BIOCHEMISTRY ATOMS ELEMENTS COMPOUNDS BONDING ATOM Smallest part

BIOCHEMISTRY ATOMS ELEMENTS COMPOUNDS BONDING
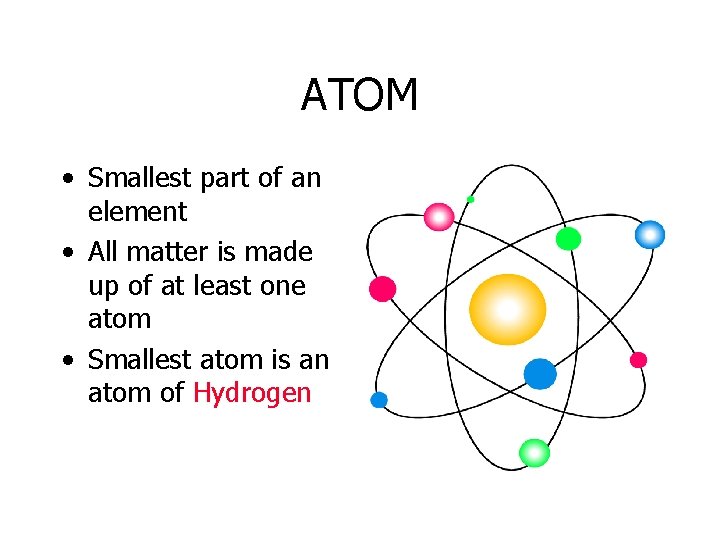
ATOM • Smallest part of an element • All matter is made up of at least one atom • Smallest atom is an atom of Hydrogen

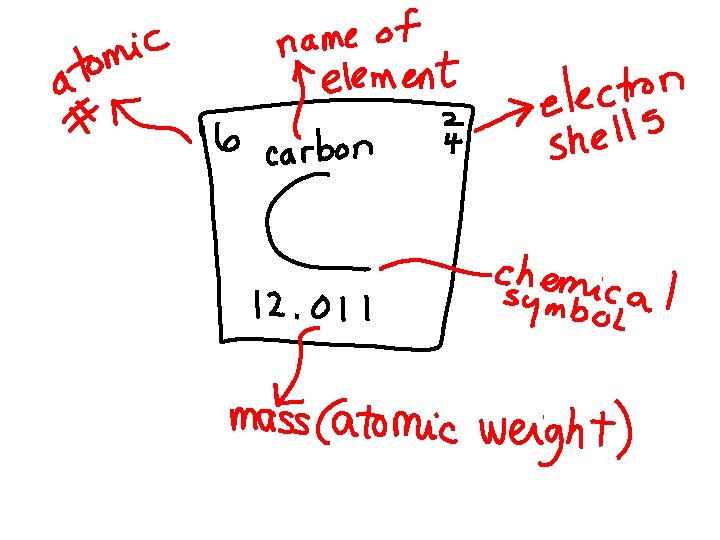
ELEMENT • There are 118 elements • They are the simplest of substances • Elements cannot be broken down into smaller substances

Elements you must know!!! • • • C – carbon O – oxygen H – hydrogen Na – sodium Cl – chlorine Mg – magnesium I – iodine Cu – copper K – potassium Co – cobalt He – helium S - sulfur N - nitrogen P - phosphorus Zn - zinc Ca - calcium Fe – iron Mn - manganese B - boron Cr – chromium F – fluorine Tl - Thallium

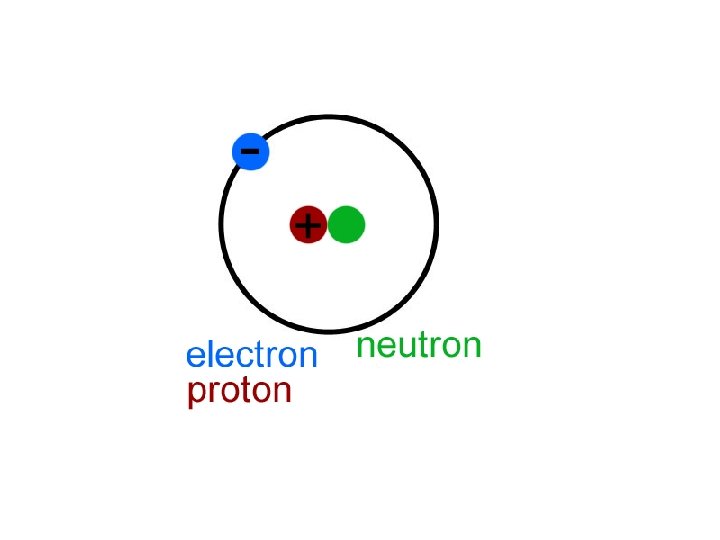
MODEL OF AN ATOM • Central Nucleus: • MODEL OF HELIUM: • protons = + charge • Neutron= neutral (no charge) ___________ • Electrons have a negative charge (they float in orbitals around 2 P the nucleus) 2 N 2 e

Protons and Neutrons • PROTONS: • The # of protons tells you what element you have • Atomic # = # of protons • PROTONS have a positive charge + • NEUTRONS • Located in the nucleus • Have no charge

CHARACTERISTICS OF PROTONS, NEUTRONS, ELECTRONS MASS PROTONS 1 NEUTRONS 1 ELECTRONS 0 CHARGE + no charge __ LOCATION NUCLEUS ORBITALS AROUND NUCLEUS


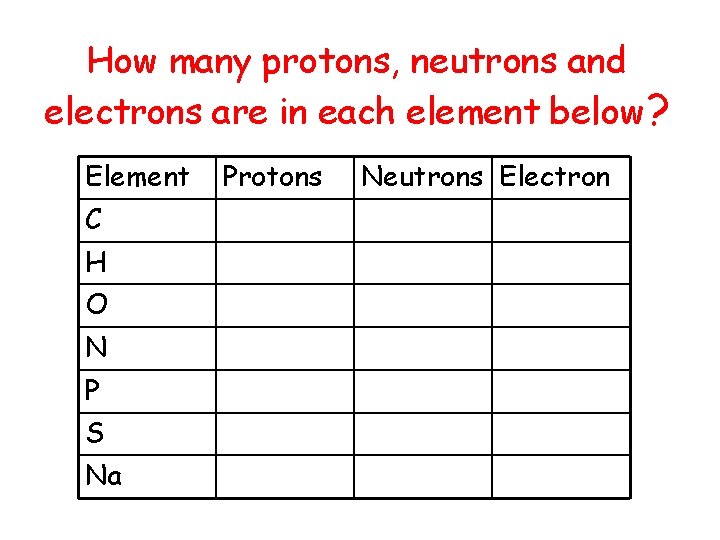
How many protons, neutrons and electrons are in each element below? Element C H O N P S Na Protons Neutrons Electron

Valence Electrons • The outermost electrons of an atom, which are important in determining how the atom reacts chemically with other atoms. • Atoms with a complete shell of valence electrons are more stable (octet rule!) • Atoms with one or two valence electrons more than a closed shell are highly reactive because the extra electrons are easily removed to form positive ions.

Electrons of Phosphorus

MOLECULE • Two or more atoms that are joined by a bond • A molecule of an element is the way the element usually exists in nature • H-H is a molecule of hydrogen (usually exists in pairs) • H-O-H is a molecule of water
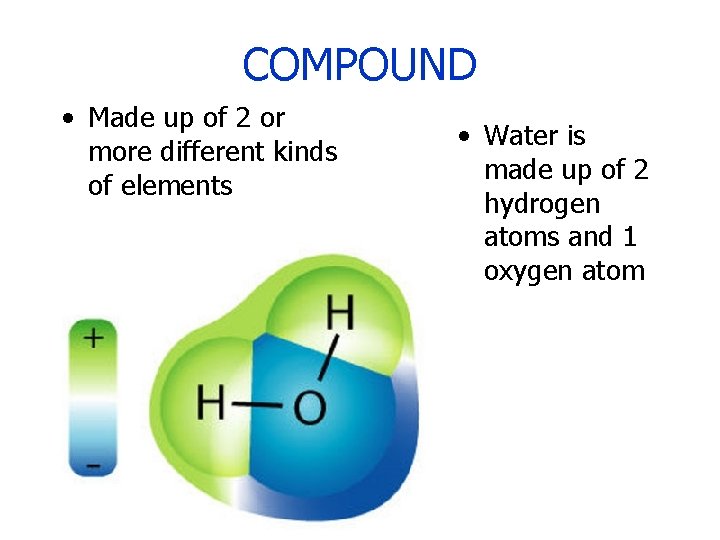
COMPOUND • Made up of 2 or more different kinds of elements • Water is made up of 2 hydrogen atoms and 1 oxygen atom

MIXTURE • Two or more elements or compounds that are not chemically combined. • Mixtures can be separated by physical means, such as filtering or boiling
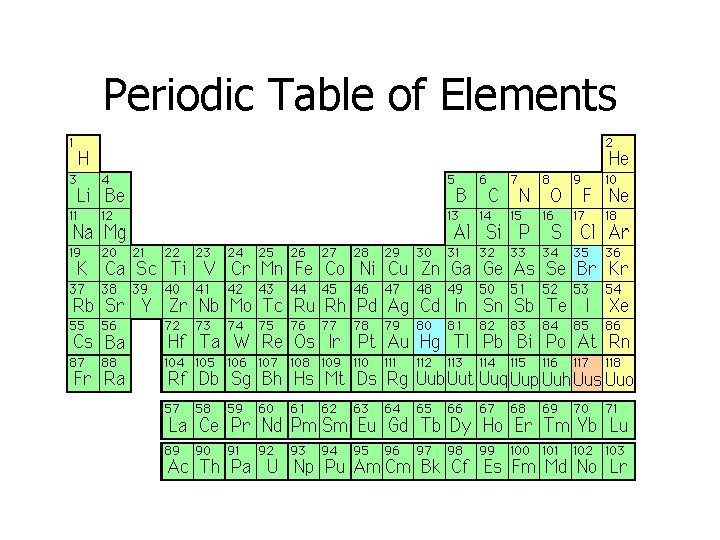
Periodic Table of Elements

ISOTOPES • Have the same atomic number (same # of protons) • Have a different atomic mass (different # of neutrons)

Radioactive Isotopes • Provides diagnostic information about the functioning of a person's specific organs. • Used as radiation therapy (treatment) for some medical conditions, especially cancer, (using radiation to weaken or destroy particular targeted cells). • Used for nuclear medicine procedures so the demand for radioisotopes is increasing rapidly.

