Biochemistry Biochemistry is the application of chemistry to

Biochemistry

* Biochemistry is the application of chemistry to the study of biological processes at the cellular and molecular level. *Basic principles of biochemistry are common to all living organism *Biochemistry

*Structure and function of biological macromolecules *Molecular Genetics – How life is replicated. Regulation of protein synthesis *Metabolism processes. – anabolic and catabolic *Principle Areas of Biochemistry

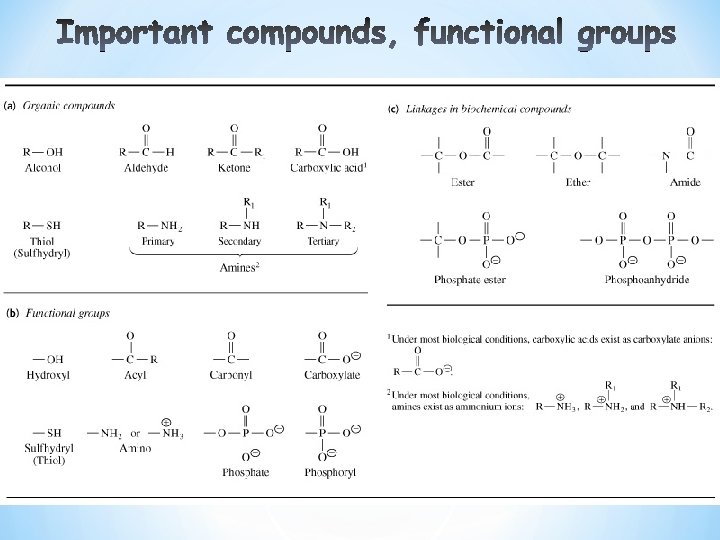
* * Just like cells are building blocks of tissues likewise molecules are building blocks of cells. * Animal and plant cells contain approximately 10, 000 kinds of molecules (bio-molecules) * Water constitutes 50 -95% of cells content by weight. * Ions like Na+, K+ and Ca+ may account for another 1% * Almost all other kinds of bio-molecules are organic (C, H, N, O, P, S) * Infinite variety of molecules contain C. * Most bio-molecules considered to be derived from hydrocarbons. * The chemical properties of organic bio-molecules are determined by their functional groups. Most bio-molecules have more than one.

Organization of Life
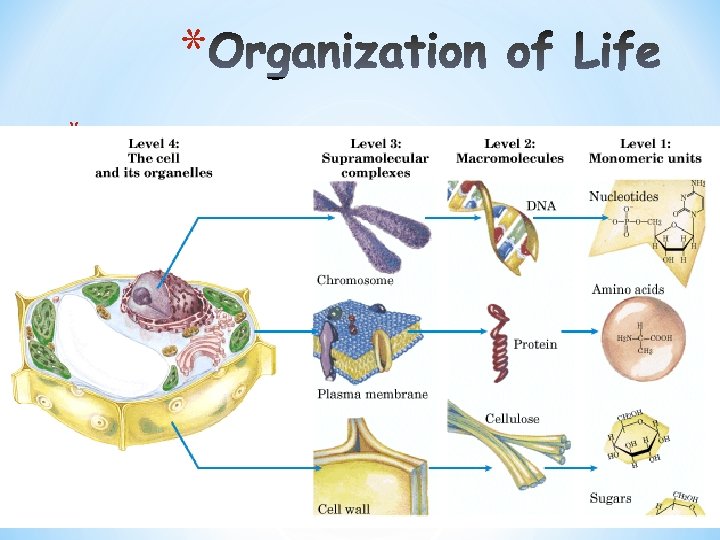
* *Elements *Simple organic compounds (monomers) *Macromolecules (polymers) *Supramolecular structures *Organelles *Cells *Tissues *Organisms N. Hamed BCH 1
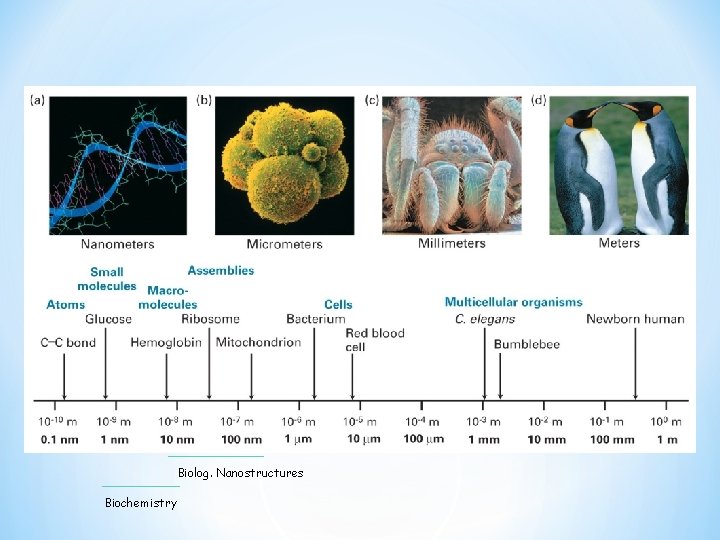
Biolog. Nanostructures Biochemistry

