Biochemistry Biochemistry Study of the chemical composition and

Biochemistry

Biochemistry • Study of the chemical composition and reactions of living matter

Organic VS Inorganic • 2 major classes of chemicals in the body • Organic- Contain carbon – All compounds have covalent bonds • Inorganic- All others – Ex: water, salts, and many acids/bases

Inorganic Compounds • Water – Most abundant and important inorganic – 60 -80% of the volume of most living cells

Properties of Water • High heat capacity/ heat storage capacity • Polar Solvent – Hydration layers – Transport • Reactivity • Cushioning – Organs • High heat of vaporization – Removal of heat through perspiration and evaporation

Salts • Ionic compound • Electrolytes: substances that conduct an electrical current in sol’n. – Nerve impulse transmission and muscle contraction • Ex: Na. Cl, Ca. CO 3, KCl – Calcium phosphates: make bone/teeth hard

Homeostasis and Ions • Kidneys help to maintain proper ionic balance in body fluids

Acids and Bases • Acids: substance that releases H+ in detectable amounts (proton donors) HCl H+ + Cl – • Bases: Proton acceptors: release OH– Ex: Bicarbonate: abundant in the blood Na. OH Na+ + OH–

p. H • Relative concentration of hydrogen ions in various body fluids
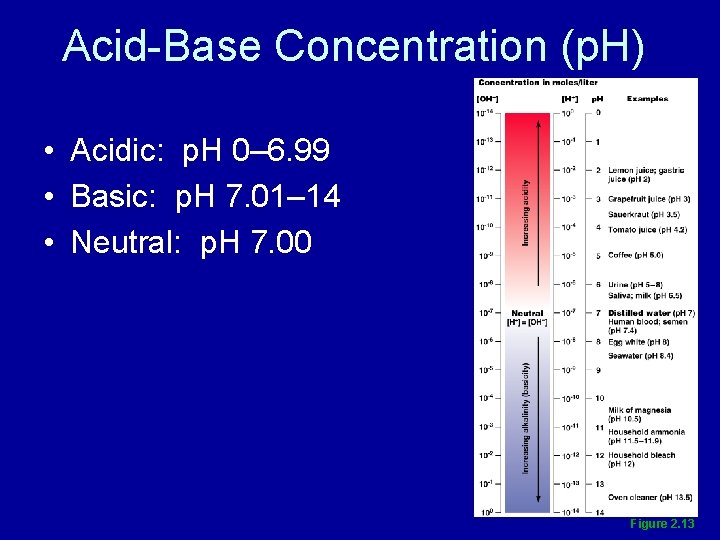
Acid-Base Concentration (p. H) • Acidic: p. H 0– 6. 99 • Basic: p. H 7. 01– 14 • Neutral: p. H 7. 00 Figure 2. 13

Neutralization Rxn Acid + Base Salt + Water

Buffers • Chemical systems that help regulate homeostatic p. H balance – Release Hydrogen ions when p. H increases and binds these ions when p. H drops.

Weak VS Strong • Strong: Dissociated completely and irreversibly- can dramatically change p. H – Hydrochloric Acid and Sulfuric Acid • Weak: Do not dissociate completely – bicarbonate

Carbonic Acid-Bicarbonate System Carbonic acid dissociates, reversibly releasing bicarbonate ions and protons The chemical equilibrium between carbonic acid and bicarbonate resists p. H changes in the blood

Organic Compounds • Unique to living systems • Contain carbon • Generally very large with functional groups – No Carbon dioxide, CO, etc

Carbohydrates • Contain carbon, hydrogen, and oxygen • Their major function is to supply a source of cellular food