RADIOACTIVE ISOTOPES: Examples used in Medicine today • Thallium-201 is a radioisotope used to determine whether a person has heart disease (caused by narrowing of the arteries to the heart • Iodine-131 is used to measure thyroid activity • Cobalt-60 is used for radiation in order to shrink cancer tumors
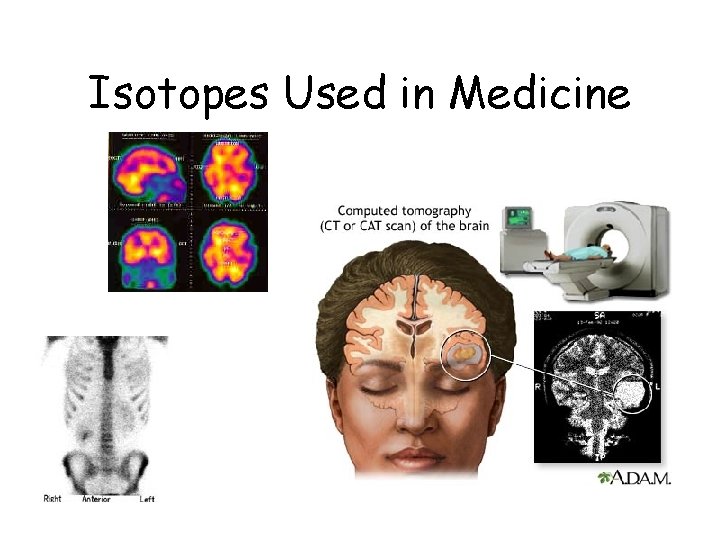
Isotopes Used in Medicine
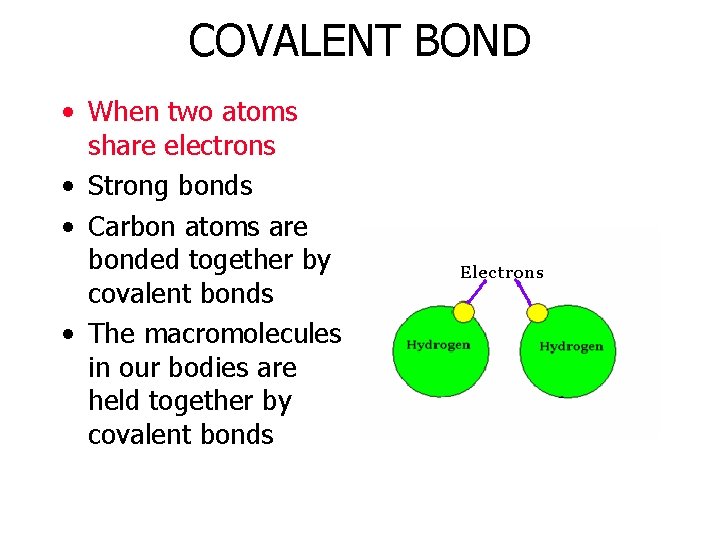
COVALENT BOND • When two atoms share electrons • Strong bonds • Carbon atoms are bonded together by covalent bonds • The macromolecules in our bodies are held together by covalent bonds

IONIC BOND • When an atom loses an electron to another atom, it gains a + charge • The atom that gained the electrons now has a - charge • + AND - ions attract each other (ionic bond) GIVE ME YOUR ELECTRON! IONIC BOND BECOMES POSITIVE BECOMES NEGATIVE
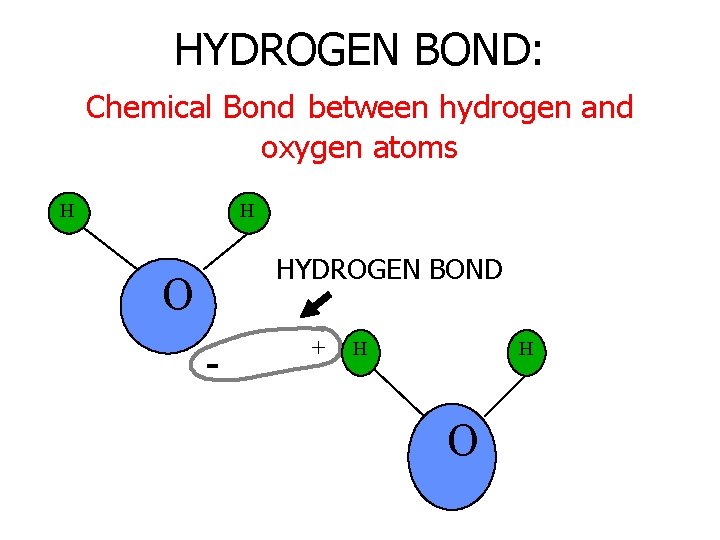
HYDROGEN BOND: Chemical Bond between hydrogen and oxygen atoms H H HYDROGEN BOND O - + H H O

PROPERTIES OF WATER • THE MOST ABUNDANT INORGANIC COMPOUND IN LIVING THINGS! • Universal Solvent: most substances dissolve in water • Water’s properties are due to its hydrogen bonding • Stable compound: Helps our cells to maintains HOMEOSTASIS • Water has a high specific heat: It takes much energy to change the temperature of water


Surface Tension of Water • Water is adhesive & elastic • Tends to aggregate in drops • This allows water to stick to the sides of vertical structures despite gravity’s downward pull

Surface Tension of Water


Water droplets forming from adhesive bonding:

Capillary Action of Water: how water moves up plant roots and through our tiny blood vessel

p. H • Measures the acidity or alkalinity of solution • Ranges from 1 to 14 • p. H levels from 1 to 6 are acids • p. H levels from 8 to 14 are bases • In pure water @ 25º C, the p. H is 7, which is neutral

p. H of pure water = 7

Some human Ph values: • p. H of blood is 7. 4 • Saliva has a p. H between 6 and 7. 4 • Urine is slightly acid in the morning, (p. H 6. 5 - 7. 0) generally becoming more alkaline (p. H = 7. 5 - 8. 0) by evening

So why all the fuss about water? ? ? 1. Water is a chemical essential to all known forms of life 2. It is a very important solvent, capable of dissolving salts, sugars, acids, alkalis, gases, and many other organic molecules 3. About 55 to 78% of our bodies is water

Organic vs. Inorganic Molecules: • ORGANIC: • Compounds contain the elements Carbon and Hydrogen Examples: • METHANE- CH 4 • Glucose: C 6 H 12 O 6 • INORGANIC: • Compounds do not have the carbon hydrogen combination • They may contain C or H, but not both • EXAMPLES: • Carbon dioxide CO 2 • WATER: H 2 O • Hydrchloric Acid: HCl

What is a Macromolecule? • Polymer = A molecule made up of a large number of elements • Also referred to as a Macromolecule • Individual parts of macromolecules are known as monomers • Most macromolecules are found in living organisms (like us)!!!

4 important macromolecules: 1. 2. 3. 4. Carbohydrates Lipids Proteins Nucleic Acids

• These macromolecules are synthesized (joined together) by monomers during a dehydration reaction • In each dehydration reaction, a molecule of water is lost • The macromolecules are broken apart into their monomers by a hydrolysis reaction, where a molecule of water is gained
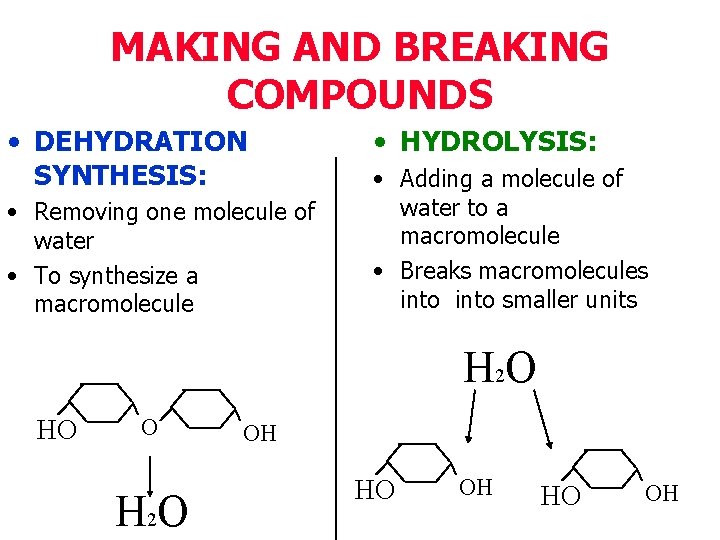
MAKING AND BREAKING COMPOUNDS • DEHYDRATION SYNTHESIS: • Removing one molecule of water • To synthesize a macromolecule • HYDROLYSIS: • Adding a molecule of water to a macromolecule • Breaks macromolecules into smaller units H 2 O HO O H 2 O OH HO OH

