Biochemistry Carbohydrates b Class of organic compounds contains

Biochemistry

Carbohydrates b Class of organic compounds contains C, H, O b monosaccharides • C 6 H 12 O 6 • hexoses – glucose – galactose – fructose
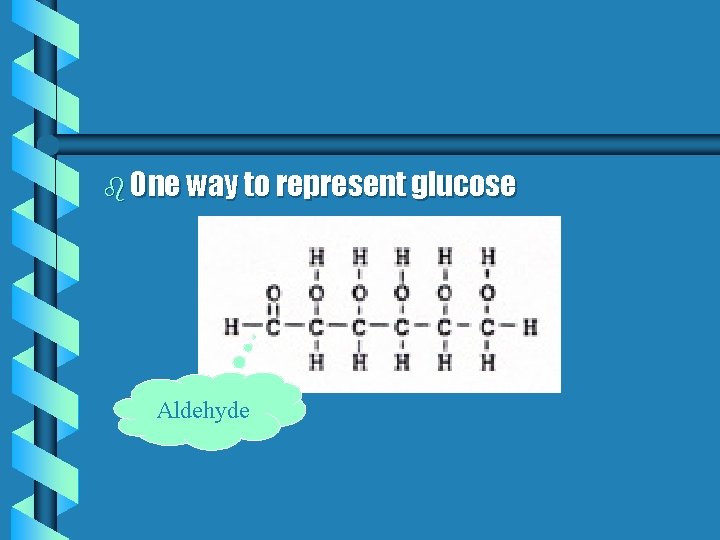
b One way to represent glucose Aldehyde
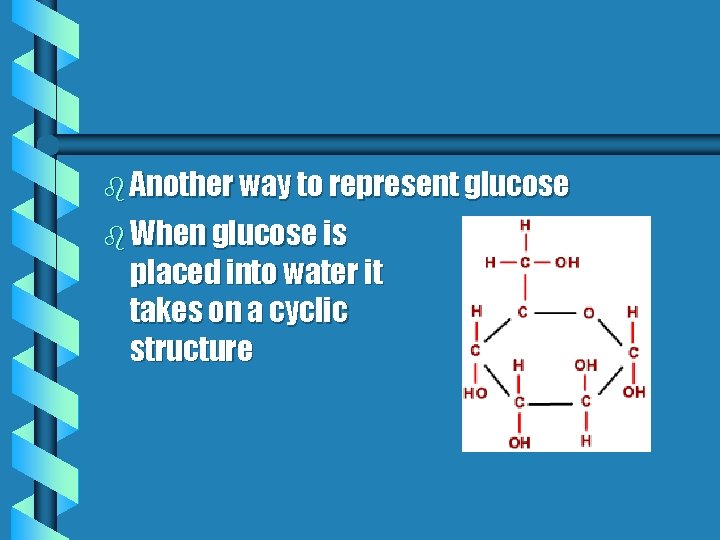
b Another way to represent glucose b When glucose is placed into water it takes on a cyclic structure