Many Important Biomolecules are Polymers lipids monomer polymer supramolecular structure proteins carbo nucleic acids
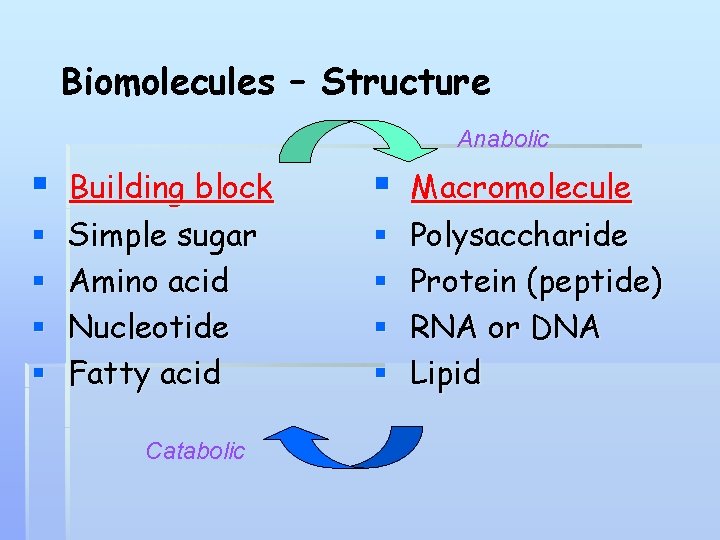
Biomolecules – Structure Anabolic § § § Building block Simple sugar Amino acid Nucleotide Fatty acid Catabolic § § § Macromolecule Polysaccharide Protein (peptide) RNA or DNA Lipid

Macromolecules
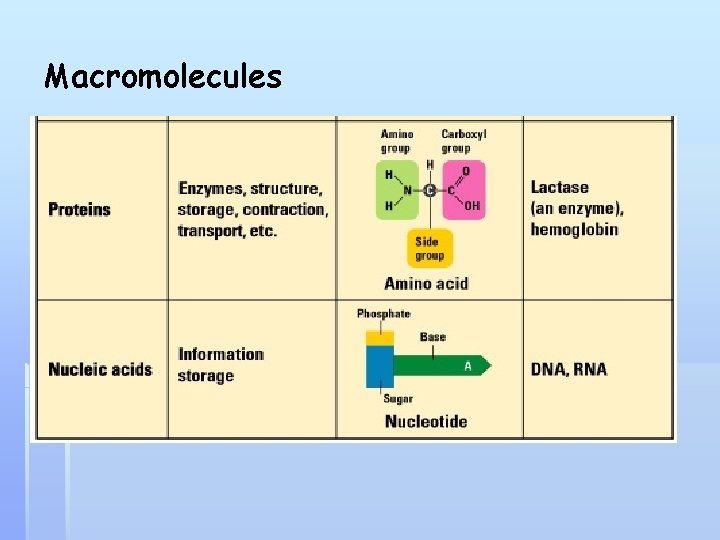
Macromolecules
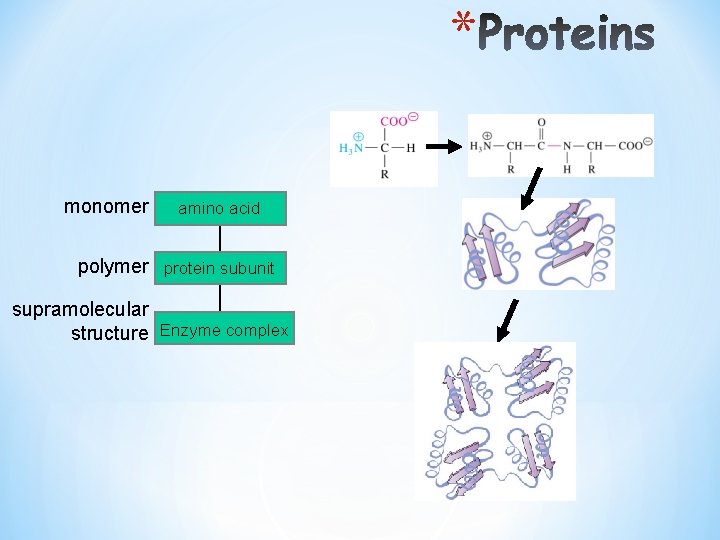
* monomer amino acid polymer protein subunit supramolecular structure Enzyme complex

Ø Amino acid Structure ØAmino acid Function Ø Amino acid chemical properties ØAmino * acid Classification