Dehydration Synthesis • How small molecules (monomers) are combined to make molecules large (polymers) • water is lost (dehydrate) • How macromolecules in our bodies are synthesized (built/combined)

Hydrolysis • How large molecules (polymers) are broken down to smaller molecules (monomers) • water is added • How our bodies digest (break down) the macromolecules that we eat

CARBOHYDRATES: ENERGY COMPOUNDS
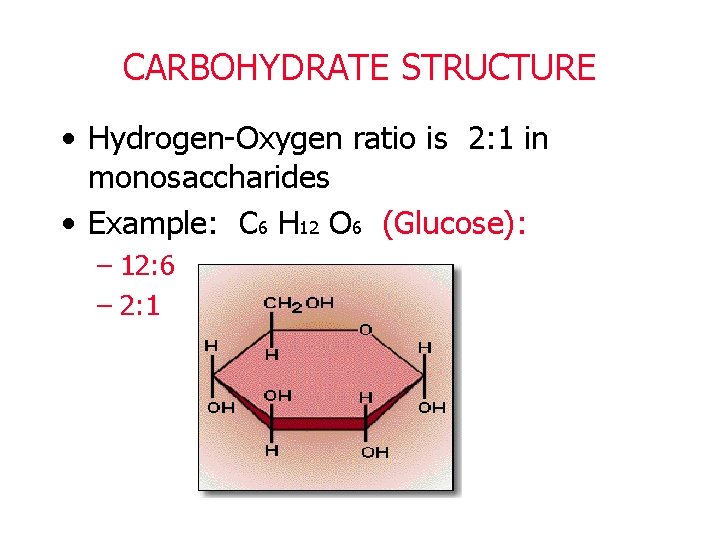
CARBOHYDRATE STRUCTURE • Hydrogen-Oxygen ratio is 2: 1 in monosaccharides • Example: C 6 H 12 O 6 (Glucose): – 12: 6 – 2: 1

Monosaccharides: simple carbohydrates • • Consist of one sugar molecule: Fructose Glucose Galactose


DISACCHARIDES (made of 2 MONOSACCHARIDES) • MALTOSE: glucose + glucose • SUCROSE: glucose + fructose • LACTOSE: glucose + galactose

Remember… • -ose • Most simple carbs end in the letters -ose
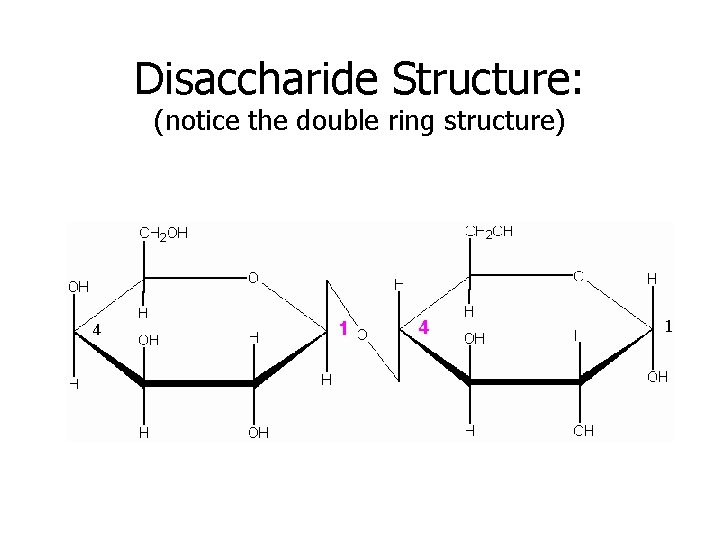
Disaccharide Structure: (notice the double ring structure)

POLYSACCHARIDES: complex carbohydrates • STARCH: Long chains of glucose – plants store sugar as starch (potato) • GLYCOGEN: chains of glucose (not as long) – animals store sugar as GLYCOGEN in the LIVER • CELLULOSE: long chains of polysaccharides that make up plant cell walls





CARBOHYDRATE TESTS *indicators • Benedict’s Solution: turns from a turquoise blue to orange/red (after heating) in the presence of a monosaccharide • Iodine: turns from a rust color to bluepurple-black in the presence of starch – a polysaccharide

Carbohydrate Indicator List Type of Original Color After Indicator Color Indicator is inserted Benedicts turquoise Orange/ solution Red (after heating) Iodine rust Purple/black solution Type of Carbohydrate: monosaccharide polysaccharide Note: a disaccharide will show no change with either indicator

Function of Carbohydrates: • Carbohydrates are an ideal source of energy for the body because they can be converted more readily into glucose, the form of sugar that is needed by the body. • Simple carbs contain 1 or 2 sugar molecules that are easily absorbed into the body and used for energy (they are readily broken down, although excess unused simple carbs turn into FAT!) • Complex carbs have larger chains of sugars that must be broken down before being used by the body for energy or stored (they take longer to break down and will give you sustained energy throughout the day)


LIPIDS: FATS, OILS, & WAXES


Why are lipids essential? 1. Energy storage for metabolism 2. Make up cell membrane structure & function 3. Fatty tissue helps regulate body temperature (insulation) 4. Protection of internal body organs 5. Necessary for hormone production in the body 6. 50% of our brain’s dry weight is lipid 7. Lipids are involved in cell signals 8. Transport fat soluble vitamins in the body
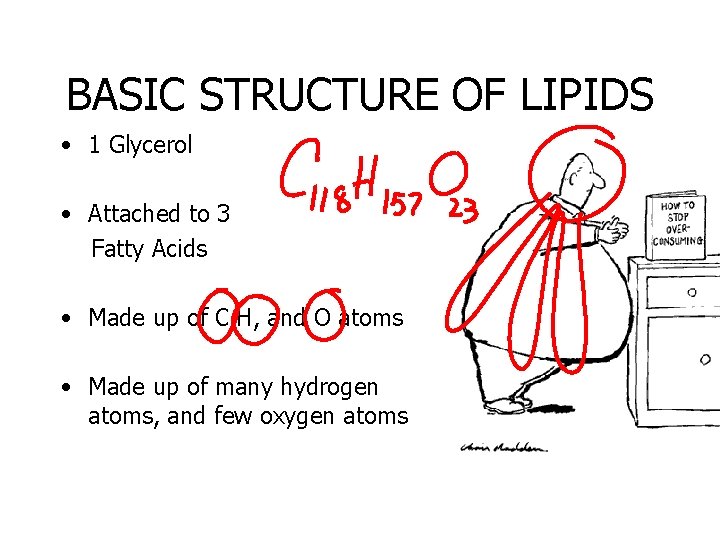
BASIC STRUCTURE OF LIPIDS • 1 Glycerol • Attached to 3 Fatty Acids • Made up of C, H, and O atoms • Made up of many hydrogen atoms, and few oxygen atoms

Dehydration synthesis Hydrolysis
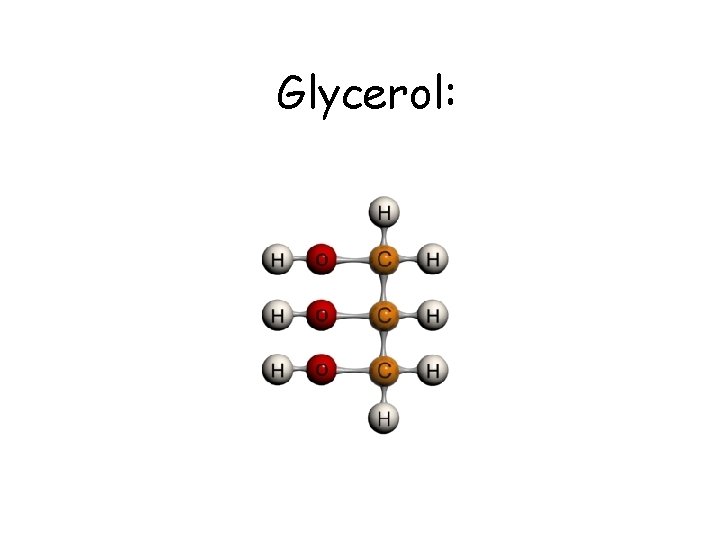
Glycerol:
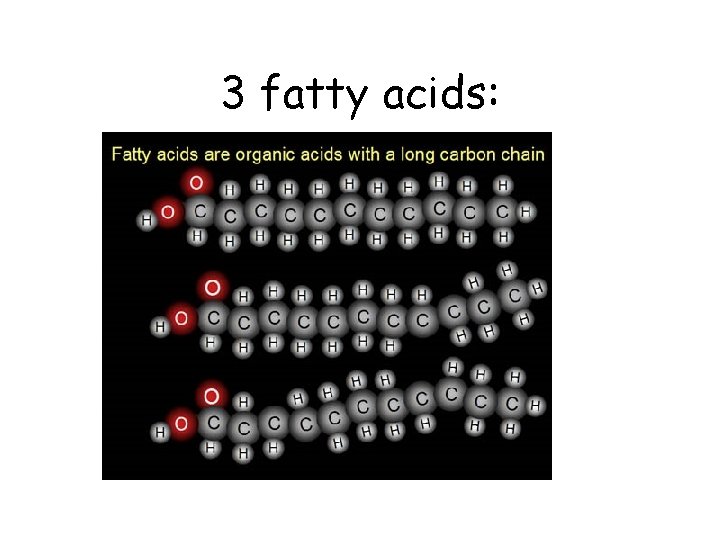
3 fatty acids:

