Biochemistry 2e Garrett Grisham CHAPTER 2 Water p

Biochemistry 2/e - Garrett & Grisham CHAPTER 2 Water, p. H, and Ionic Equilibria to accompany Biochemistry, 2/e by Reginald Garrett and Charles Grisham All rights reserved. Requests for permission to make copies of any part of the work should be mailed to: Permissions Department, Harcourt Brace & Company, 6277 Sea Harbor Drive, Orlando, Florida 32887 -6777 Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Outline • • 2. 1 Properties of Water 2. 2 p. H 2. 3 Buffers 2. 4 Water's Unique Role in the Fitness of the Environment Copyright © 1999 by Harcourt Brace & Company

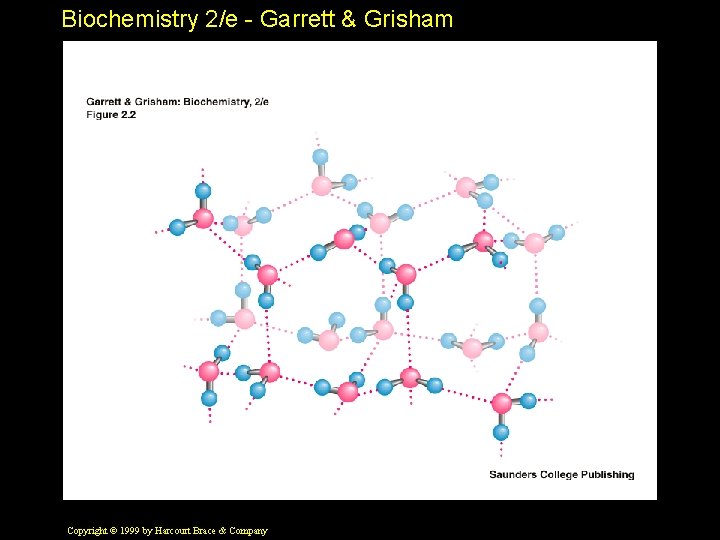
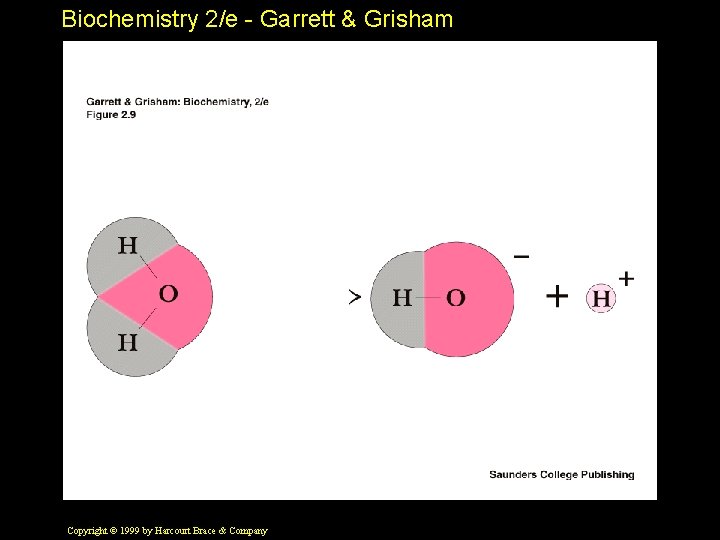
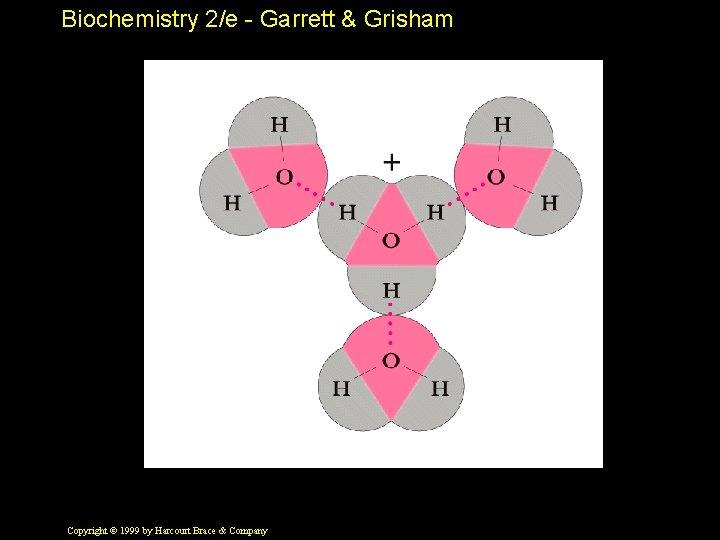
Biochemistry 2/e - Garrett & Grisham Properties of Water • High b. p. , m. p. , heat of vaporization, surface tension • Bent structure makes it polar • Non-tetrahedral bond angles • H-bond donor and acceptor • Potential to form four H-bonds per water Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Comparison of Ice and Water • • • Issues: H-bonds and Motion Ice: 4 H-bonds per water molecule Water: 2. 3 H-bonds per water molecule Ice: H-bond lifetime - about 10 microsec Water: H-bond lifetime - about 10 psec (10 psec = 0. 000001 sec) Thats "one times ten to the minus eleven second"! Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

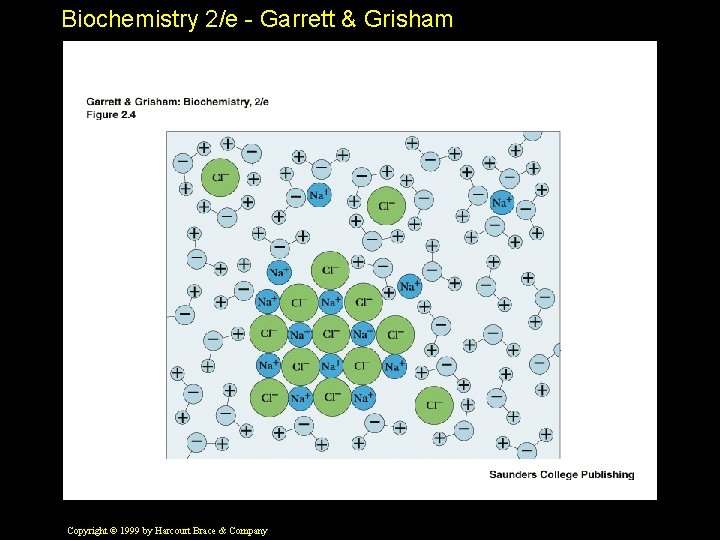
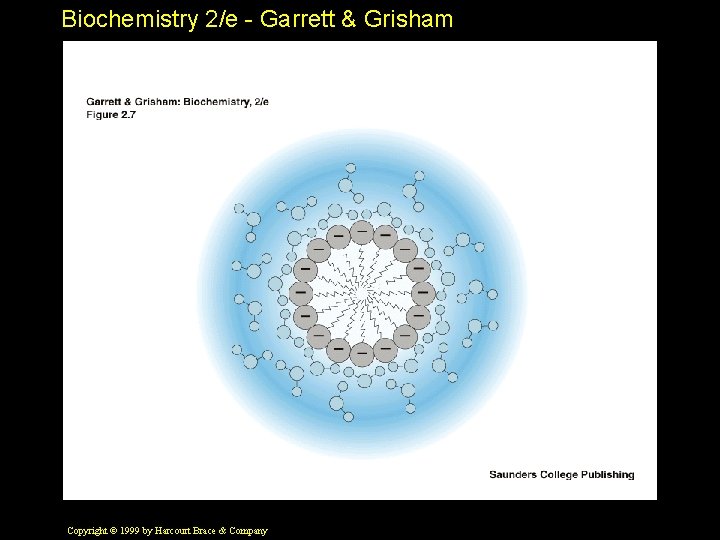
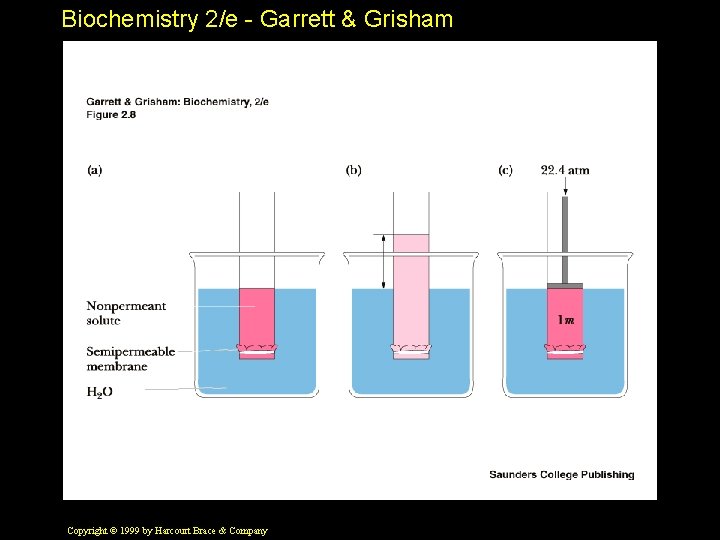
Biochemistry 2/e - Garrett & Grisham Solvent Properties of Water • Ions are always hydrated in water and carry around a "hydration shell" • Water forms H-bonds with polar solutes • Hydrophobic interactions - a "secret of life" Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

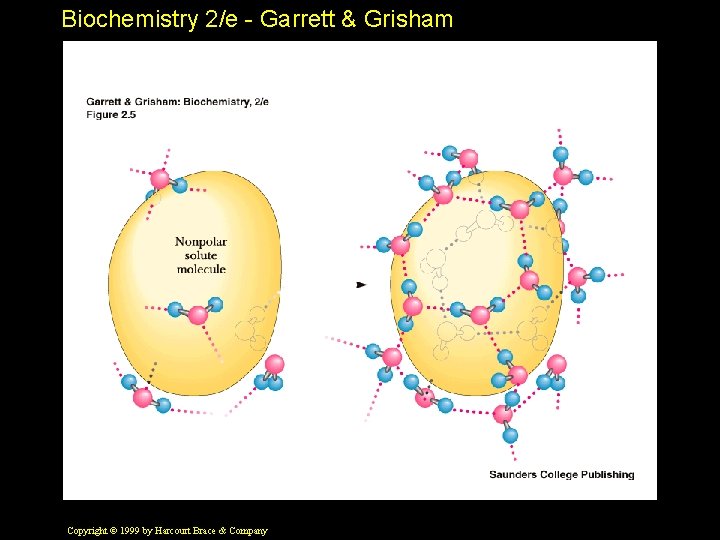
Biochemistry 2/e - Garrett & Grisham Hydrophobic Interactions • A nonpolar solute "organizes" water • The H-bond network of water reorganizes to accommodate the nonpolar