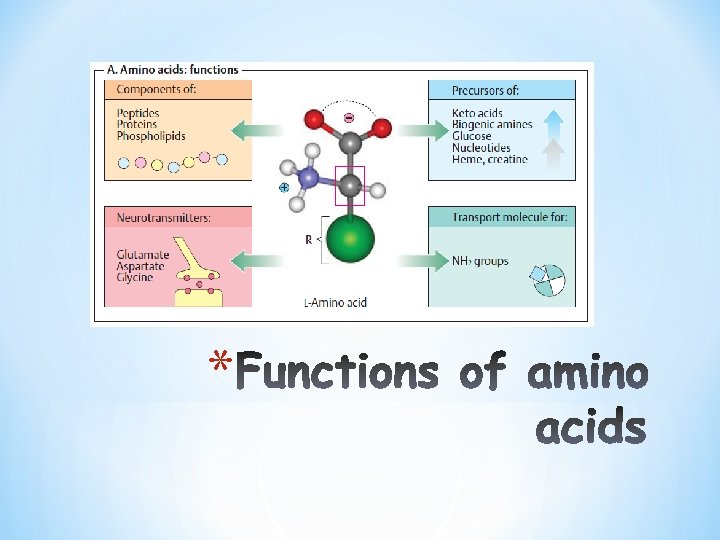
*Building blocks for Proteins and Peptides *How important are they? *proteins; enzymes, hormones direct and regulate metabolism in the body. *contractile proteins in muscle movement. *In bone Collagen *In the bloodstream hemoglobin, plasma albumin and immunoglobulin fight infectious bacteria and viruses. *20 amino-acids/ proteins/ coded for by DNA *What are Amino (are these the only amino acids in nature? >300 AA) Acids?
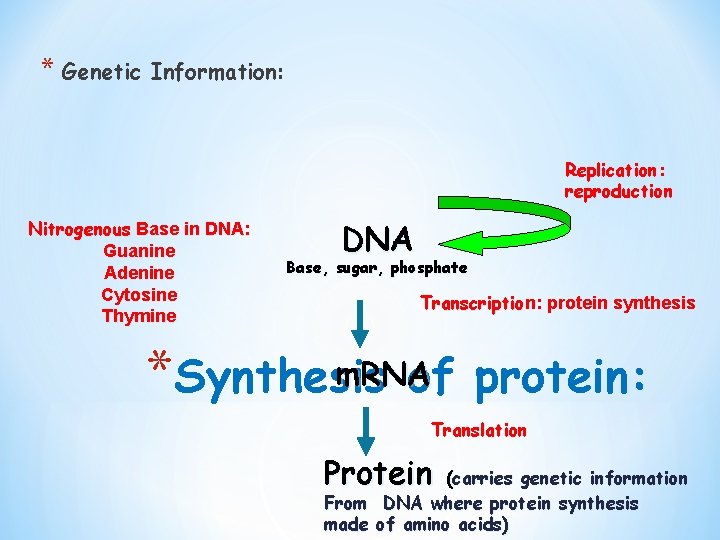
* Genetic Information: Replication: reproduction Nitrogenous Base in DNA: Guanine Adenine Cytosine Thymine DNA Base, sugar, phosphate Transcription: protein synthesis m. RNA *Synthesis of protein: Translation Protein (carries genetic information From DNA where protein synthesis made of amino acids)

The central concept of genetics involves the DNA-to-protein sequence (transcription and translation) n DNA has a sequence of bases that is transcribed into a sequence of bases in m. RNA. n Every 3 nitrogenous bases (G, A, C, T) make genetic codon that stands for a particular amino acid. Is it one codon/one amino acid? e. g. AAG = Lysine , CAG = Glutamine n

*Structural Function * Peptides, proteins (enzymes, plasma and tissue proteins) * Hormones as thyroxine. * Histamine (synthesised from histidine ) is the mediator of allergic reactions. *Neurotransmitters (glycine , glutamate) *Health and growth *