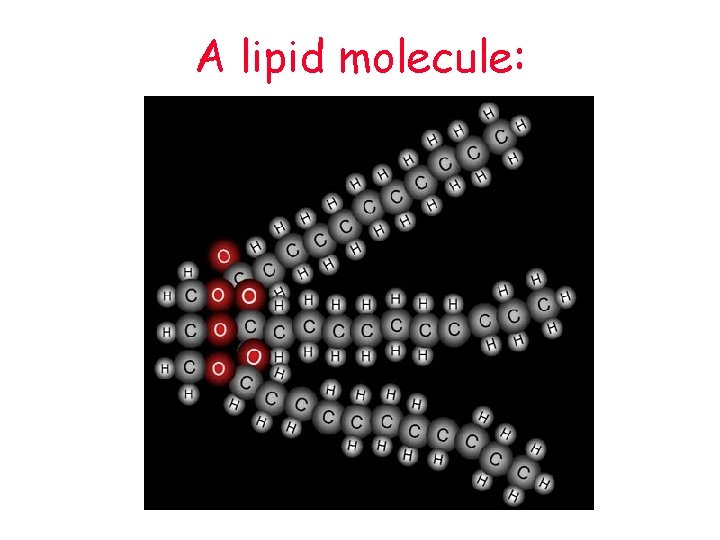
A lipid molecule:
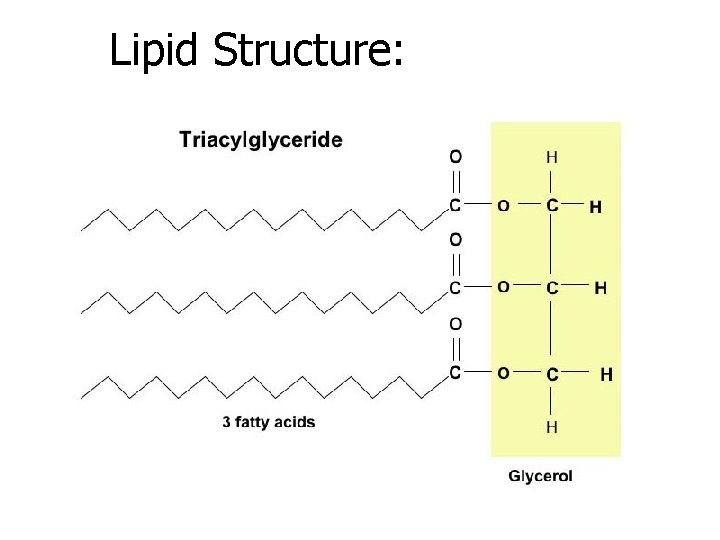
Lipid Structure:

Triglycerides • Triglycerides are the chemical form in which most fat exists in our bodies • They are the storage form of fat when we eat calories in excess of our energy needs • Burning up the stored fat allows us to live without food for periods of time • A high triglyceride level is not healthy!

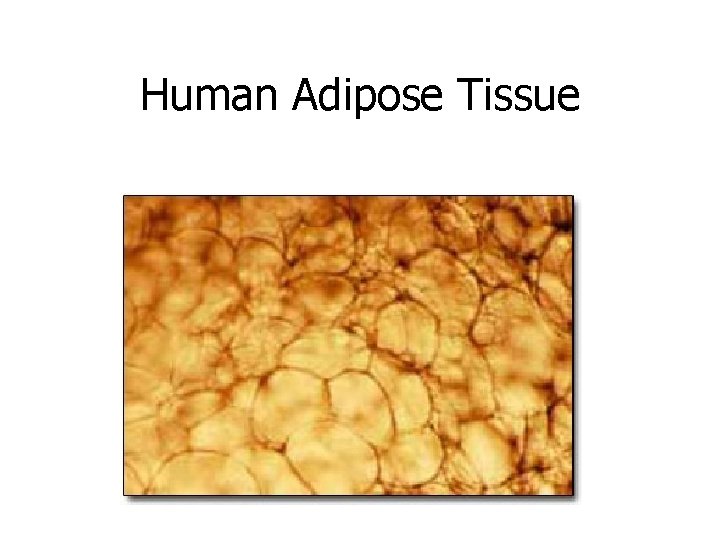
Human Adipose Tissue

Classification of Fats: 1. Saturated fats 2. Unsaturated fats (monounsaturated & polyunsaturated) 3. Trans fats

Saturated Fats • Formed by single bonds only (more difficult to break apart = stronger bonds)! • solid at room temperature • Derived from animal products (meat, dairy, eggs) • Lard, butter, coconut and palm oils • Clogs our arteries and directly raises LDL cholesterol levels

Unsaturated Fats • Formed by double bonds (easier to break apart = weaker bonds) • Derived from vegetables & plants • Two varieties: monounsaturated & polyunsaturated • Mono-unsaturated fats are the best fats to eat…they lower total cholesterol levels – Ex. Canola, & olive oils • Polyunsaturated fats: soybean oil, peanut, corn, & cottonseed oil

Trans Fats • Unsaturated fats that can raise total cholesterol levels • Trans fats - used to extend the shelf life of processed foods, typically cookies, cakes, fries and donuts • Any item that contains “hydrogenated oil” or “partially hydrogenated oil” likely contains trans fats • The tide is turning against trans fats…Since January 2006, all food manufacturers are required to list trans fat content on food labels.

Cholesterol • Soft, waxy substance in our bloodstream and in our cells • Used to form cell membranes and produce some hormones • High levels are a major risk for heart attack • Cannot dissolve in the blood so it must be transported to and from the cells by carriers called lipoproteins • LDL = bad cholesterol • HDL = good cholesterol

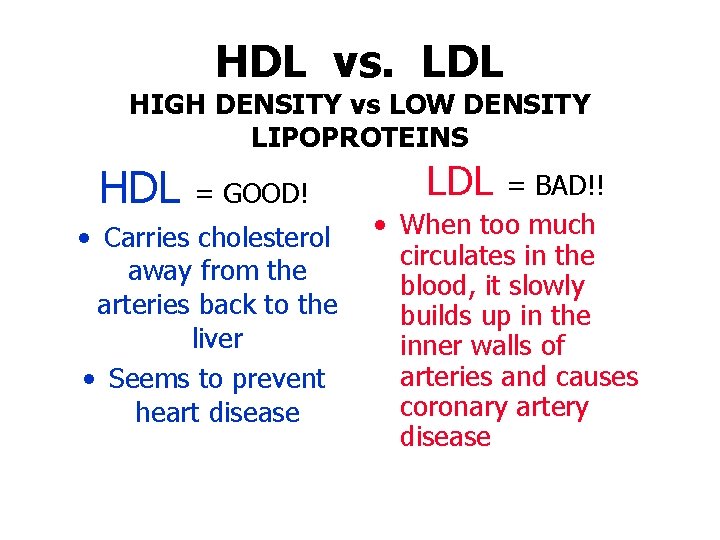
HDL vs. LDL HIGH DENSITY vs LOW DENSITY LIPOPROTEINS HDL = GOOD! • Carries cholesterol away from the arteries back to the liver • Seems to prevent heart disease LDL = BAD!! • When too much circulates in the blood, it slowly builds up in the inner walls of arteries and causes coronary artery disease


High Cholesterol Foods: • • Eggs Organ Meats (Liver, Brain) Red meat Whole-milk dairy products Butter Cream Ice cream






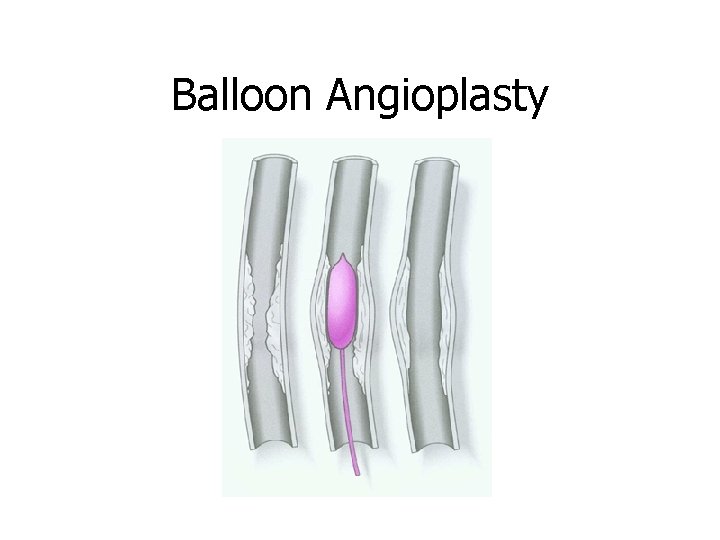
Balloon Angioplasty


A stent is made of stainless steel and can be inserted in the artery to keep it open


Proteins… The building blocks of life

Why is Protein important/FUNCTIONS? ? 1. Building blocks of the body – growth & repair of muscles, hair, aid in blood clotting, fingernails, skin, etc. 2. Create antibodies, (Immune System) 3. Make hormones (insulin, estrogen, testosterone) 4. Cell communication, (neurotransmitters) 5. Transport substances into and out of cells (proteins in the cell membrane) 6. Speed up reaction rate of metabolism ENZYMES!

