Biochemistry Bioc 212 Acknowledgment Addisa Ababa University Haramaya

Biochemistry Bioc 212

Acknowledgment • • Addisa Ababa University Haramaya University Hawassa University Jimma University of Gondar American Society for Clinical Pathology Center for Disease Control and Prevention-Ethiopia

Course title: Biochemistry • • Course Number: Credit Hours: Lecture: Prerequisite: Bioc 212 3 3 hours/week None

Course information • • • Teaching Methods: Lecture Demonstration Presentation and group discussion Assessment Method: Assignment & Quiz (10%) Mid examination (30%) Final examination (60%) Time Allocation: 48 contact hours Grading: Fixed scale

Course objectives At the end of the course the students should be able to: • Describe the classification, properties and function of the four major biomolecules • Gain basic insight into the dynamic events of metabolism, so characteristic of every living cell • Identify the common tests used for carbohydrates and protein • Discuss the role of enzymology in pharmacology and clinical diagnosis • Understand the storage and expression of genetic information • Discuss vitamins and minerals

Course outline 1. 0. Introduction 1. 1. Definition of biochemistry 1. 2. The scope of biochemistry 1. 3. Significance of biochemistry in medicine 1. 4. The cell and major chemical constituents of cells (water, acid and base) 1. 5. Water as a solvent and as a medium for biochemical reactions

Outline cont’d 1. 6. Chemical properties and structure of water 1. 7. Water and thermal regulation 1. 8. Electrolyte 1. 9. Osmolality and water movement 2. 0. Acid and base

Outline cont’d 2. 0. Carbohydrates 2. 1. Introduction and classification-mono, di- and polysaccharides 2. 2. Chemistry and properties and their test 2. 3. Carbohydrate metabolism 2. 4. Hormonal regulations of carbohydrate metabolism 2. 5. Clinical significances in carbohydrate metabolism

Outline cont’d 2. 6. Glycolysis 2. 7. Rapaport luebering shunt 2. 8. Gluconeogenesis 2. 9. Glycogen metabolism 2. 10. Pentose monophosphate pathway 2. 11. TCA cycle

Outline cont’d 3. 0. Proteins 3. 1. Classification, naming, Structure and function of amino acids 3. 2. Structure of proteins (conformation) 3. 3. Functions of proteins 3. 4. Myoglobin and Hemoglobin Structure

Outline cont’d 3. 5. Protein metabolism 3. 6. Essential and non-essential aminoacids 3. 7. Catabolism aminoacids 3. 8. Transamination and oxidative deamination 3. 9. Urea cycle 3. 10. Catabolism of aminoacid carbon skeleton 3. 11. Inborn errors of aminoacid metabolism 3. 12. Heme Metabolism

Outline cont’d 4. 0. Enzymes 4. 1. Nomenclature 4. 2. Properties of enzymes 4. 3. Theory of enzyme catalysis 4. 4. Factors affecting the velocity of enzyme-catalyzed reactions 4. 5. Enzyme inhibitors 4. 6. Enzymes in clinical diagnosis and pharmacology

Outline cont’d 5. 0. Vitamins 5. 1. Classification-water soluble and fat soluble 5. 2. Sources, physiological functions and deficiency of water soluble Vitamins- B vitamins and vitamin C 5. 3. Sources, physiological functions and deficiency of fat soluble vitamins- vitamin A, vitamin D, vitamin K and vitamin E

Outline cont’d 6. Lipids 6. 1. Classification 6. 2. Structure and properties of fatty acids, phospholipids, triacylglycerol, Cholesterol, lipoproteins and ketone bodies 6. 3. Lipid metabolism

Outline cont’d 6. 4. Lipid transport 6. 5. Lipoproteins 6. 6. Clinical correlates 6. 7. Ketosis 6. 8. Disorders of plasma lipoprotein (hypercholeste- rolenuria and atherosclerosis)

outline cont’d 7. 0. Oxidative phosphorylation and electron transport chain Oxidation and Reduction 7. 1. The Electron Transport Chain 7. 2. Connections and Regulation 7. 3. Ratios of phosphate to oxygen 7. 4. Uncouplers 7. 5. Inhibitors

Outline cont’d 8. 0. Nucleic acids 8. 1. Classification and chemistry of nucleic acids-RNA and DNA 8. 2. Metabolism of nucleotides 8. 3. Clinical significance (Hyperuricemia and gout and Orotic aciduria 8. 4. DNA replication 8. 5. Transcription 8. 6. Translation