* *Amino Acid pool (present in body/cells) * Food * Synthesized from other metabolic compounds inside the body * Degradation of proteins inside the cell
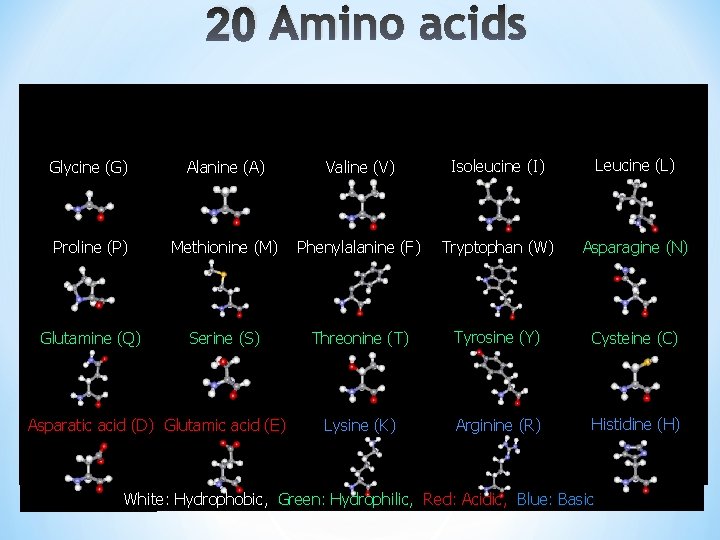
20 Amino acids Glycine (G) Alanine (A) Valine (V) Isoleucine (I) Leucine (L) Proline (P) Methionine (M) Phenylalanine (F) Tryptophan (W) Asparagine (N) Glutamine (Q) Serine (S) Threonine (T) Tyrosine (Y) Cysteine (C) Lysine (K) Arginine (R) Histidine (H) Asparatic acid (D) Glutamic acid (E) White: Hydrophobic, Green: Hydrophilic, Red: Acidic, Blue: Basic
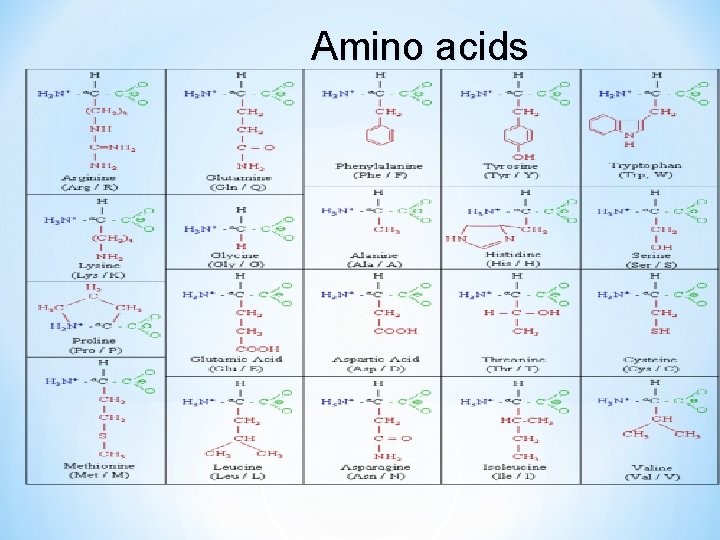
Amino acids