Monosaccharides
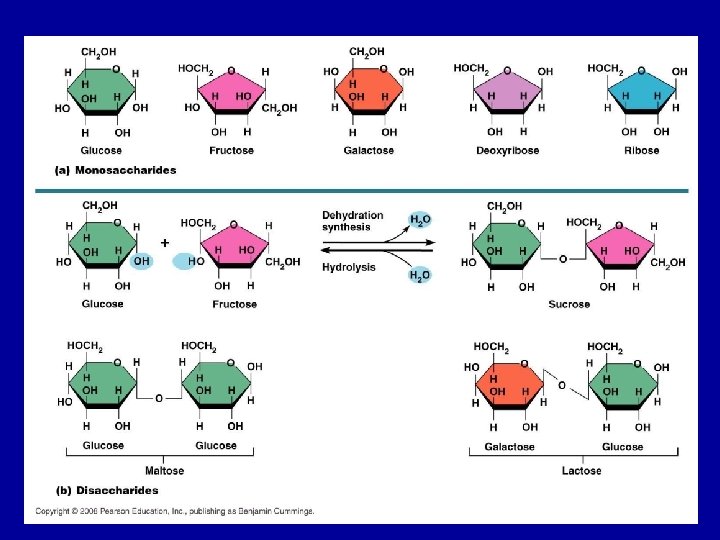
Disaccharides
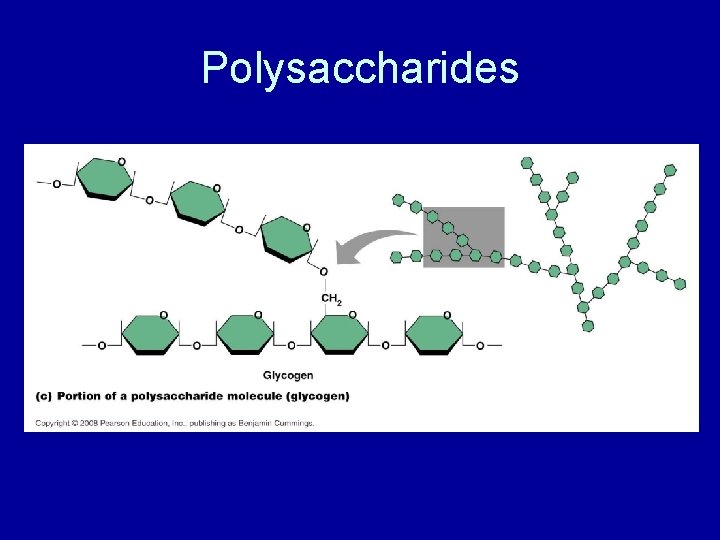
Polysaccharides

Functions of Carbs • Small amounts used for structural purposes • Ready, easily used cellular fuel – ATP synthesis • Converted to fat when ATP supplies are sufficient

