BIOCHEMISTRY AP Biology Life requires 25 elements u

BIOCHEMISTRY AP Biology

Life requires ~25 elements u u 4 elements make up 96% of living matter: • carbon (C) • hydrogen (H) • oxygen (O) • nitrogen (N) 4 elements make up most of remaining 4%: AP Biology • phosphorus (P) • calcium (Ca) • sulfur (S) • potassium (K)

Chemical reactivity § Atoms tend to complete a partially filled valence shell or u empty a partially filled valence shell u This tendency drives chemical reactions… and creates bonds – – – AP Biology
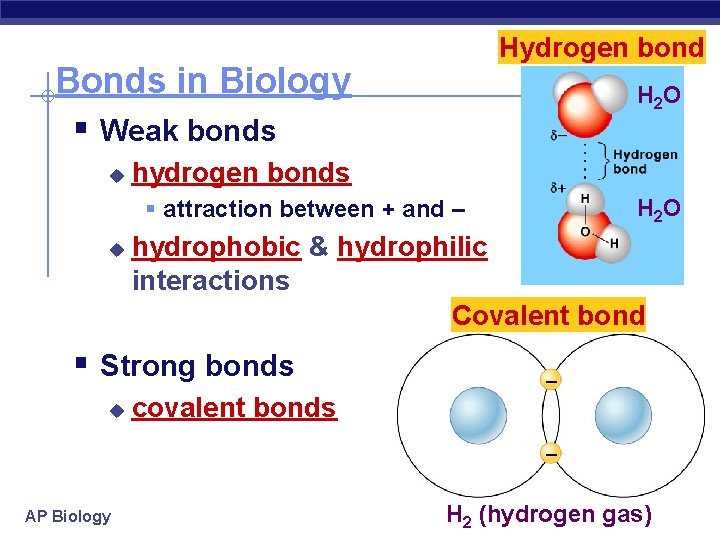
Hydrogen bond Bonds in Biology § Weak bonds u H 2 O hydrogen bonds H 2 O § attraction between + and – u hydrophobic & hydrophilic interactions Covalent bond § Strong bonds u – covalent bonds – AP Biology H 2 (hydrogen gas)
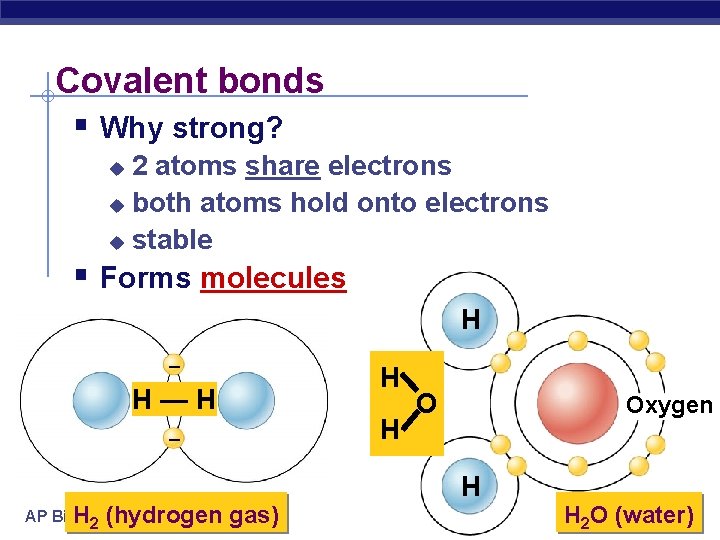
Covalent bonds § Why strong? 2 atoms share electrons u both atoms hold onto electrons u stable u § Forms molecules H – H H—H H – AP Biology H 2 (hydrogen gas) O Oxygen H H 2 O (water)
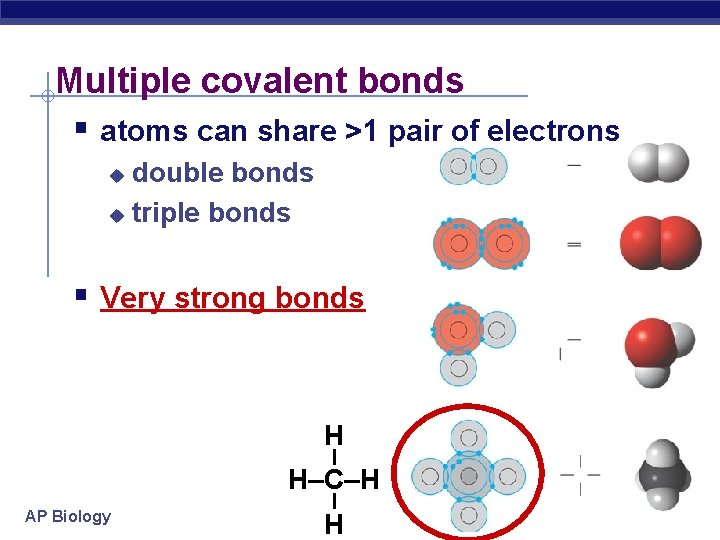
Multiple covalent bonds § atoms can share >1 pair of electrons double bonds u triple bonds u § Very strong bonds – H AP Biology – H–C–H H