Three letter (examples) * Alanine, Ala * Glycine, Gly * Aspartate, Asp * Aspargine, Asn * Glutamate, Glu * Glutamine, Gln * Tyrosine, Tyr * Tryptophan, Trp One letter (examples) * Alanine, A * Glycine, G * Aspartate, D * Aspargine, N * Glutamate, E * Glutamine, Q * Tyrosine, Y * Tryptophan, W
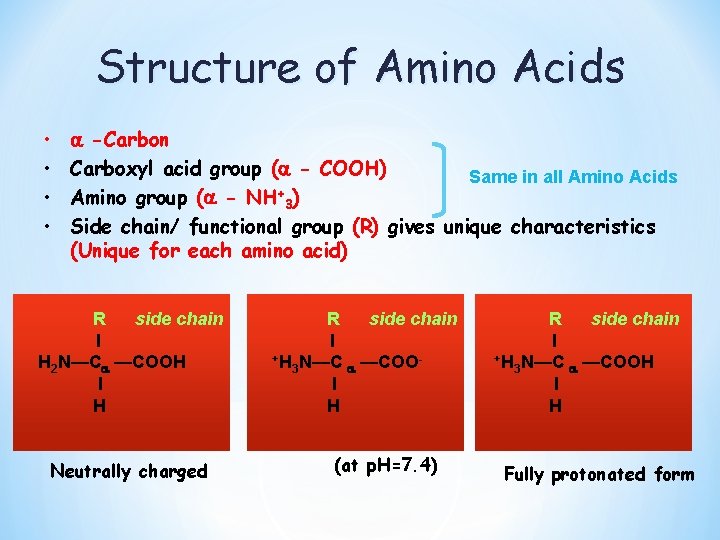
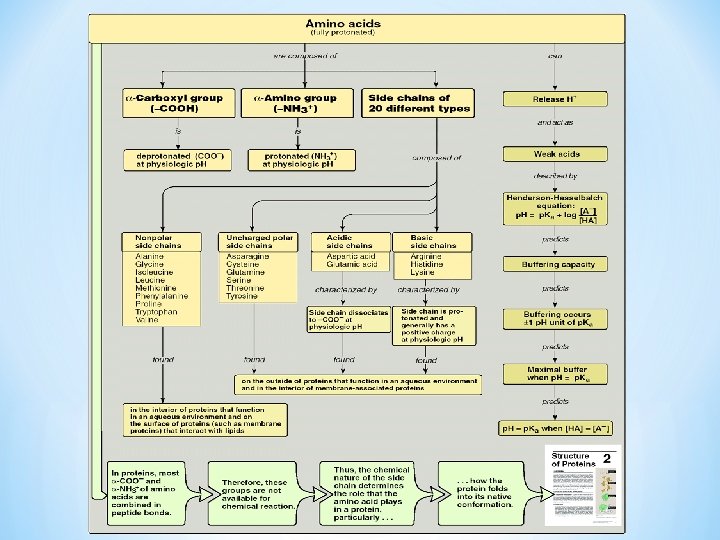
Structure of Amino Acids Aci • • -Carbon Carboxyl acid group ( - COOH) Same in all Amino Acids Amino group ( - NH+3) Side chain/ functional group (R) gives unique characteristics (Unique for each amino acid) R side chain I H 2 N—C —COOH I H Neutrally charged R side chain I +H N—C —COO 3 I H (at p. H=7. 4) R side chain I +H N—C —COOH 3 I H Fully protonated form
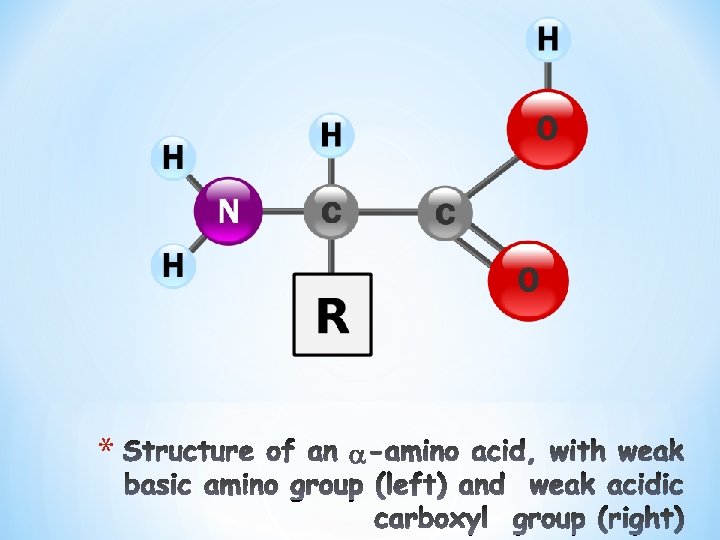

Carboxyl and amino groups • At physiologic p. H (p. H 7. 4), the carboxyl group is dissociated forming the : Ønegatively charged carboxylate ion (─COO- ) Øamino group is protonated (─NH 3+). • In proteins, all the carboxyl and amino groups are combined in peptide linkage and, available only for hydrogen bond formation.

* *Unique to each amino acid *Determine the role that the amino acid plays in a protein *Different amino acids have interesting properties because they have a variety of structural parts which result in different polarities and solubility.

*-COOH, -NH 2 so amino acids will give all reactions of carboxyl and amino group *Transamination *Decarboxylation *Deamination *Peptide bond *Reaction with Ninhydrin (Lab) blue color indicates the quantity of AA present *Absorption of light (Lab) *

1. Iso-electric point *Amino acids can exist as Zwitterions in solution, depending on the p. H of the medium. The p. H at which the molecule carries no net charge is known as iso-electric point. *