Lipids • Contain C, H, and O, but the proportion of oxygen in lipids is less than in carbohydrates • Examples: • Neutral fats or triglycerides • Phospholipids • Steroids • Eicosanoids
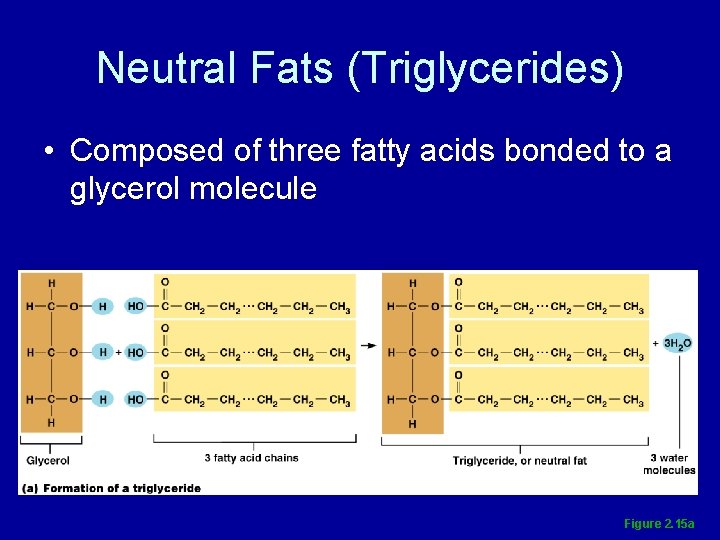
Neutral Fats (Triglycerides) • Composed of three fatty acids bonded to a glycerol molecule Figure 2. 15 a
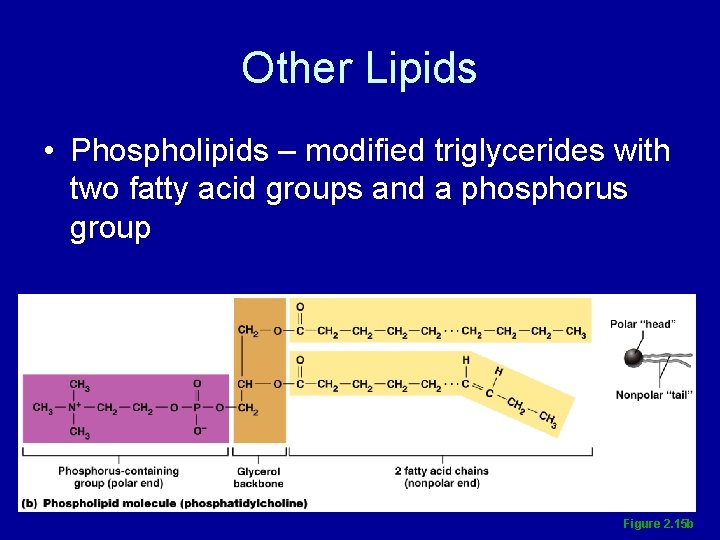
Other Lipids • Phospholipids – modified triglycerides with two fatty acid groups and a phosphorus group Figure 2. 15 b
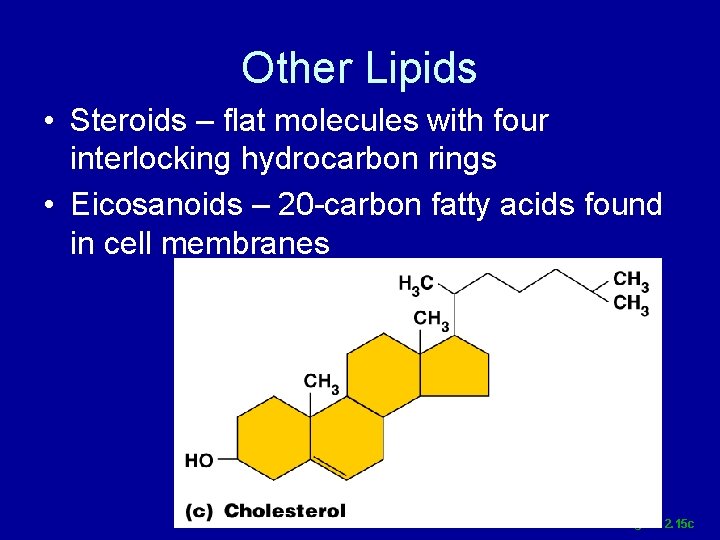
Other Lipids • Steroids – flat molecules with four interlocking hydrocarbon rings • Eicosanoids – 20 -carbon fatty acids found in cell membranes Figure 2. 15 c

Representative Lipids Found in the Body • Neutral fats – found in subcutaneous tissue and around organs • Phospholipids – chief component of cell membranes • Steroids – cholesterol, bile salts, vitamin D, sex hormones, and adrenal cortical hormones

Representative Lipids Found in the Body • Fat-soluble vitamins – vitamins A, E, and K • Eicosanoids – prostaglandins, leukotrienes, and thromboxanes • Lipoproteins – transport fatty acids and cholesterol in the bloodstream