solute • This is an increase in "order" of water • This is a decrease in ENTROPY Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Amphiphilic Molecules Also called "amphipathic" • Refers to molecules that contain both polar and nonpolar groups • Equivalently - to molecules that are attracted to both polar and nonpolar environments • Good examples - fatty acids Copyright © 1999 by Harcourt Brace & Company
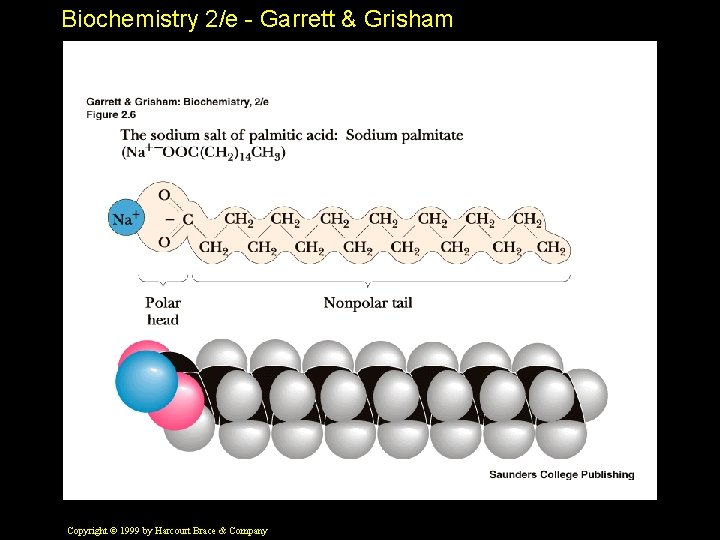
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Acid-base Equilibria • • • The p. H Scale A convenient means of writing small concentrations: p. H = -log 10 [H+] Sørensen (Denmark) If [H+] = 1 x 10 -7 M Then p. H = 7 Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

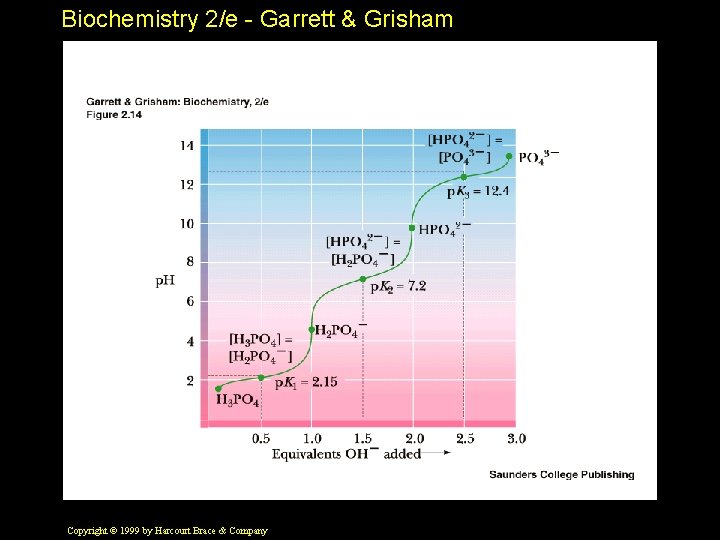
Biochemistry 2/e - Garrett & Grisham Dissociation of Weak Electrolytes Consider a weak acid, HA • The acid dissociation constant is given by: • HA H+ + A • Ka = [ H + ] [ A - ] __________ [HA] Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham The Henderson-Hasselbalch Equation Know this! You'll use it constantly. • For any acid HA, the relationship between the p. Ka, the concentrations existing at equilibrium and the solution p. H is given by: • p. H = p. Ka + log 10 [A¯ ] ¯¯¯¯¯ [HA] Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

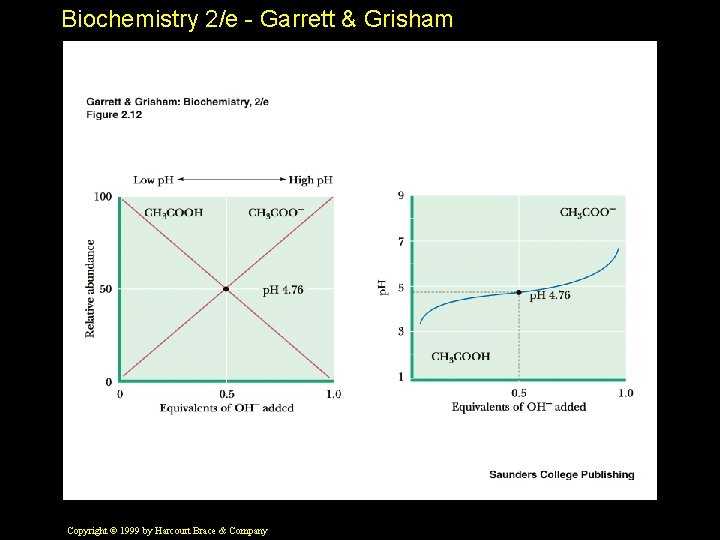
Biochemistry 2/e - Garrett & Grisham Consider the Dissociation of Acetic Acid Assume 0. 1 eq base has been added to a fully protonated solution of acetic acid • The Henderson-Hasselbalch equation can be used to calculate the p. H of the solution: With 0. 1 eq OH¯ added: • p. H = p. Ka + log 10 [0. 1 ] ¯¯¯¯¯ [0. 9] • p. H = 4. 76 + (-0. 95) • p. H = 3. 81 Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Consider the Dissociation of Acetic Acid Another case. . • What happens if exactly 0. 5 eq of base is added to a solution of the fully protonated acetic acid? • With 0. 5 eq OH¯ added: • p. H = p. Ka + log 10 [0. 5 ] ¯¯¯¯¯ [0. 5] • p. H = 4. 76 + 0 • p. H = 4. 76 = p. Ka Copyright © 1999 by Harcourt Brace & Company