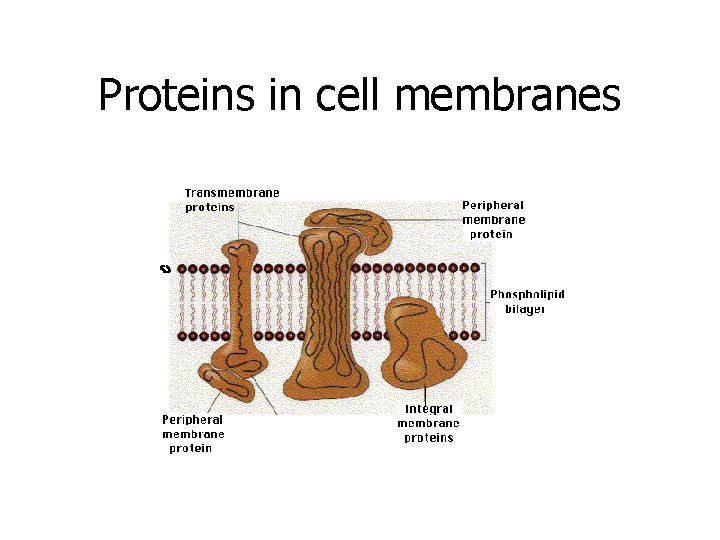
Proteins in cell membranes

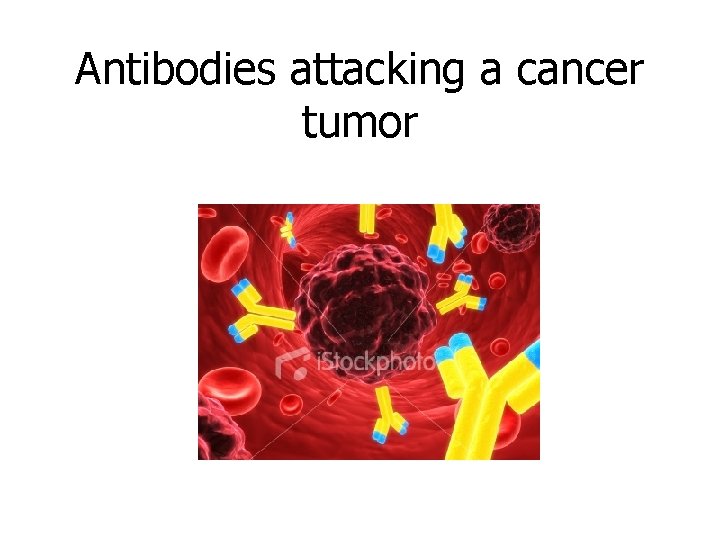
Antibodies attacking a cancer tumor
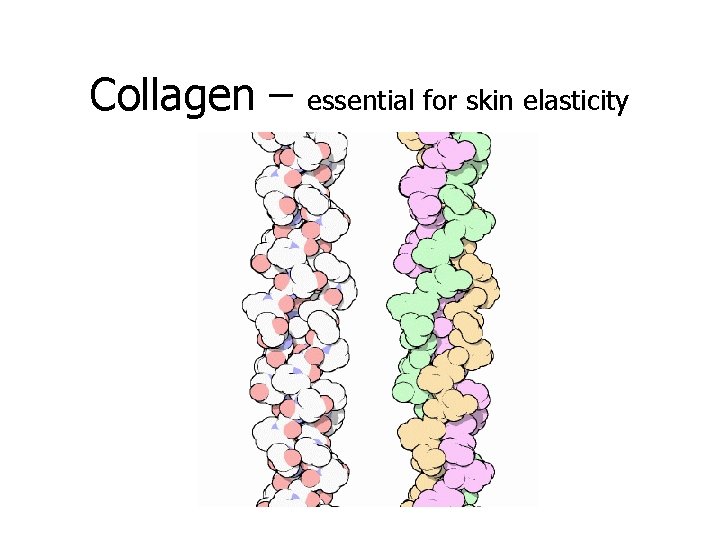
Collagen – essential for skin elasticity

Proteins = Muscle
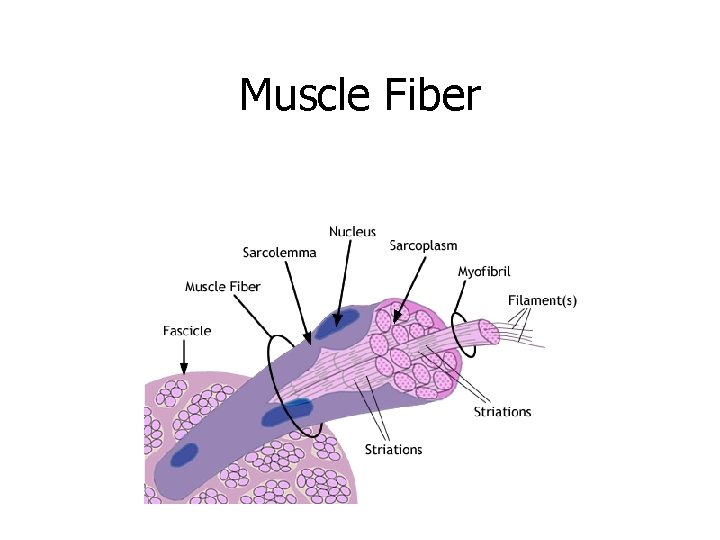
Muscle Fiber

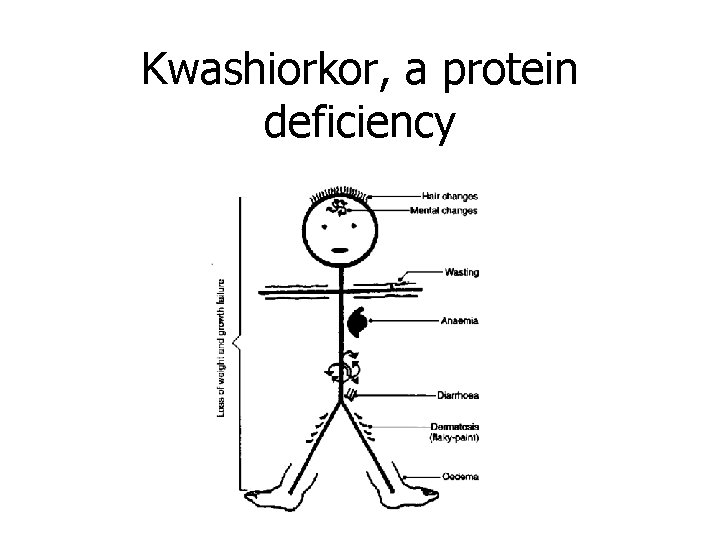
Kwashiorkor, a protein deficiency

• • • Symptoms of Kwashiorkor Swollen abdomen Failure to produce antibodies Failure to gain weight Decreased muscle mass swelling Lethargy Irritability Diarrhea Thin, brittle hair Changes in skin pigment Mortality rates as high as 60%


Why do starving children have fat bellies? • In Kwashiorkor, protein deficiency results in the body 'digesting' its own for energy. • Chronic malnutrition reduces the muscle tone of the abdominal muscles, so you get more protrusion of the abdomen (rounded, flaccid appearance of the belly). • Kids with Kwashiorkor may appear 'fat', but in reality their limbs are swollen with fluids from the ongoing processes. Also, they develop a swollen, fatty liver, which may add to the appearance.


PROTEIN • Proteins contain carbon, hydrogen, oxygen, nitrogen and sometimes sulfur, CHON(S) • The basic monomers of proteins are amino acids • Two amino acids joined together are called a dipeptide • Many amino acids joined together are called a polypeptide

PROTEIN STRUCTURE • Each polypeptide chain contains many arrangements of amino acids • The sequence of amino acids in a protein is what determines the shape of the protein • The shape of the protein determines the function of the protein • Sequence = Shape = FUNCTION of Protein!!!
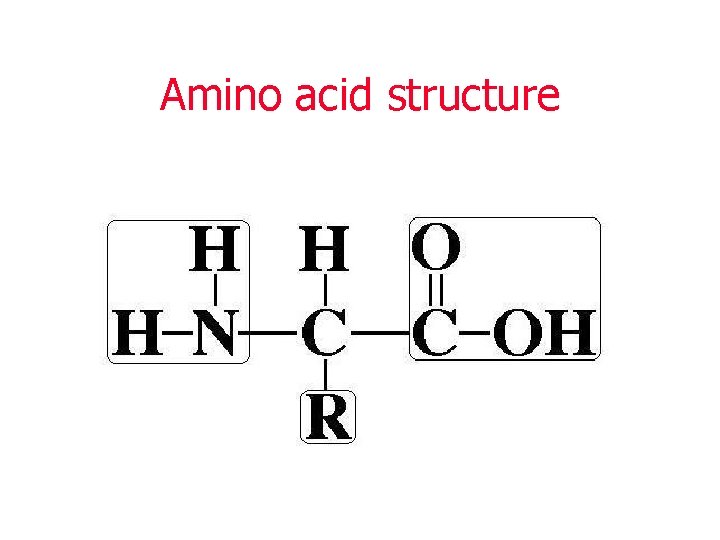
Amino acid structure

• There are 20 common amino acids (20 different “R” groups = variable groups) • In different combinations, they make thousands of proteins essential for life!