Amino Acids • Building blocks of protein, containing an amino group and a carboxyl group • Amino group NH 2 • Carboxyl groups COOH
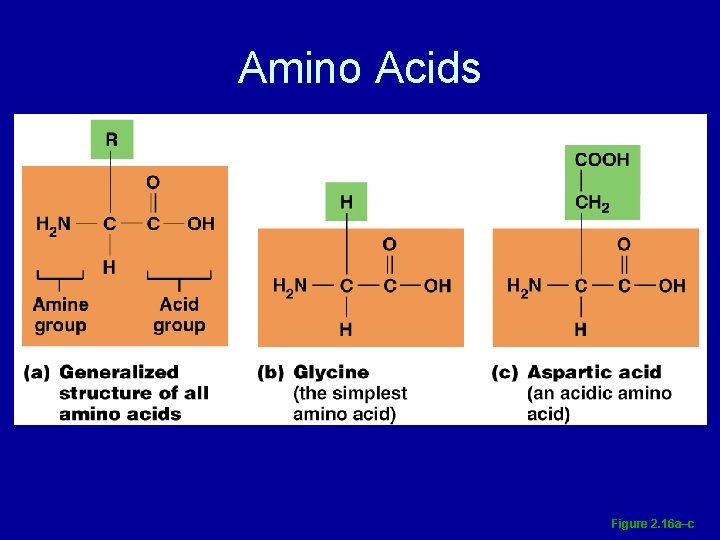
Amino Acids Figure 2. 16 a–c
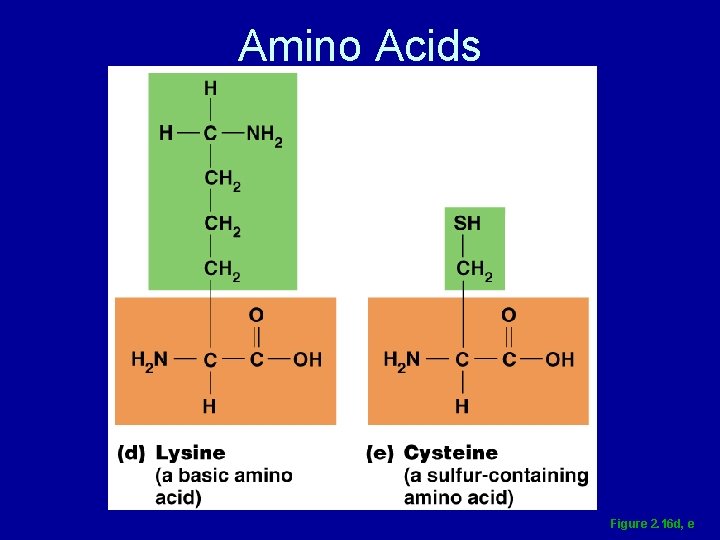
Amino Acids Figure 2. 16 d, e

Protein • Macromolecules composed of combinations of 20 types of amino acids bound together with peptide bonds Figure 2. 17
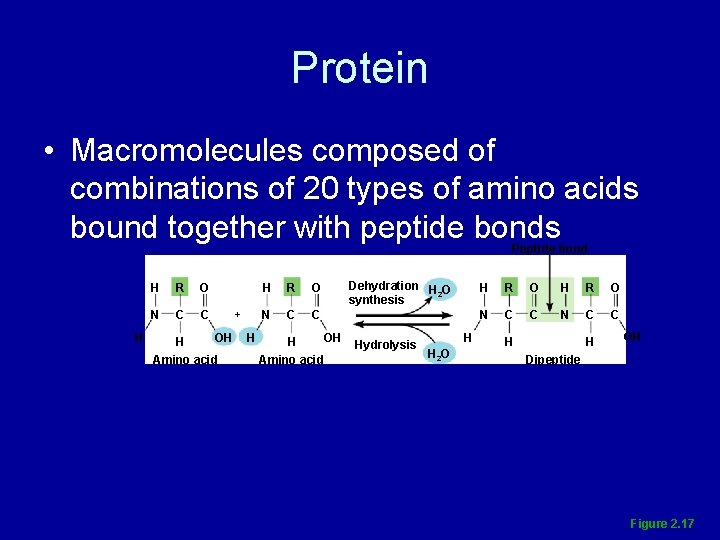
Protein • Macromolecules composed of combinations of 20 types of amino acids bound together with peptide bonds Peptide bond H H R O N C C OH H Amino acid + H H R O N C C OH H Amino acid Dehydration H O 2 synthesis Hydrolysis H H 2 O H R O N C C H H OH Dipeptide Figure 2. 17

