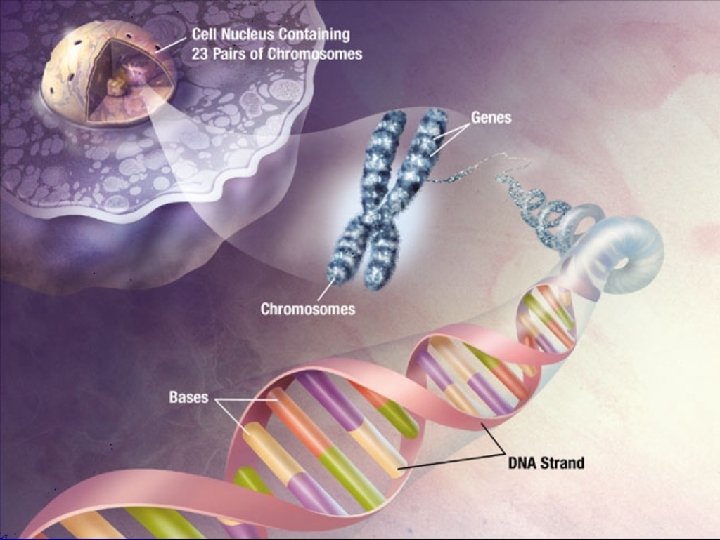
III. Macromolecules l E. Nucleic Acids nucleotide (5) l 1. Monomer: ________ nucleic acid l 2. Polymer: ______ 5 -carbon sugar attached l 3. Structure: ___________ to nitrogen base and phosphate group ________________ stores genetic code l 4. Uses: __________ DNA and RNA l 5. Examples: ________
37

Examples of Nucleic Acids
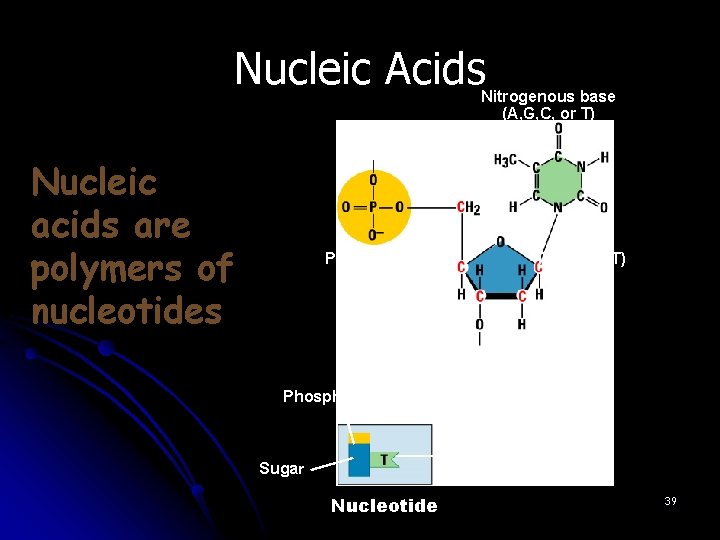
Nucleic Acids Nitrogenous base (A, G, C, or T) Nucleic acids are polymers of nucleotides Phosphate group Thymine (T) Sugar (deoxyribose) Phosphate Base Sugar Nucleotide 39
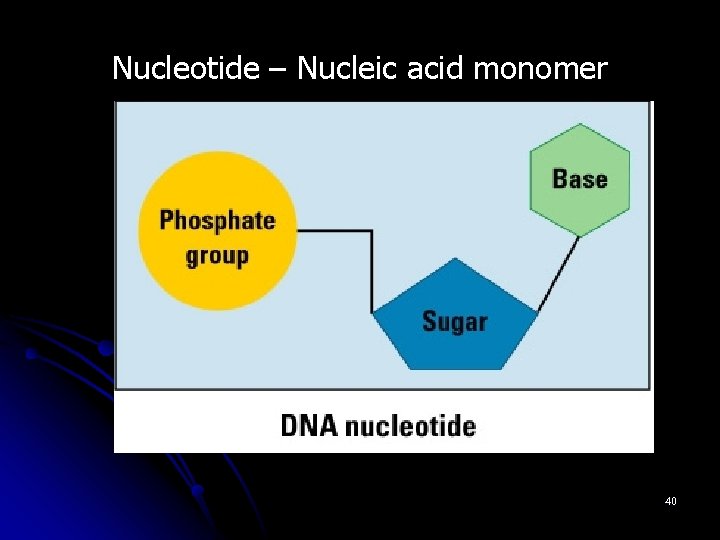
Nucleotide – Nucleic acid monomer 40
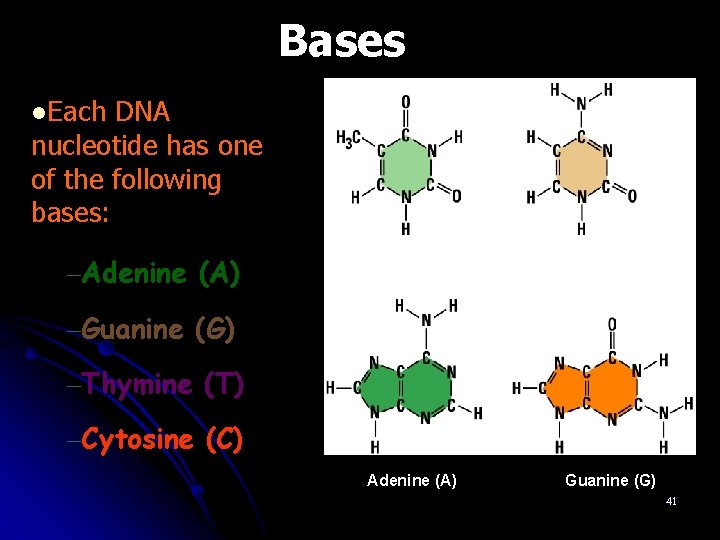
Bases l. Each DNA nucleotide has one of the following bases: –Adenine (A) Thymine (T) Cytosine (C) –Guanine (G) –Thymine (T) –Cytosine (C) Adenine (A) Guanine (G) 41
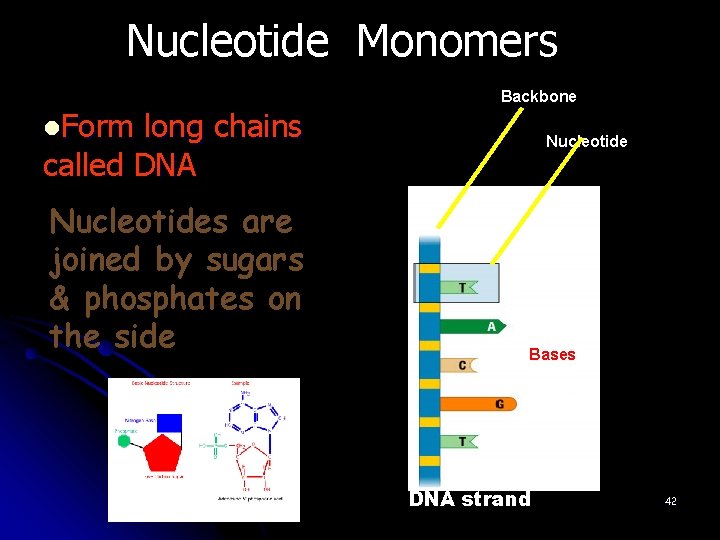
Nucleotide Monomers Backbone l. Form long chains called DNA Nucleotides are joined by sugars & phosphates on the side Nucleotide Bases DNA strand 42