Protein Synthesis • These amino acids are joined together by peptide bonds • This happens in a reaction called dehydration synthesis • In dehydration synthesis, a water molecule is removed to link up amino acids to build a protein
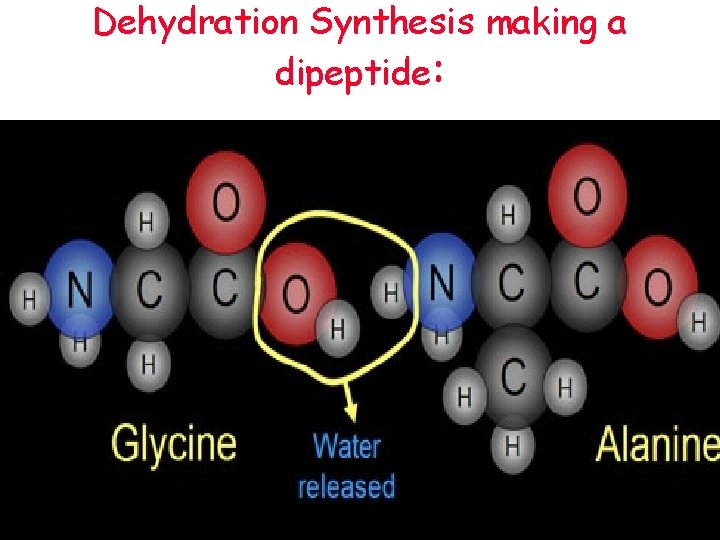
Dehydration Synthesis making a dipeptide:

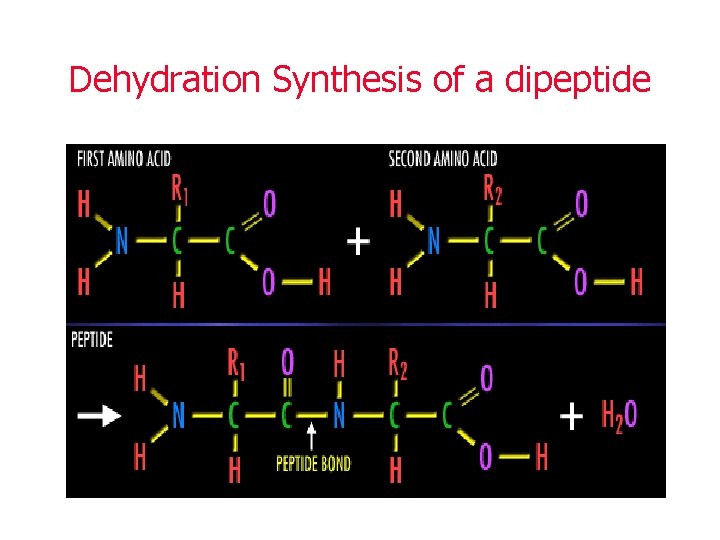
Dehydration Synthesis of a dipeptide

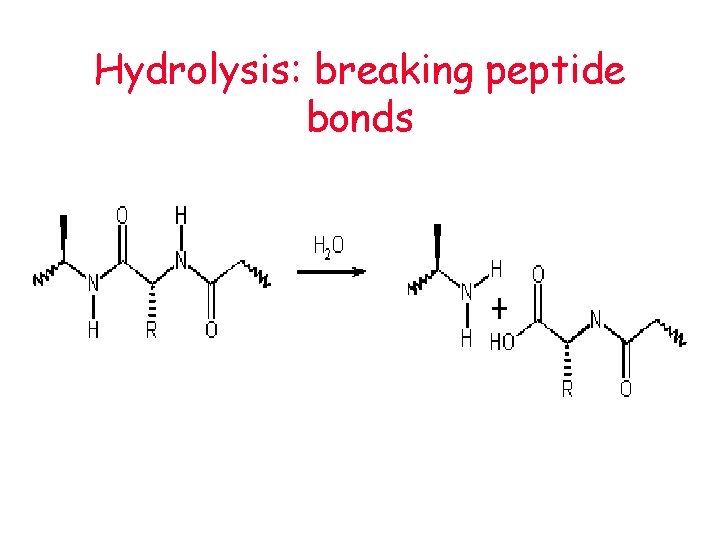
Hydrolysis: breaking peptide bonds

ENZYMES • • Enzymes are special proteins They are called catalysts Catalysts speed up chemical reactions Example: Salivary Amylase is an enzyme in our saliva that speeds up the initial breakdown of carbohydrates into glucose as we chew our food • Usually end in -ase

The enzymes that digest some molecules (break bonds) by hydrolysis are: – Maltase digests maltose – Sucrase digests sucrose – Lactase digests lactose – Lipase digests lipids


Lock & Key Model of Enzyme Activity: • The lock and key model of how an enzyme works shows us how they link up with substrates to form an enzymesubstrate complex • substrates/reactants ------ products • NOW MAKE AN ENZYME!
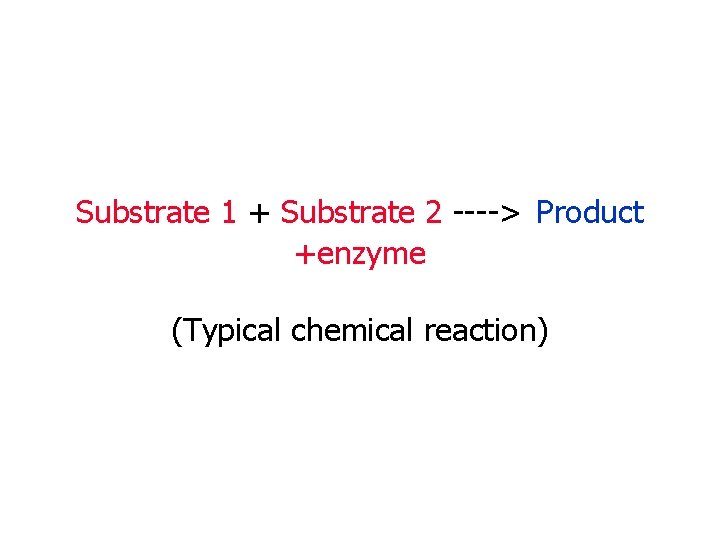
Substrate 1 + Substrate 2 ----> Product +enzyme (Typical chemical reaction)

Lock & Key Model of Enzyme Activity: • A substrate is a molecule that undergoes a chemical reaction • Each enzyme has an active site • which is a region on the surface of the enzyme where the substrate fits into (much like jigsaw puzzle pieces) • to form an enzyme-substrate complex


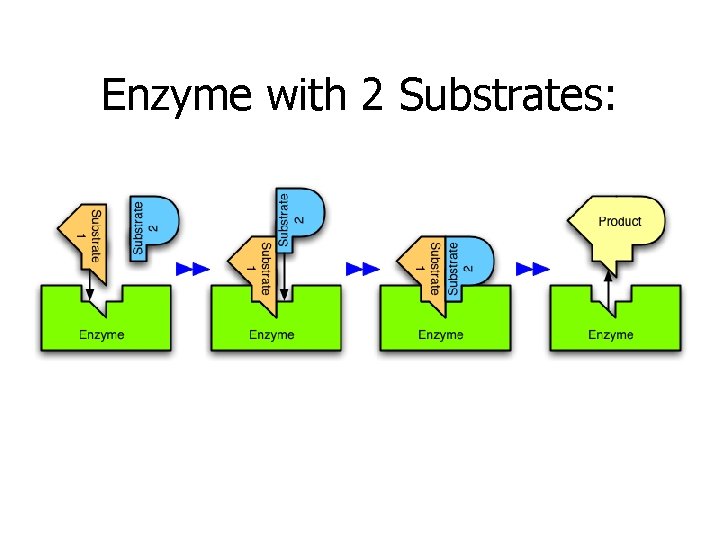
Enzyme with 2 Substrates:




Enzymes • Are involved in nearly all metabolic processes (digestion, absorption, synthesis, hydrolysis) • Are not changed by reactions • Are used again & again for the same chemical reaction • Enzymes are site specific (stomach vs. intestines) • Can become DENATURED if heated too much (change shape and cannot be used again)

Enzymes are affected by changes in : 1. 2. 3. 4. Temperature p. H Concentration of enzyme Concentration of substrate