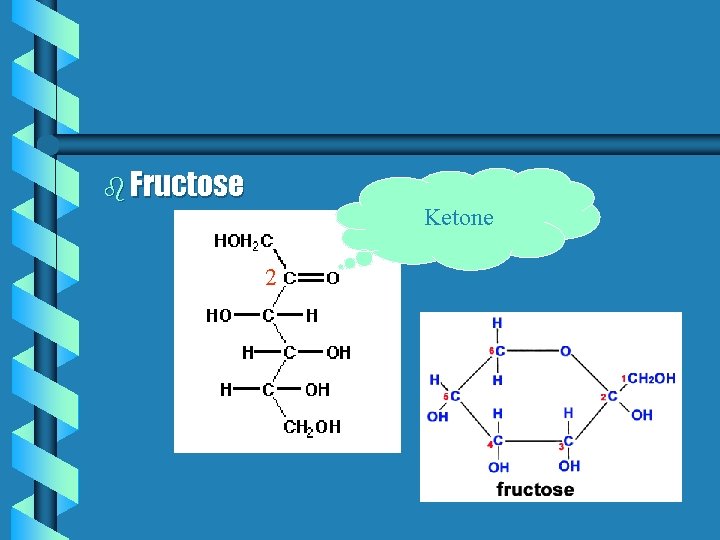
b Fructose Ketone 2

Polymerization b When small subunits are joined into longer compounds • When monosaccharides are in cyclic form they can bond together in looooong chains b many monosaccharides bonded together = polysaccharide
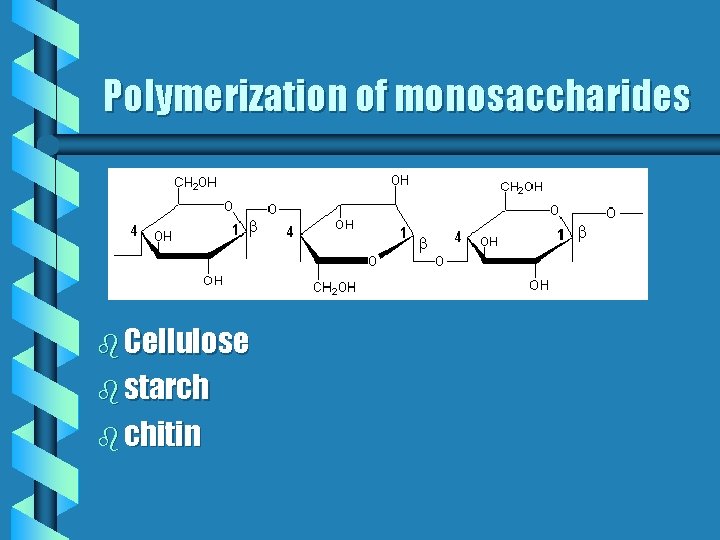
Polymerization of monosaccharides b Cellulose b starch b chitin
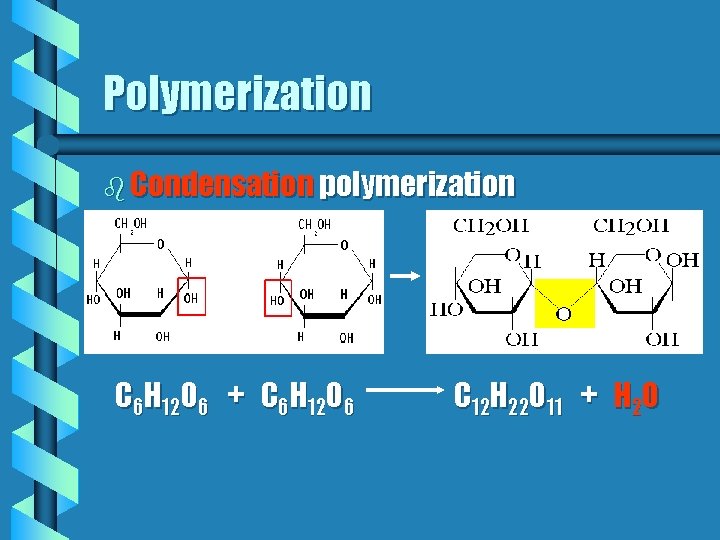
Polymerization b Condensation polymerization C 6 H 12 O 6 + C 6 H 12 O 6 C 12 H 22 O 11 + H 2 O