Structural Levels of Proteins • Primary – amino acid sequence • Secondary – alpha helices or beta pleated sheets • Tertiary – superimposed folding of secondary structures • Quaternary – polypeptide chains linked together in a specific manner
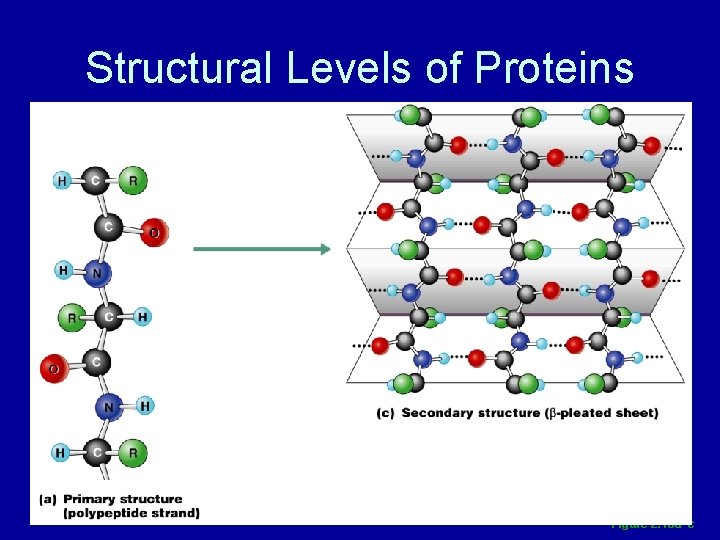
Structural Levels of Proteins Figure 2. 18 a–c
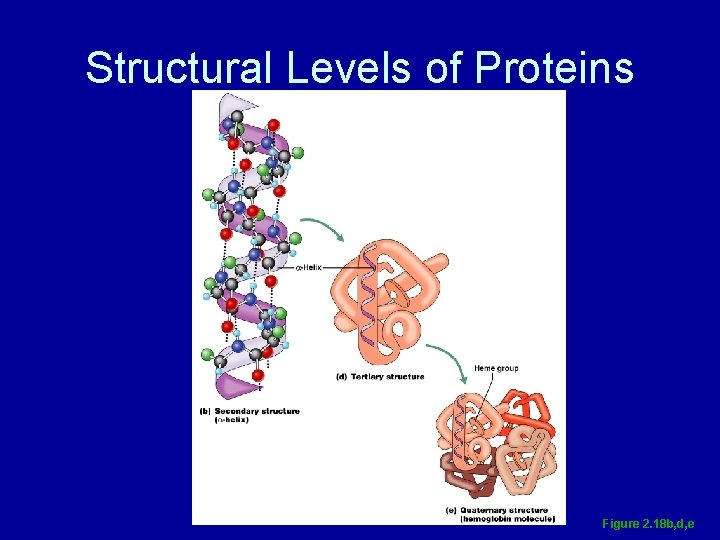
Structural Levels of Proteins Figure 2. 18 b, d, e

Fibrous and Globular Proteins • Fibrous proteins – Extended and strand-like proteins – Examples: keratin, elastin, collagen, and certain contractile fibers

Fibrous and Globular Proteins • Globular proteins – Compact, spherical proteins with tertiary and quaternary structures – Examples: antibodies, hormones, and enzymes