Enzymes are affected by changes in : 1. Temperature • Enzyme action depends on the random motion of molecules • This motion increases as the temperature rises • Each enzyme works best at an ideal temperature
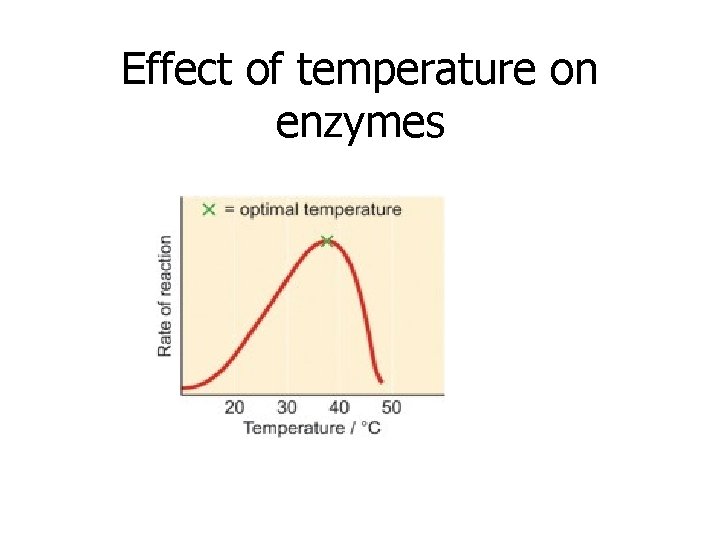
Effect of temperature on enzymes
2. p. H • Effectiveness of an enzyme depends on the p. H of the surrounding medium • Human stomach is acidic and pepsin (stomach enzyme) starts the digestion of proteins in the stomach • Therefore pepsin is more effective at lower p. H levels • Human intestines is basic (high p. H) • Intestinal enzyme trypsin works well in basic mediums
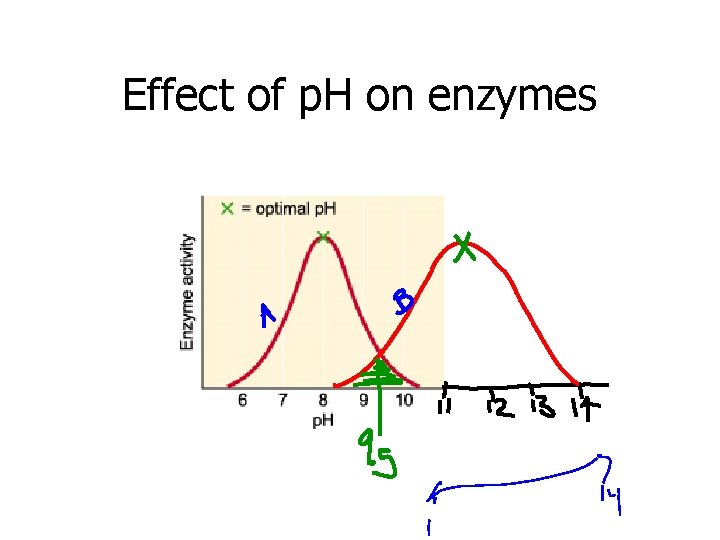
Effect of p. H on enzymes

3. Concentration of Enzyme • Adding more enzyme to a solution increases the # of substrate molecules that will react with that enzyme • Therefore reaction rate increases when more enzyme is added until all of the available substrate molecules are used up
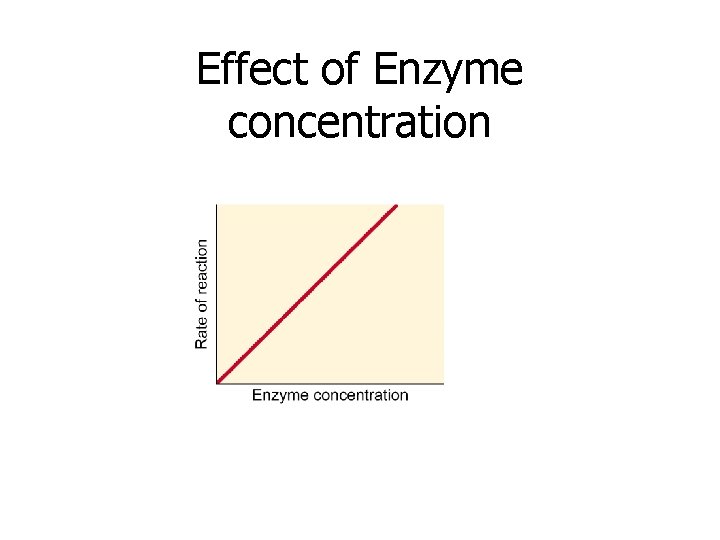
Effect of Enzyme concentration

4. Concentration of Substrate • Adding more substrate increases the reaction rate • until point of saturation is reached • This occurs when all enzyme molecules are occupied
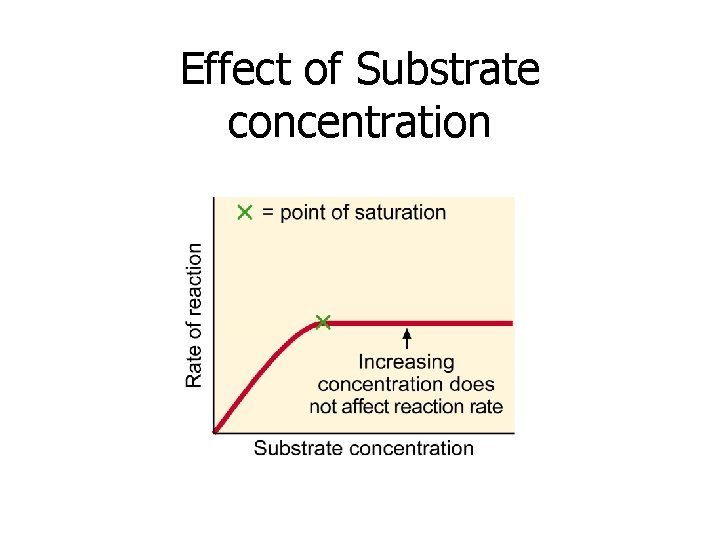
Effect of Substrate concentration

Facts about protein: • Protein is what makes up your ligaments, tendons, muscles, hair, nails, skin, teeth, tissue, organs, and bones. • Besides water, proteins are the most plentiful substance in the body!!! • Since most of the body's proteins are continually broken down, the body needs to manufacture thousands of proteins every day to replace them

Some vital proteins in our bodies: • • Actin & myosin allows our muscles to contract Collagen in our skin gives it elasticity Cardiac muscle allows our heart to pump Digestive muscles allow food to pass through our system • Blood vessels, organs, & skin have structural proteins • Hemoglobin transports O 2 from the lungs to the rest of the body (via red blood cells)

Best sources of protein • • • Beef Poultry Fish Eggs dairy products Nuts, seeds, legumes

How much protein do we need? • About 0. 4 gram per pound of body weight • That is how many grams of protein you need per day • The more active you are, the more protein your body needs to repair body cells and make new ones

Straight talk about soy… • One protein source that has been getting a lot of attention is soybeans. • Regularly eating soy-based foods lowers cholesterol, prevents breast and prostate cancer, aids weight loss, and wards off osteoporosis. • This is because soybeans have a high concentration of isoflavones, a type of plant-made estrogen (phytoestrogen). • Food makers are churning out new soy products that are moving into the mainstream. • In Boston, for example, soymilk is now advertised on the radio during Boston Red Sox games, alongside donuts, and oil additives.

NUCLEIC ACIDS • TWO TYPES: DNA & RNA • DNA - the hereditary information passed on from parent to offspring • RNA- Carries the genetic code for amino acids – puts the machinery to work in your body so that proteins can be synthesized – Carries the genetic code from the nucleus to the ribosomes (where proteins are made)
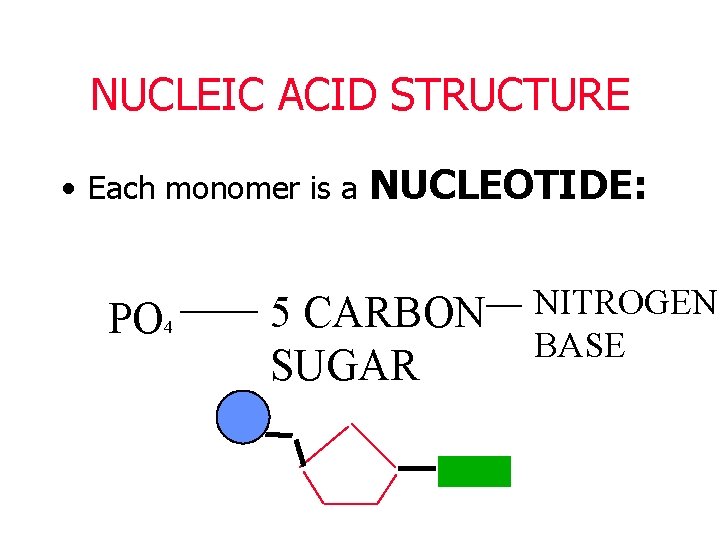
NUCLEIC ACID STRUCTURE • Each monomer is a PO 4 NUCLEOTIDE: 5 CARBON SUGAR NITROGEN BASE

Nucleotides consist of: 1. Phosphate group – (PO 4) 2. 5 -carbon sugar (deoxyribose) 3. Nitrogen base (A T, C, G)
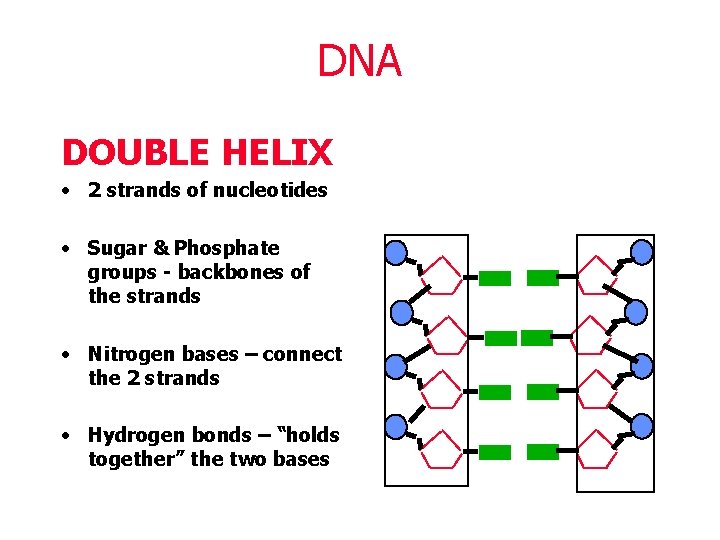
DNA DOUBLE HELIX • 2 strands of nucleotides • Sugar & Phosphate groups - backbones of the strands • Nitrogen bases – connect the 2 strands • Hydrogen bonds – “holds together” the two bases

• DNA – (deoxyribonucleic acid) - the hereditary material in living organisms • Procaryotes – DNA is in middle of the cell (nucleoid) • Eucaryotes – DNA is in the nucleus • Genetic information stored in a code made up of four chemical bases: adenine (A), guanine (G), cytosine (C), and thymine (T) • Human DNA consists of about 3 billion bases, and more than 99% of those bases are the same in all people.