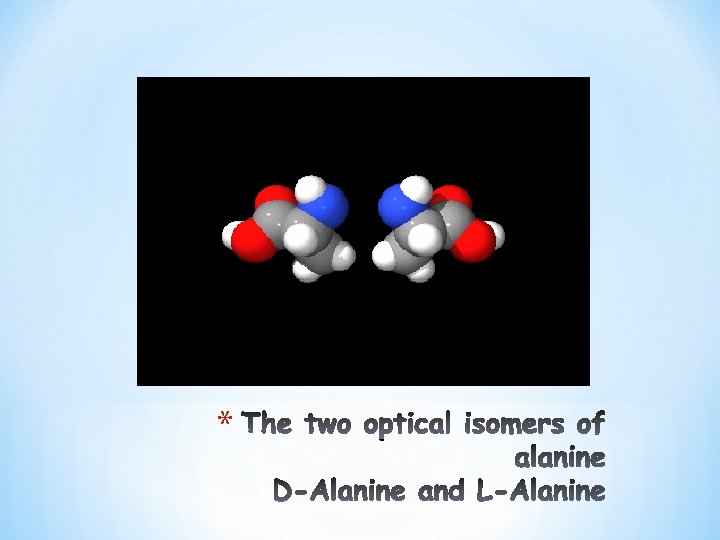
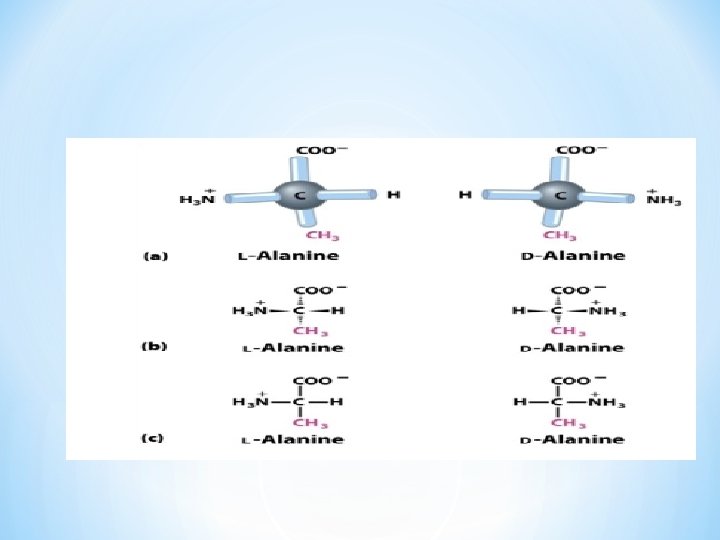
COOH I +H N—C — H 3 I R L-Form COOH I H—C —NH+3 I R D-Form • Amino Acids that have four different chemical groups can exist in two forms (D and L) • What if the R-group is an “H+”? Is there going to be an L and D form? (Glycine) No optical activity. • In proteins in mammals (all amino acids are in the L-form) • Are there any D-from amino acids? Yes, synthetics (antibiotics, in plant and bacterial walls) *

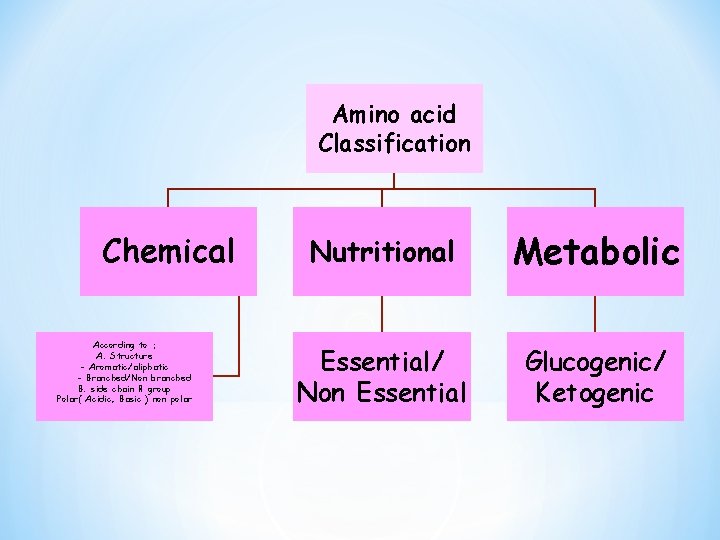
Amino acid Classification Chemical According to : A. Structure - Aromatic/aliphatic - Branched/Non branched B. side chain R group Polar( Acidic, Basic ) non polar Nutritional Metabolic Essential/ Non Essential Glucogenic/ Ketogenic

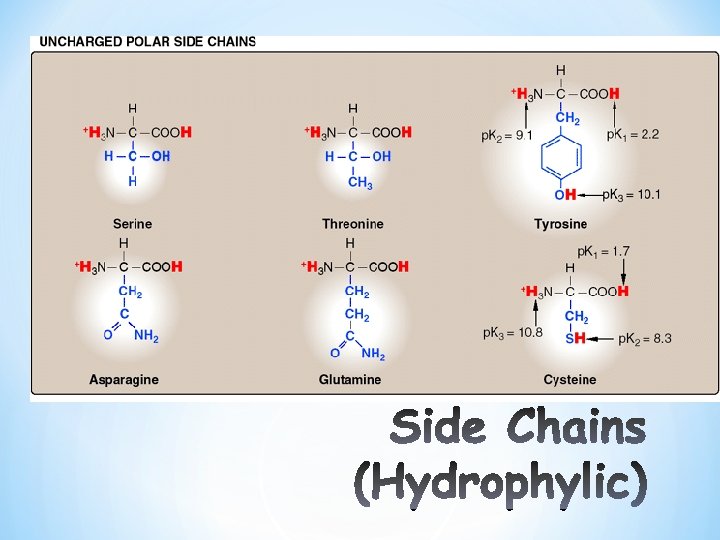
Classification of A. A according to Side chain Polar Non polar Neutral, uncharged Polar Acidic ( negatively charged) Polar Basic ( positively charged)