Protein Denuaturation • Reversible unfolding of proteins due to drops in p. H and/or increased temperature Figure 2. 19 a
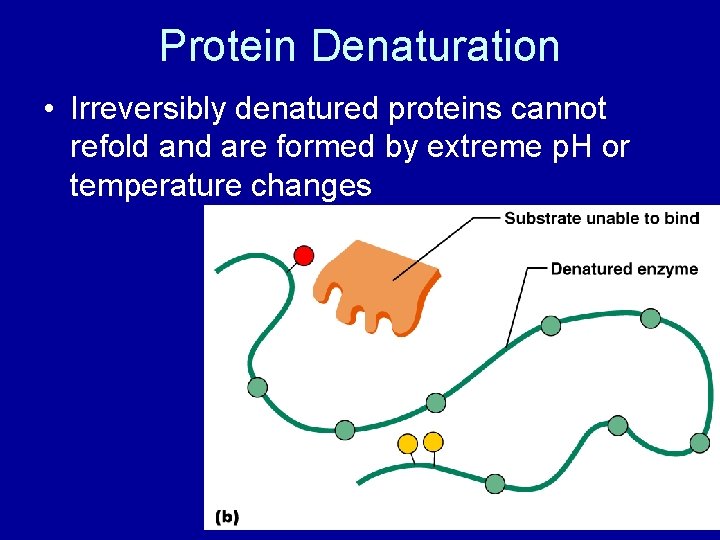
Protein Denaturation • Irreversibly denatured proteins cannot refold and are formed by extreme p. H or temperature changes Figure 2. 19 b

Molecular Chaperones (Chaperonins) • Help other proteins to achieve their functional three-dimensional shape • Maintain folding integrity • Assist in translocation of proteins across membranes • Promote the breakdown of damaged or denatured proteins

Characteristics of Enzymes • Most are globular proteins that act as biological catalysts • Holoenzymes consist of an apoenzyme (protein) and a cofactor (usually an ion) • Enzymes are chemically specific

Characteristics of Enzymes • Frequently named for the type of reaction they catalyze • Enzyme names usually end in -ase • Lower activation energy
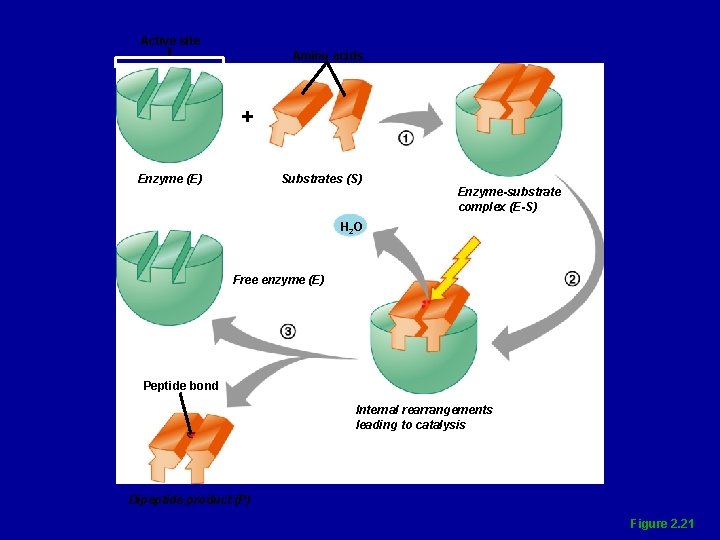
Active site Amino acids + Enzyme (E) Substrates (S) Enzyme-substrate complex (E-S) H 2 O Free enzyme (E) Peptide bond Internal rearrangements leading to catalysis Dipeptide product (P) Figure 2. 21

Nucleic Acids • Composed of carbon, oxygen, hydrogen, nitrogen, and phosphorus • Their structural unit, the nucleotide, is composed of N-containing base, a pentose sugar, and a phosphate group

Nucleic Acids • Five nitrogen bases contribute to nucleotide structure – adenine (A), guanine (G), cytosine (C), thymine (T), and uracil (U) • Two major classes – DNA and RNA