Procaryote DNA


• Order of the bases form a code that directs a blueprint for how an organism is built • DNA does this be directing which proteins to make and where to place them • DNA is like an instruction manual for an organism!!! • Complementary Nitrogen base pairs: • A–T • C-G




Let’s review…


Are you ready for your biochemistry quiz?
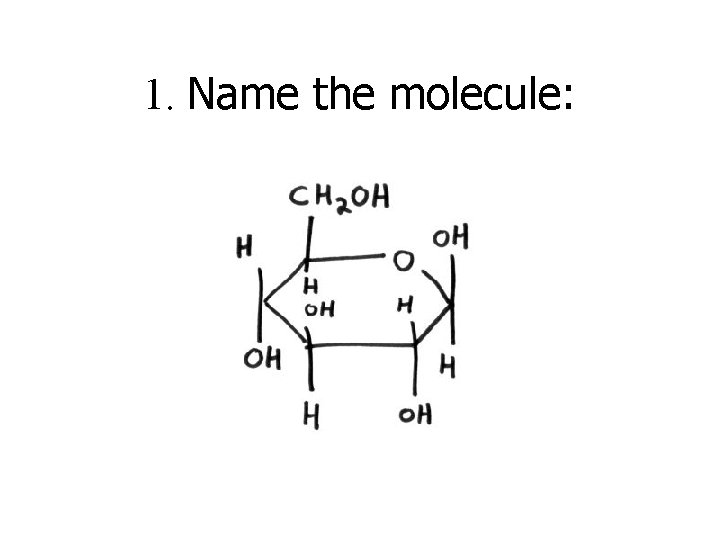
1. Name the molecule:
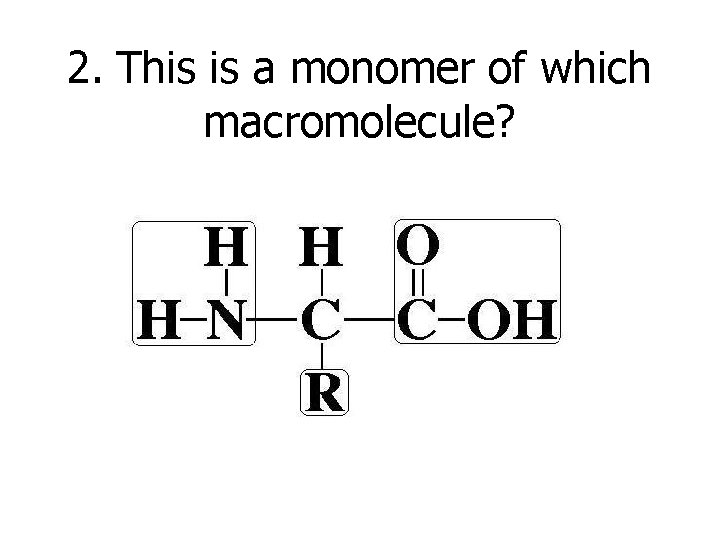
2. This is a monomer of which macromolecule?
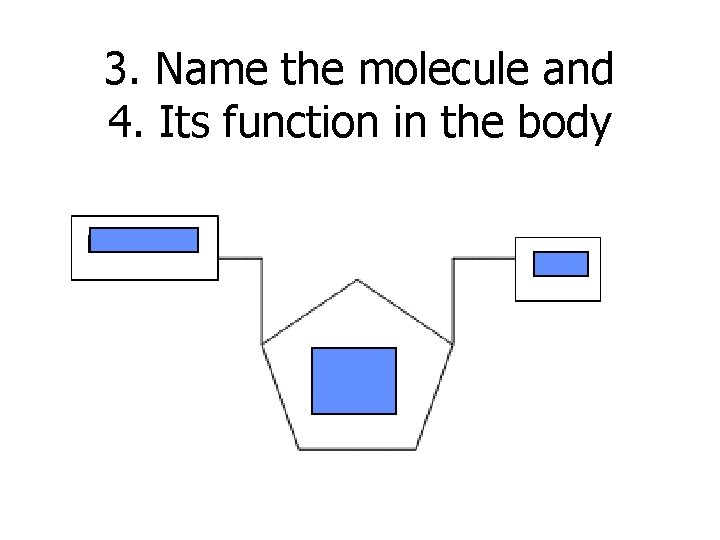
3. Name the molecule and 4. Its function in the body
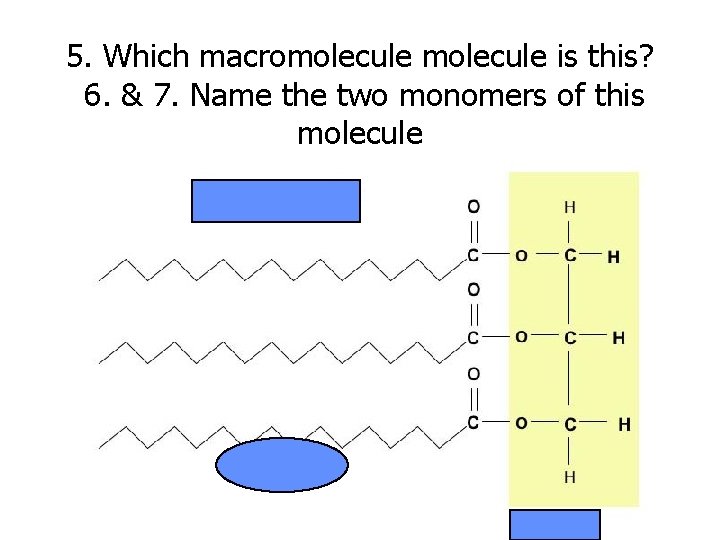
5. Which macromolecule is this? 6. & 7. Name the two monomers of this molecule
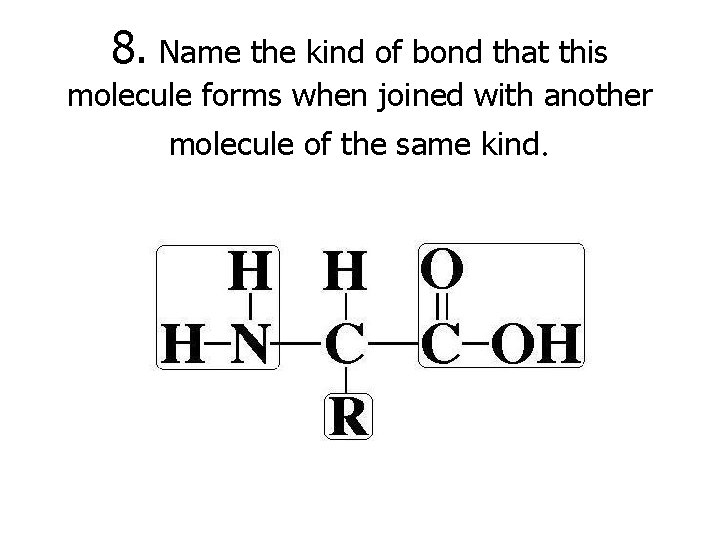
8. Name the kind of bond that this molecule forms when joined with another molecule of the same kind.
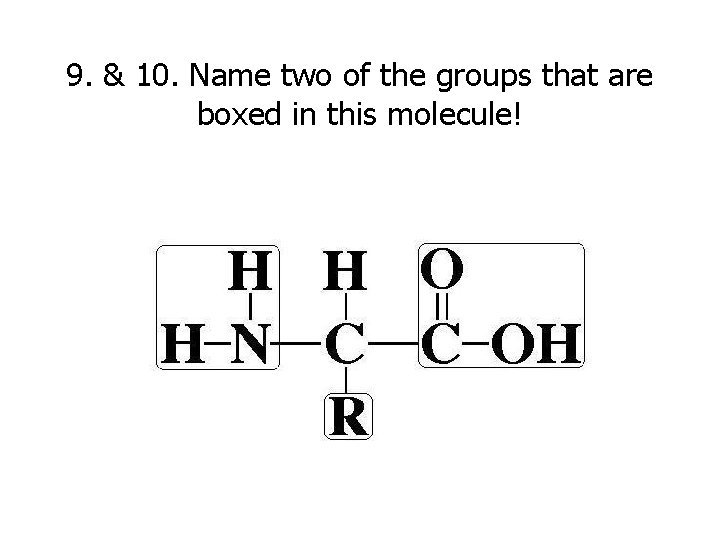
9. & 10. Name two of the groups that are boxed in this molecule!
- Slides: 163