Why study CARBON? § All of life is built on carbon § Cells ~72% H 2 O u ~25% carbon compounds u § § u AP Biology carbohydrates lipids proteins nucleic acids ~3% salts § Na, Cl, K…
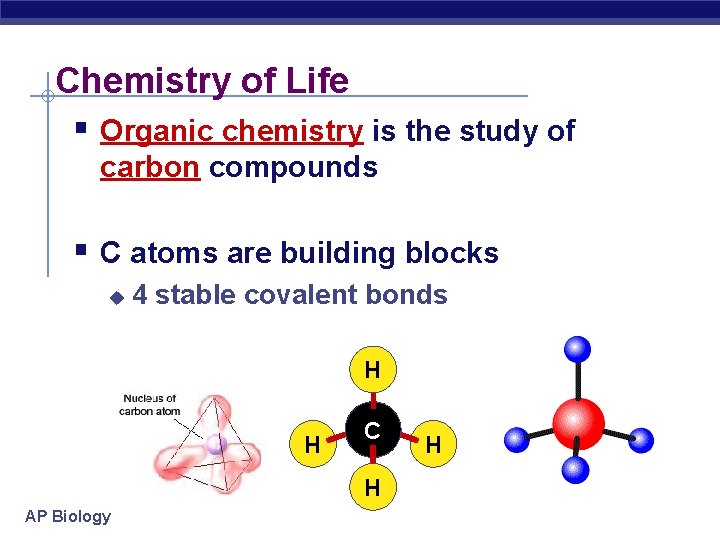
Chemistry of Life § Organic chemistry is the study of carbon compounds § C atoms are building blocks u 4 stable covalent bonds H H C H AP Biology H

Hydrocarbons § Combinations of C & H u non-polar § not soluble in H 2 O § hydrophobic methane (simplest HC) stable u very little attraction between molecules u AP Biology
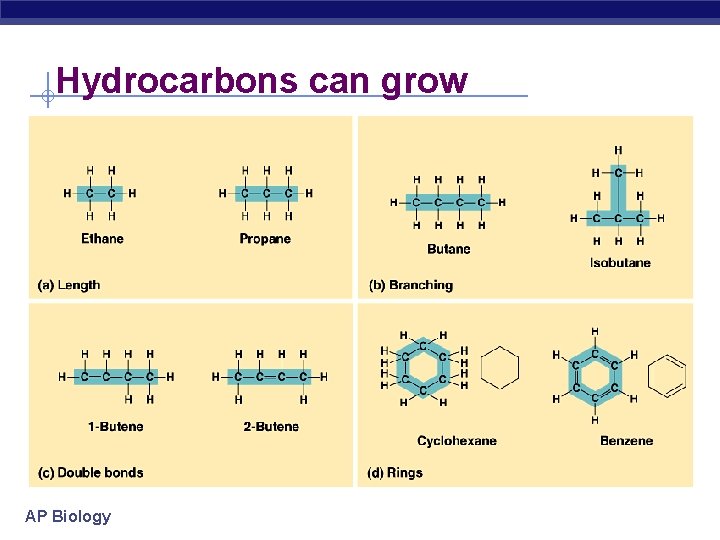
Hydrocarbons can grow AP Biology
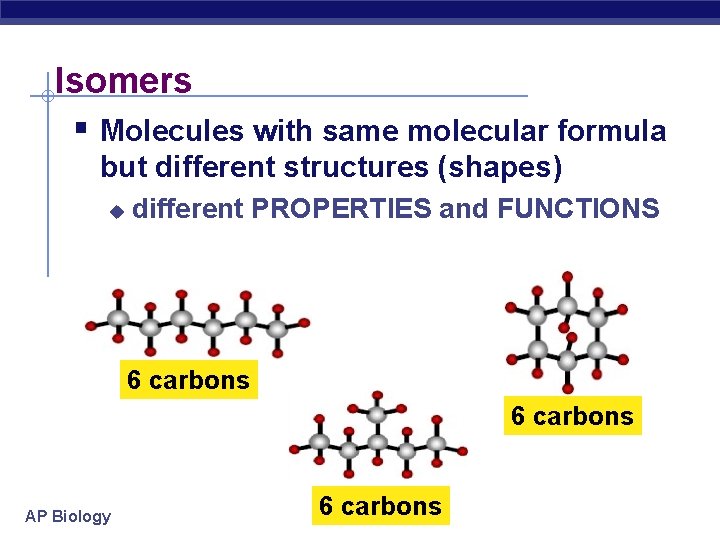
Isomers § Molecules with same molecular formula but different structures (shapes) u different PROPERTIES and FUNCTIONS 6 carbons AP Biology 6 carbons