DNA l. Two strands of DNA join together to form a double helix Base pair Double helix 43
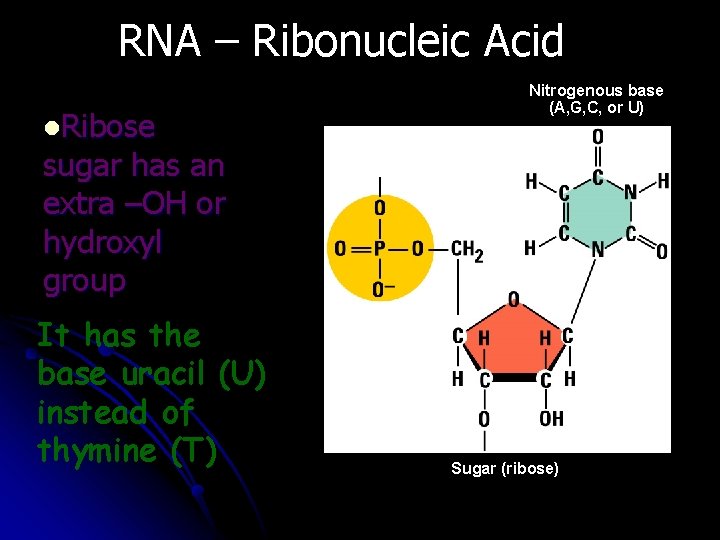
RNA – Ribonucleic Acid Nitrogenous base (A, G, C, or U) l. Ribose sugar has an extra –OH or hydroxyl group It has the base uracil (U) instead of thymine (T) Uracil Phosphate group Sugar (ribose)

IV. ATP adenosine triphosphate A. ATP stands for ___________ l B. Cells use ATP as a _________ rechargeable battery 3 phosphates l C. Made of adenine with ___ l D. Lots of energy is stored in the bond the second and third phosphates between _______________ l E. When this bond is broken, tremendous energy is released. l F. The pieces are then reassembled, storing more energy for another use. l

Question? ? l. Explain l. ATP the ATP/ADP process. has 3 phosphate groups. The energy is stored in the bond. When 1 phosphate group breaks off it releases energy and forming ADP. The addition of 1 phosphate to ADP forms ATP and the process continuous over and over.

Macromolecules

Macromolecules Copyright Cmassengale 48
- Slides: 48