1. 0. Introduction

Out line 1. 0. Introduction 1. 1. Definition of biochemistry 1. 2. The scope of biochemistry 1. 3. The cell 1. 4. Water 1. 5. Water as a solvent and as a medium for biochemical reactions

Outline cont’d 1. 6. Chemical properties and structure of water 1. 7. Water and thermal regulation 1. 8. Electrolyte 1. 9. Osmolality and water movement 2. 0. Acid and base

Chapter learning objective • At the end of this chapter the students should be able to: • Explain the definition , the scope and significance of biochemistry in medicine • Discuss the major chemical constituents of cells

Learning objective cont’d · Discuss the chemical properties and structure of water · Explain about water and thermal regulation · Discuss electrolytes · Discuss osmolality and water movement · Discuss acid and base

1. 0. Introduction • 1. 1. Definition of biochemistry • Biochemistry is defined as the science concerned with the chemical constituents of living cells and with the reactions and processes they undergo. • Biochemistry encompasses large areas of cell biology, of molecular biology, and of molecular genetics.

• 1. 2. Scope of biochemistry • Biochemistry provides important insights and practical applications in medicine, agriculture, nutrition, and industry • Medicine depends on understanding the roles and interactions of the enormous number of different chemicals enabling our bodies to function.

• 1. 3. The cell • Cells Are the Structural and Functional Units of all Living Organisms • The plasma membrane defines the periphery of the cell, separating its contents from the surroundings. • It is composed of lipid and protein molecules that form a thin, tough, pliable, hydrophobic barrier around the cell.

• Transport proteins in the plasma membrane allow the passage of certain ions and molecules; • Receptor proteins transmit signals into the cell; and membrane enzymes participate in some reaction pathways. • Because the individual lipids and proteins of the plasma membrane are not covalently linked, the entire structure is remarkably flexible, allowing changes in the shape and size of the cell.
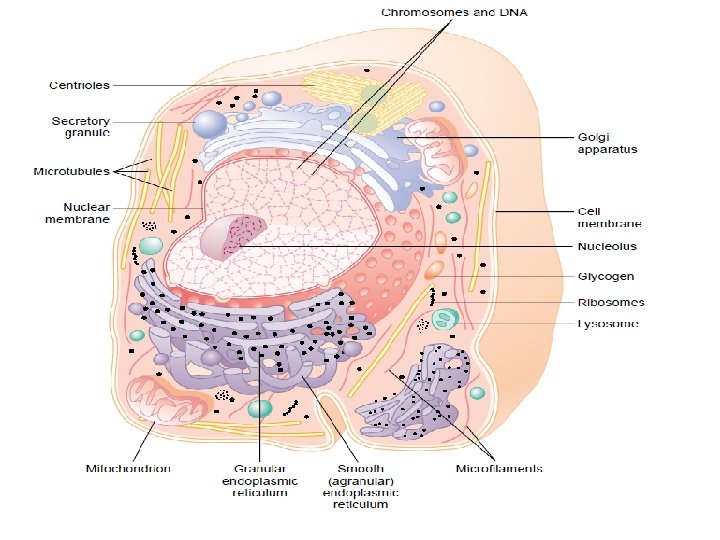

• Cytoplasm is composed of an aqueous solution, the cytosol, and a variety of suspended particles with specific functions. • The cytosol is a highly concentrated solution containing enzymes and the RNA molecules that encode them. • Ribosomes are small particles (composed of protein and RNA molecules) that are the sites of protein synthesis.

• 1. 4. Water • Water is the solvent of life. It bathes the cells, dissolves and transports compounds in the blood, and provides a medium for movement of molecules into and throughout cellular compartments. • Separates charged molecules, dissipates heat, and participates in chemical reactions. • The body maintains a nearly constant amount of water that is approximately 60% of our body weight

• Approximately 40% of the total body water is intracellular and 60% extracellular. • The extracellular water includes the fluid in plasma (blood after the cells have been removed) and interstitial water (the fluid in the tissue spaces, lying between cells).

• Transcellular water is a small, specialized portion of extracellular water that includes gastrointestinal secretions, urine, sweat, and fluid that has leaked through capillary walls because of such processes as increased hydrostatic pressure or inflammation.