*Non Polar least soluble * R= H+, CH 3, alkyl, aromatic. ( 9 amino acids in this group) • Polar Uncharged more soluble O – R = –CH 2 OH, –CH 2 SH, C–NH 2, (polar groups with hydroxyl–O-, sulfur (6 amino acids in this group) -SH, Amide-N-) • Acidic/ Polar: – R= -COOH ( 2 amino acids in this group) • Basic/ Polar: – R= -NH 2 ( 3 amino acids in this group)

*These are more soluble in water than the Non polar( Hydrophilic). * have an uneven distribution of electrons, such as acids and bases. *Side chains which have various functional groups such as Sulfur, amides, alcohols will impart a more polar character to the amino acid. *

* • Disulfide bonds: – Cysteine (-SH group) – 2 Cysteine (2 –SH groups, become oxidized to give SS disulfied bond) H I – Play important role in protein +H N—C —COOH 3 stability I H — C—H I s Disulfide bond/bridge) I H—C—H I +H N—C —COOH 3 I H

*Contains more than one –NH 2 group. *Proton acceptors. *(+ve) at physiological p. H. *
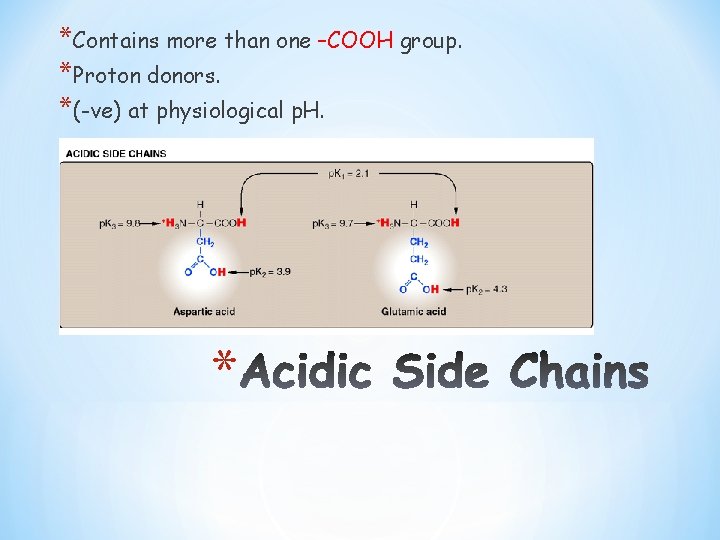
*Contains more than one –COOH group. *Proton donors. *(-ve) at physiological p. H. *

* Each of these A. A. has a nonpolar side chain that does not bind or give off protons or participate in hydrogen or ionic bonds. *have an even distribution of electrons. *The side chains can be thought of as "oily" or lipid-like, a property promotion of hydrophobic interactions. *

*One of the 20 AA called proline is not an amino acid *It is an Imino acid as it contains imino group ( -NH). *It has a secondary amino group. *Fibrous structures of collagen. *

*Glucogenic: gives * Essential glucose * Non essential *Ketogenic: gives ketone bodies *

• Can not be synthesized by humans (have to get them from diet) • Their deficiencies affect growth, health, protein synthesis • Isoleucine • Threonine • 10 amino acids: • Lysine • Phenylalanine • Methionine • Leucine • Valine * • Tryptophan • Arginine* • Histidine*