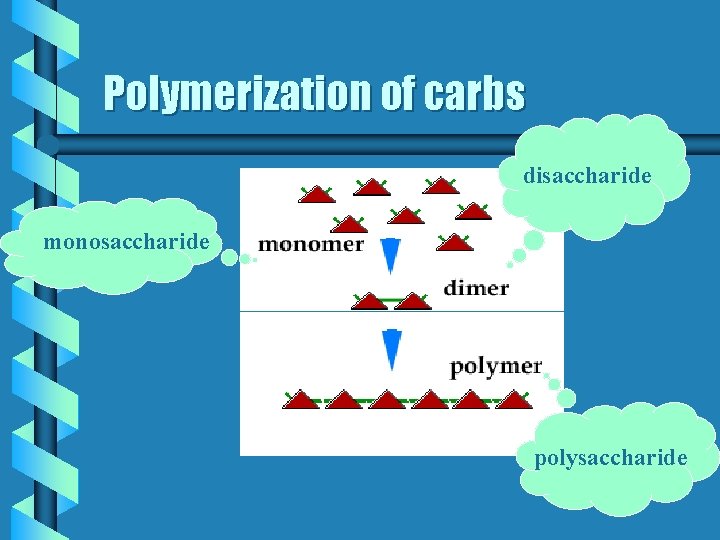
Polymerization of carbs disaccharide monosaccharide polysaccharide
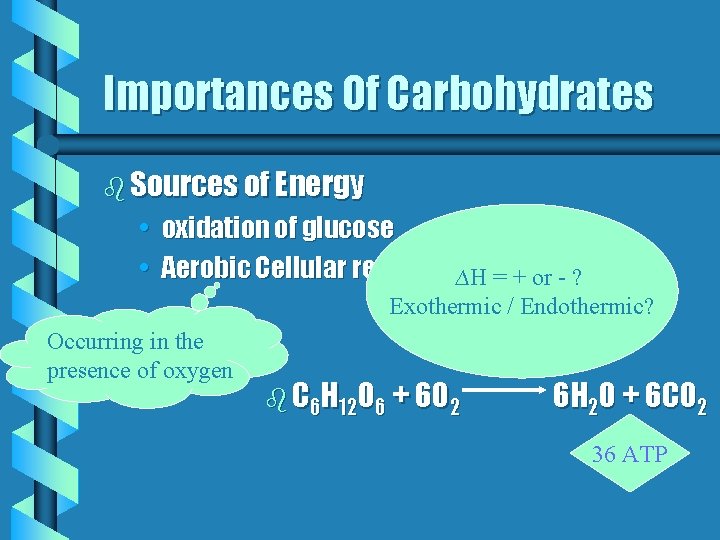
Importances Of Carbohydrates b Sources of Energy • oxidation of glucose • Aerobic Cellular respiration ∆H = + or - ? Exothermic / Endothermic? Occurring in the presence of oxygen b C 6 H 12 O 6 + 6 O 2 6 H 2 O + 6 CO 2 36 ATP