Functional groups § parts of organic molecules that are involved in chemical reactions u give organic molecules distinct properties hydroxyl carbonyl carboxyl amino sulfhydryl phosphate § affect reactivity makes hydrocarbons hydrophilic u increase solubility in water u AP Biology

Viva la difference! § Basic structure of male & female hormones is identical u u AP Biology identical carbon skeleton attachment of different functional groups interact with different targets in the body = different effects

Hydroxyl § –OH alcohols u names typically end in –ol u Example = ethanol u AP Biology

Carbonyl § C=O u O double bonded to C § if C=O at end molecule = aldehyde § if C=O in middle of molecule = ketone AP Biology
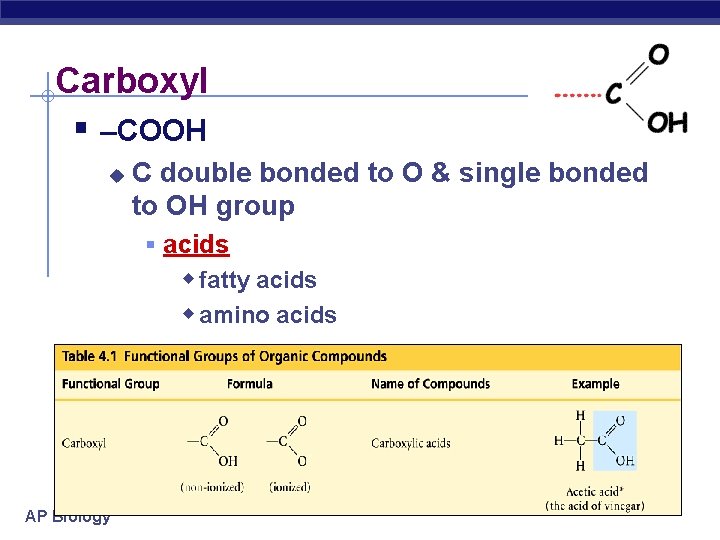
Carboxyl § –COOH u C double bonded to O & single bonded to OH group § acids w fatty acids w amino acids AP Biology
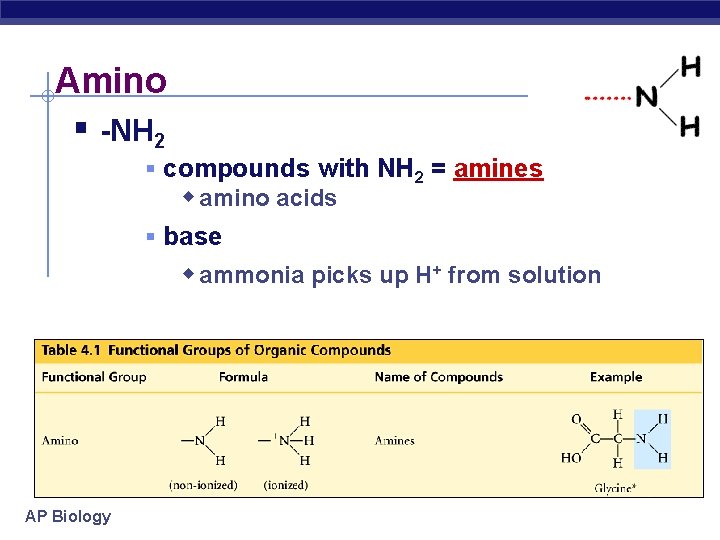
Amino § -NH 2 § compounds with NH 2 = amines w amino acids § base w ammonia picks up H+ from solution AP Biology

Sulfhydryl § –SH u S bonded to H § compounds with SH = thiols § SH groups stabilize the structure of proteins AP Biology

Phosphate § –PO 4 u P bound to 4 O § connects to C through an O § lots of O = lots of negative charge w highly reactive § transfers energy between organic molecules w ATP, GTP, etc. AP Biology
- Slides: 19