• 10 amino acids that can be formed from aketo acids by transamination and subsequent reactions • • • Alanine Glycine Serine Proline Glutamate * • • • Glutamine Aspartate Asparagine Cysteine (from Met*) Tyrosine (from Phe*)
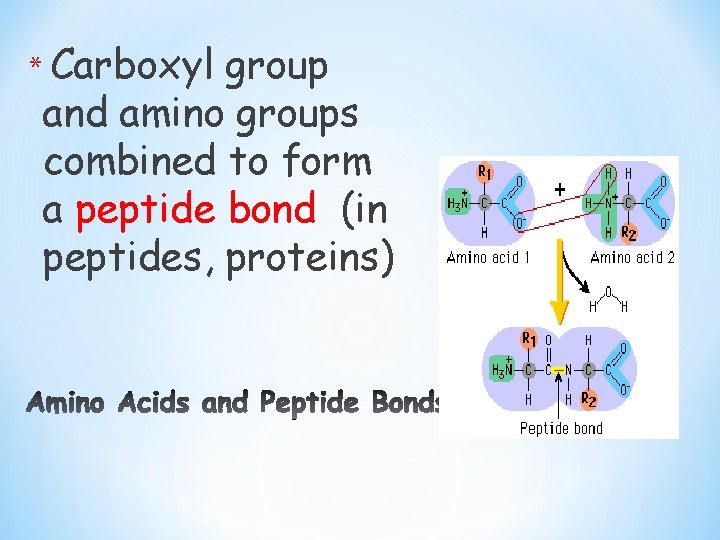
* Carboxyl group and amino groups combined to form a peptide bond (in peptides, proteins)
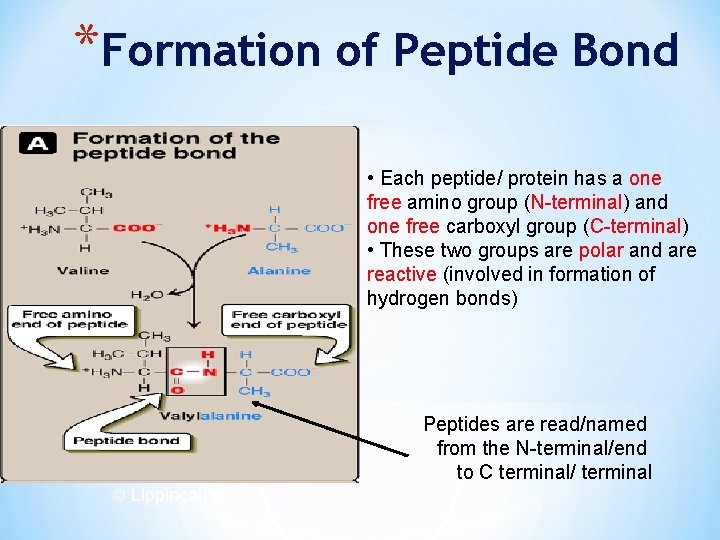
*Formation of Peptide Bond • Each peptide/ protein has a one free amino group (N-terminal) and one free carboxyl group (C-terminal) • These two groups are polar and are reactive (involved in formation of hydrogen bonds) Lippincott’s Peptides are read/named from the N-terminal/end to C terminal/ terminal
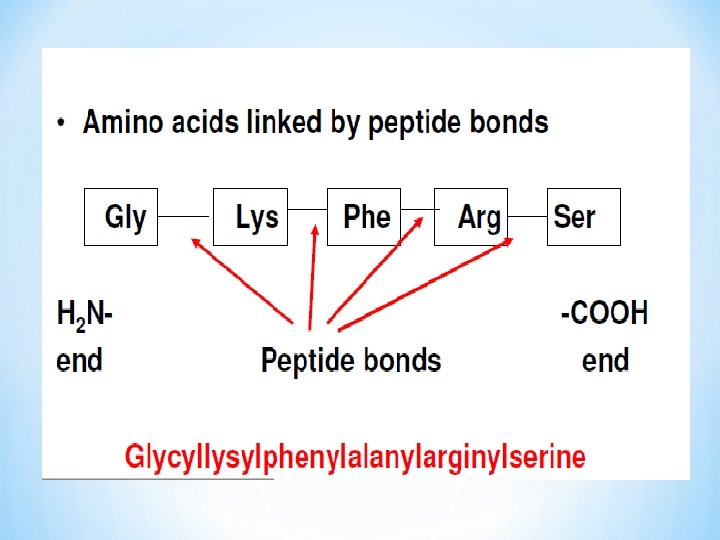

I. Amino terminal end (N-terminal), usually the n-terminal amino acid is written on the left hand side. II. Carboxy terminal end (C- terminal) *
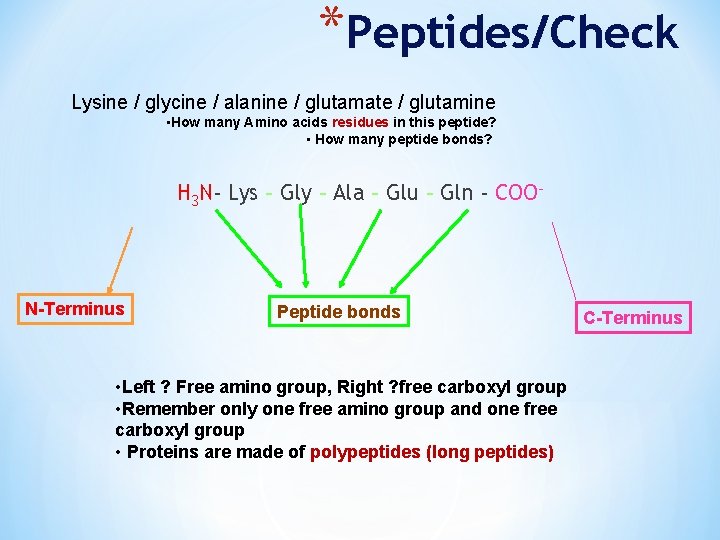
*Peptides/Check Lysine / glycine / alanine / glutamate / glutamine • How many Amino acids residues in this peptide? • How many peptide bonds? H 3 N- Lys – Gly – Ala – Glu – Gln - COO- N-Terminus Peptide bonds • Left ? Free amino group, Right ? free carboxyl group • Remember only one free amino group and one free carboxyl group • Proteins are made of polypeptides (long peptides) C-Terminus
- Slides: 52