Deoxyribonucleic Acid (DNA) • Double-stranded helical molecule found in the nucleus of the cell • Replicates itself before the cell divides, ensuring genetic continuity • Provides instructions for protein synthesis
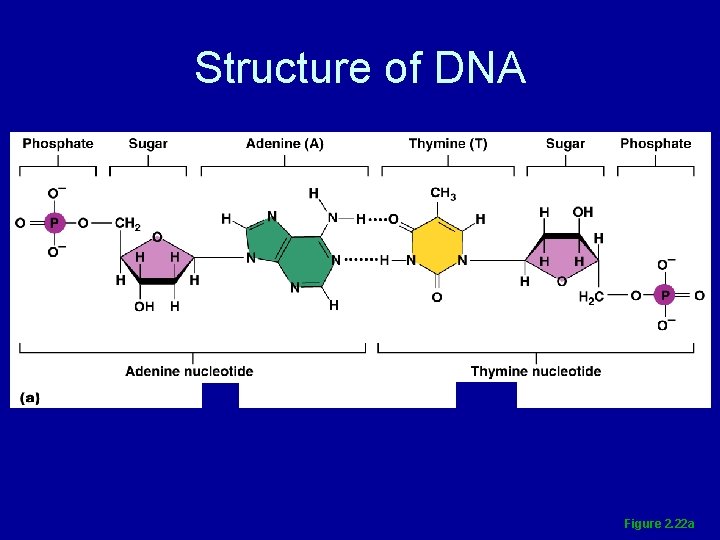
Structure of DNA Figure 2. 22 a
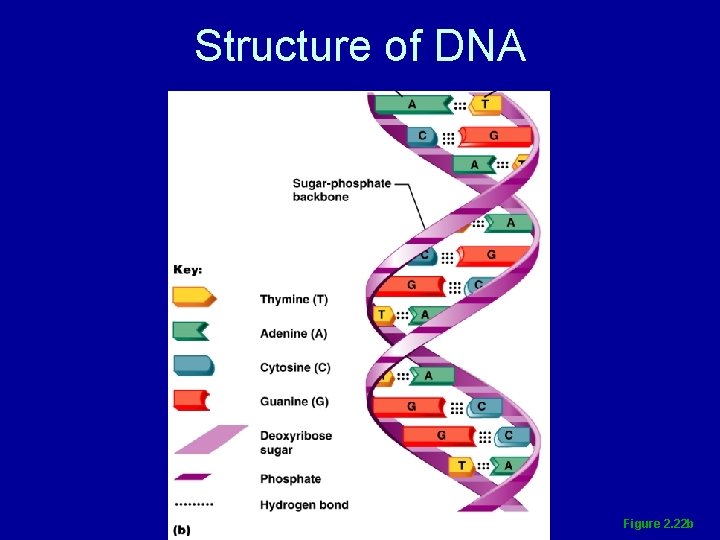
Structure of DNA Figure 2. 22 b

Ribonucleic Acid (RNA) • Single-stranded molecule found in both the nucleus and the cytoplasm of a cell • Uses the nitrogenous base uracil instead of thymine • Three varieties of RNA: messenger RNA, transfer RNA, and ribosomal RNA

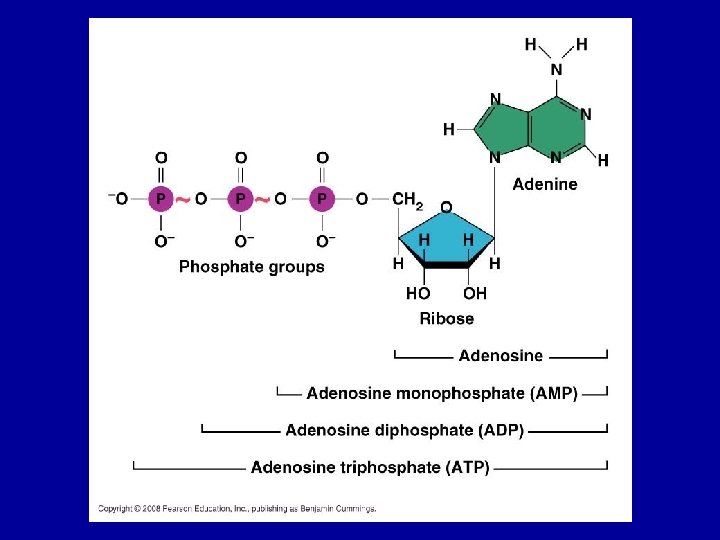
Adenosine Triphosphate (ATP) • Source of immediately usable energy for the cell • Adenine-containing RNA nucleotide with three phosphate groups

Hooray for the end of Chapter 2!!!!
- Slides: 51