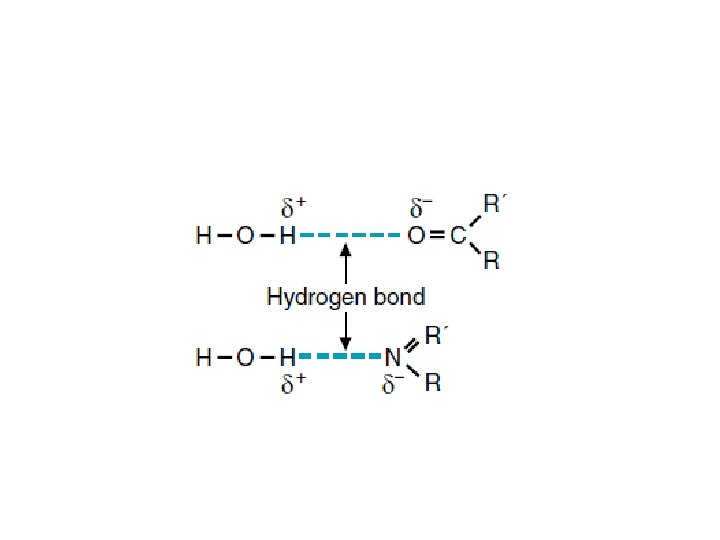
• 1. 5. Water as a solvent • Polar organic molecules and inorganic salts can readily dissolve in water because water also forms hydrogen bonds and electrostatic interactions with these molecules. • Organic molecules containing a high proportion of electronegative atoms (generally oxygen or nitrogen) are soluble in water because these atoms participate in hydrogen bonding with water molecules.

• 1. 6. Electrolytes • Both extracellular fluid (ECF) and intracellular fluid (ICF) contain electrolytes, a general term applied to bicarbonate and inorganic anions and cations. • The electrolytes are unevenly distributed between compartments; Na and Cl are the major electrolytes in the ECF (plasma and interstitial fluid).

• 1. 7. Acids and bases • Acids are compounds that donate a hydrogen ion (H) to a solution, and bases are compounds (such as the OH ion) that accept hydrogen ions. • Water itself dissociates to a slight extent, generating hydrogen ions (H), which are also called protons, and hydroxide ions (OH).

• The extent of dissociation by water molecules into H and OH is very slight, and the hydrogen ion concentration of pure water is only 0. 0000001 M, or 10 -7 mol/L. • The concentration of hydrogen ions in a solution is usually denoted by the term p. H, which is the negative log 10 of the hydrogen ion concentration expressed in mol/L. Therefore, the p. H of pure water is 7.

• The dissociation constant for water, Kd, expresses the relationship between the hydrogen ion concentration [H], the hydroxide ion concentration [OH]. • Because Kw, the product of [H] and [OH], is always constant, a decrease of [H] must be accompanied by a proportionate increase of [OH].


• The strength of weak acids is expressed by p. Ka, the negative log of the acid dissociation constant. • Strong acids have low p. Ka values and weak acids have high p. Ka values. • Buffers resist a change in p. H when protons are produced or consumed. Maximum buffering capacity occurs ± 1 p. H unit on either side of p. Ka. • Physiologic buffers include bicarbonate, orthophosphate, and proteins.

• The tendency of the acid (HA) to dissociate and donate a hydrogen ion to solution is denoted by its Ka, the equilibrium constant for dissociation of a weak acid. • The higher the Ka, the greater is the tendency to dissociate a proton.

Review questions • Describes a universal property of buffers? • Calculate the PH of the following solutions A. 0. 02 NHCl B. 0. 0003 M Na. OH

Summary • Water forms hydrogen-bonded clusters with itself and with other proton donors or acceptors. Hydrogen bonds account for the surface tension, viscosity, liquid state at room temperature, and solvent power of water. • Macromolecules exchange internal surface hydrogen bonds for hydrogen bonds to water. Entropic forces dictate that macromolecules expose polar regions to an aqueous interface and bury nonpolar regions. • Salt bonds, hydrophobic interactions, and van der Waals forces participate in maintaining molecular structure.

• Alberts B, Johnson A, Lewis J, et al: Molecular Biology of the Cell. New York: Garland Science, 2002. • Bonifacino JS, Glick BS: The mechanisms of vesicle budding and fusion. Cell 116: 153, 2004. • Calakos N, Scheller RH: Synaptic vesicle biogenesis, docking, and fusion: a molecular description. Physiol Rev 76: 1, 1996. • Danial NN, Korsmeyer SJ: Cell death: critical control points. Cell 116: 205, 2004.
- Slides: 43