Biochemistry 2/e - Garrett & Grisham Consider the Dissociation of Acetic Acid A final case to consider. . • What is the p. H if 0. 9 eq of base is added to a solution of the fully protonated acid? • With 0. 9 eq OH¯ added: • p. H = p. Ka + log 10 [0. 9 ] ¯¯¯¯¯ [0. 1] • p. H = 4. 76 + 0. 95 • p. H = 5. 71 Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company

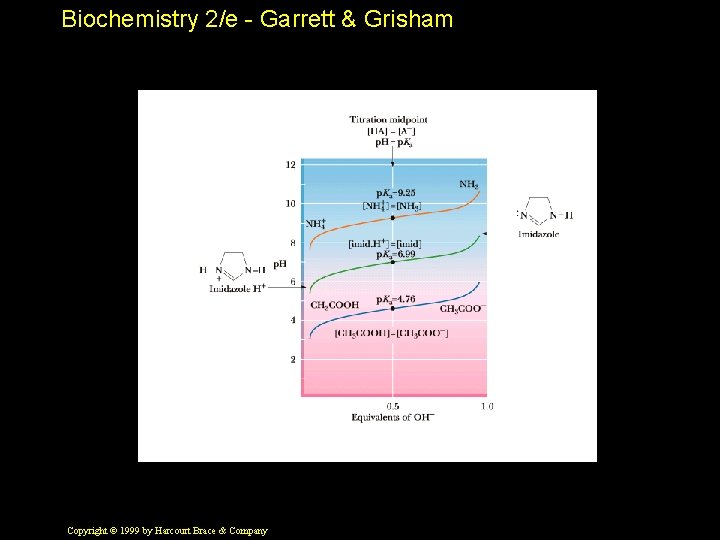
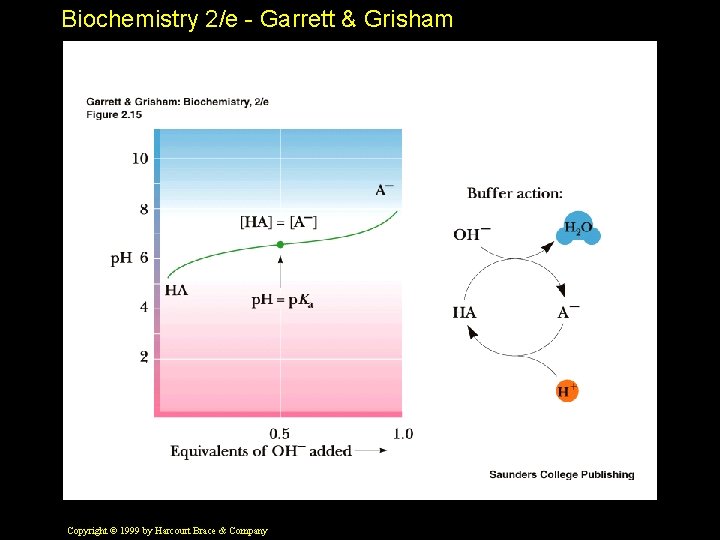
Biochemistry 2/e - Garrett & Grisham Buffers • Buffers are solutions that resist changes in p. H as acid and base are added • Most buffers consist of a weak acid and its conjugate base • Note in Figure 2. 15 how the plot of p. H versus base added is flat near the p. Ka • Buffers can only be used reliably within a p. H unit of their p. Ka Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
Biochemistry 2/e - Garrett & Grisham Copyright © 1999 by Harcourt Brace & Company
- Slides: 32