Impt #2 Occurs without oxygen b Fermentation • • anaerobic respiration process occurs with yeast b C 6 H 12 O 6 2 C 2 H 5 OH + 2 CO 2 2 ATP

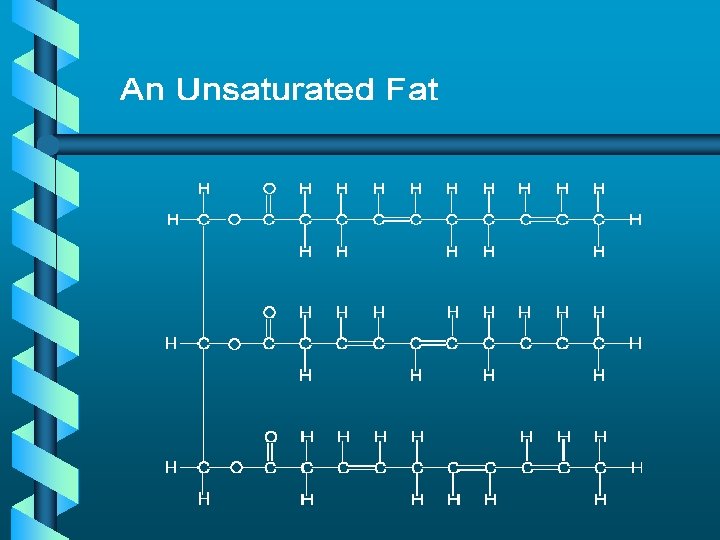
Lipids a. k. a. Fats b Monomers • 3 fatty acids • 1 glycerol
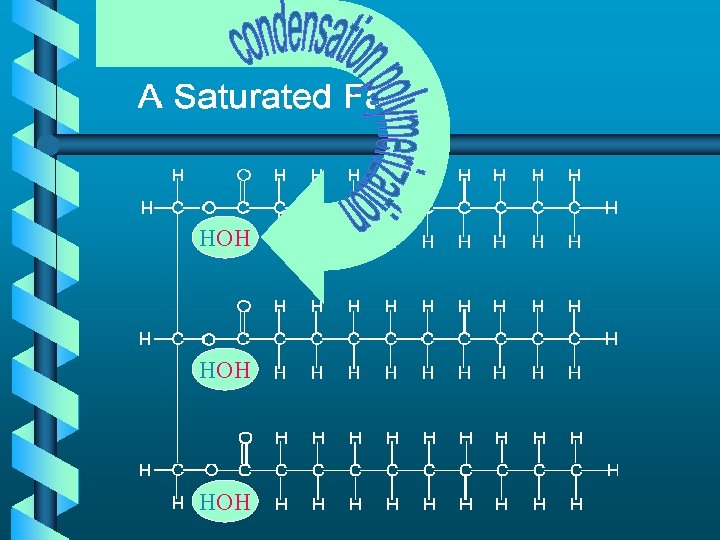
HOH HOH
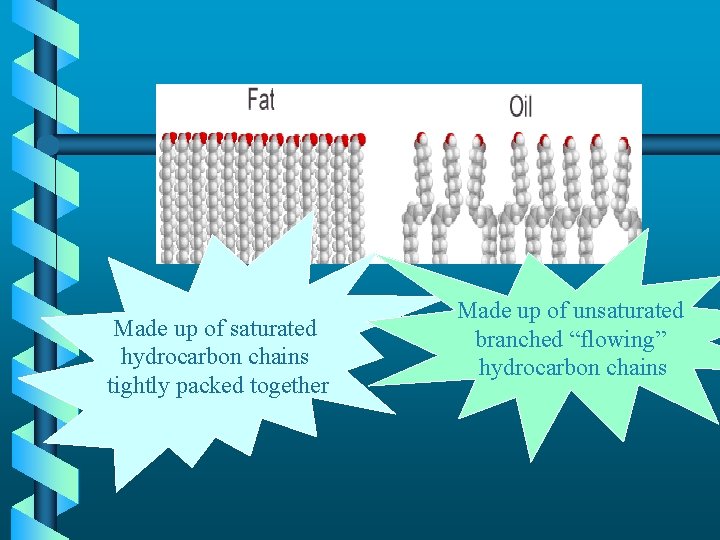
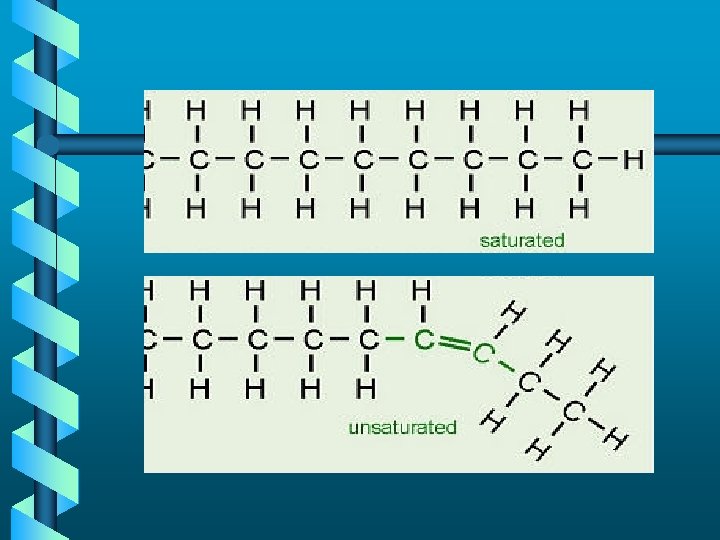
Made up of saturated hydrocarbon chains tightly packed together Made up of unsaturated branched “flowing” hydrocarbon chains
- Slides: 16